Does ionic compounds have empirical formula
 John Peck
John Peck The formula for ionic compounds is always an empirical formula because it shows the smallest, whole-number ratio between the cations and anions. The actual number of ions in a sample of an ionic compound will be very large and will vary from sample to sample.
Is empirical formula covalent or ionic?
An empirical formula is the lowest ratio of the atoms within a molecule. The empirical formula is accurate when describing ionic compounds, which cannot be broken into a single molecule unit. But when describing covalent compounds we use a molecular formula which describes the atoms within a single molecule.
Do all compounds have empirical formula?
Chemical Formula from Empirical Formula. The chemical formula for a compound obtained by composition analysis is always the empirical formula.
How do you find the empirical formula of an ionic compound?
To write the empirical formula for an ionic compound, we use the periodic table to identify the metallic cation and the nonmetallic anion and write their symbols and charges. We then determine the ratio of each element to ensure a total charge of zero.Can ionic compounds have both molecular and empirical formulas?
Yes, ionic compounds have only empirical formulas.
Are ionic compounds molecular formulas?
Ionic compounds do not exist as molecules. In the solid state, ionic compounds are in crystal lattice containing many ions each of the cation and anion. An ionic formula, like NaCl, is an empirical formula. This formula merely indicates that sodium chloride is made of an equal number of sodium and chloride ions.
How do you find the empirical formula?
- In any empirical formula problem you must first find the mass % of the elements in the compound. …
- Then change the % to grams. …
- Next, divide all the masses by their respective molar masses. …
- Pick the smallest answer of moles and divide all figures by that.
Do empirical formulas have charges?
empirical formula – the simplest formula we can write. molecular formula – tells us exactly how many atoms of each element are in a single molecule of that compound. … Since these compounds involve charges, they are also considered polar.What does the formula of an ionic compound tell us?
Ionic compounds can be described using chemical formulas, which represent the ratios of interacting elements that are found in the ionic solid or salt. … In formula notation, the elements are represented by their chemical symbols followed by numeric subscripts that indicate the relative ratios of the constituent atoms.
Which of the following is an ionic compound?Ionic CompoundUseNaCl, sodium chlorideordinary table saltCaCO3 calcium carbonateingredient in antacidsMg(OH)2, magnesium hydroxideingredient in antacidsAl(OH)3, aluminum hydroxideingredient in antacids
Article first time published onWhich substances have the same empirical formula?
