Why is chloroform used in NMR
 John Peck
John Peck Deuterated chloroform is a deuterated compound that is an isotopologue of chloroform in which the hydrogen atom is replaced with deuterium. Commonly used as a solvent in proton NMR spectroscopy. It has a role as a non-polar solvent. It is a deuterated compound and a member of chloromethanes.
Why is CDCl3 used in NMR?
In proton NMR spectroscopy, deuterated solvent (enriched to >99% deuterium) must be used to avoid recording a large interfering signal or signals from the proton(s) (i.e., hydrogen-1) present in the solvent itself.
Why CDCl3 is as a solvent in NMR spectroscopy instead of CHCl3?
Most 1H – NMR spectra are therefore recorded in a deuterated solvent, because deuterium atoms absorb at a completely different frequency. But deuteration is never complete, so in CDCl3 , for example, there is always some residual CHCl3 . You always get a solvent signal from CHCl3 at 7.26 ppm.
Can CHCl3 used in NMR spectroscopy?
The most widely used example of such a solvent is CDCl3 (chloroform-d, or “deuterochloro- form”), the deuterium analog of chloroform, CHCl3. This solvent is so widely used for NMR spectra that it is a relatively inexpensive article of commerce. Most of the spectra in this text were taken in CDCl3.What kind of solvent is suitable for NMR spectroscopy?
Only the deutero-solvents are suitable for 1H NMR spectroscopy .
Why are deuterated solvents needed in NMR?
Expensive deuterated solvents have traditionally been used for NMR spectroscopy in order to facilitate locking and shimming, as well as to suppress the large solvent signal that would otherwise occur in the proton NMR spectrum.
Is CDCl3 chloroform?
Deuterated chloroform (CDCl3), also known as chloroform-d, is an isotopically enriched form of chloroform (CHCl3) in which most of its hydrogen atoms consist of the heavier nuclide deuterium (heavy hydrogen) (D = 2H) rather than the natural isotopic mixture in which protium (1H) is predominant.
Which solvent is not used in NMR spectroscopy?
To avoid spectra dominated by the solvent signal, most 1H NMR spectra are recorded in a deuterated solvent. However, deuteration is not “100%”, so signals for the residual protons are observed. In chloroform solvent (CDCl3), this corresponds to CHCl3, so a singlet signal is observed at 7.26 ppm.What is chloroform used for?
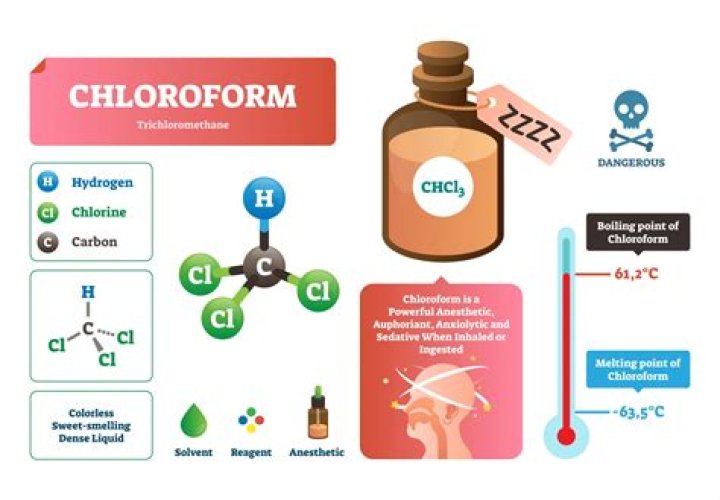
Chloroform is used as a solvent, a substance that helps other substances dissolve. Also, it is used in the building, paper and board industries, and in pesticide and film production. It is used as a solvent for lacquers, floor polishes, resins, adhesives, alkaloids, fats, oils and rubber.
What is chemical shift in NMR spectroscopy?In nuclear magnetic resonance (NMR) spectroscopy, the chemical shift is the resonant frequency of a nucleus relative to a standard in a magnetic field. Often the position and number of chemical shifts are diagnostic of the structure of a molecule. … This is reflected in the spin energy levels (and resonance frequencies).
Article first time published onHow do you neutralize deuterated chloroform?
Testing Deuterated Chloroform Acidity be easily neutralized using the following procedure: Place 3-5 grams of a 5Å molecular sieves into a 50 or 100 g bottle of the solvent. Swirl slightly and allow to stand overnight. Excess water and traces of acidity will be removed.
Is CDCl3 soluble in water?
NamesBoiling point61.15 °C (142.07 °F; 334.30 K) decomposes at 450 °CSolubility in water10.62 g/L (0 °C) 8.09 g/L (20 °C) 7.32 g/L (60 °C)SolubilitySoluble in benzene Miscible in diethyl ether, oils, ligroin, alcohol, CCl4, CS2Solubility in acetone≥ 100 g/L (19 °C)
How is deuterated chloroform made?
Through the utilization of the polarization and weakening effects of connecting bond of the carbonyl carbon of the hexachloroacetone and trichloromethyl, the carbonyl carbon of the hexachloroacetone and trichloromethyl are hydrolyzed in an alkalescence condition to produce deuterated chloroform, and the reaction …
What is shielding and Deshielding in NMR?
On Professor Hardinger’s website, shielded is defined as “a nucleus whose chemical shift has been decreased due to addition of electron density, magnetic induction, or other effects.” What is Deshielding? Downfield The Nucleus feels stronger magnetic field. Deshielding is the opposite of shielding.
What is relaxation process in NMR?
NMR relaxation is the processes by which an excited magnetic state returns to its equilibrium distribution. NMR relaxation measurement can be used for spectral assignment and the study of quadrupolar and paramagnetic interactions, and exchange dynamics.
Is chloroform polar or nonpolar?
Chloroform the solvent is “nonpolar” because it has a low dielectric constant. The polarity of a solvent is highly dependent on its dielectric constant with high dielectric constants correlating with polar solvents and low dielectric constants correlating with nonpolar solvents.
What means deuterated?
deuterated in American English (ˈdutərˌeɪtɪd ; ˈdjutərˌeɪtɪd ) adjective. designating or of a substance, compound, or organism in which part or all of the normal hydrogen atoms are replaced with deuterium. containing deuterium.
What is meant by deuterated solvents?
Deuterated solvents are a group of compounds where one or more hydrogen atoms are substituted by deuterium atoms. These compounds are often used in Nuclear magnetic resonance spectroscopy.
Why 12C is NMR inactive?
There is no NMR spin capability with 12C (6 protons and 6 neutrons). There must be an odd number of a) protons, b) neutrons, or c) both in order for there to be spin activity and thus NMR detectability.
Is deutero chloroform toxic?
Inhalation May be harmful if inhaled. Causes respiratory tract irritation. Skin Harmful if absorbed through skin. … Eyes Causes eye irritation.
What is the effect of chloroform?
Summary of Health Effects Local effects following inhalation of chloroform include shortness of breath and irritation of the nose and throat. Acute inhalation can cause systemic effects such as, excitement, nausea and vomiting followed by ataxia, dizziness, drowsiness.
How does chloroform affect the environment?
Chloroform is used in some countries to produce a refrigerant known as R-22. The use of R-22 is gradually decreasing, however, since it causes ozone depletion in the atmosphere. Chloroform is also released into the air from pulp and paper mills and from landfills and hazardous waste sites.
What works faster chloroform?
Induction with fluothane was faster than with chloroform and ether. Fluothane seems slightly more potent than chloroform and has an equal margin of safety.
Why water is not used in NMR?
Not only water even any material peaks changes with respect to the solvent. … In water proton attached to the electronegative atom so it is considered as a exchangeable proton. If the NMR solvent is able to make hydrogen bond the then peak value going to be more down field, if not peak value going to be low.
What is called coupling constant?
The distance between any two adjacent lines in the NMR peaks of two sets of equivalent hydrogen nuclei coupled only to each other is the same, which, when expressed in hertz, is called the coupling constant (symbol: J) of the two sets of equivalent hydrogen nuclei.
How many NMR signals are in methanol?
For example methanol has two different sets of protons (methyl and hydroxyl) and therefore is expected to show two signals in the proton NMR spectrum.
What is upfield and downfield in NMR?
The terms “upfield” and “downfield” describe the relative location of peaks. Upfield means to the right. Downfield means to the left. • NMR absorptions are measured relative to the position of a reference peak at 0 ppm on the. δ scale due to tetramethylsilane (TMS).
How do you predict chemical shift in NMR?
H NMR Chemical Shifts Tetramethylsilane [TMS;(CH3)4Si] is generally used for standard to determine chemical shift of compounds: δTMS=0ppm. In other words, frequencies for chemicals are measured for a 1H nucleus of a sample from the 1H or resonance of TMS.
Is CDCl3 acidic or basic?
The chemistry behind is as follows; CDCl3 is of acidic nature, so there is proton exchange, proton of acid is exchanged with Deuterated (D) solvent. As a result, the peak is not visible. In chloroform it appears as a very broad signal around 11 ppm.
What is the Colour of chloroform?
Chloroform appears as a clear colorless liquid with a characteristic odor.
Is deuterated chloroform soluble in water?
Chloroform-D is less soluble with water and the water content is low, so its water peak near 1.59 ppm. Deuterated acetone is about 2.8 ppm and the deuterated dimethylsulfoxide has a water peak of about 3.4 ppm.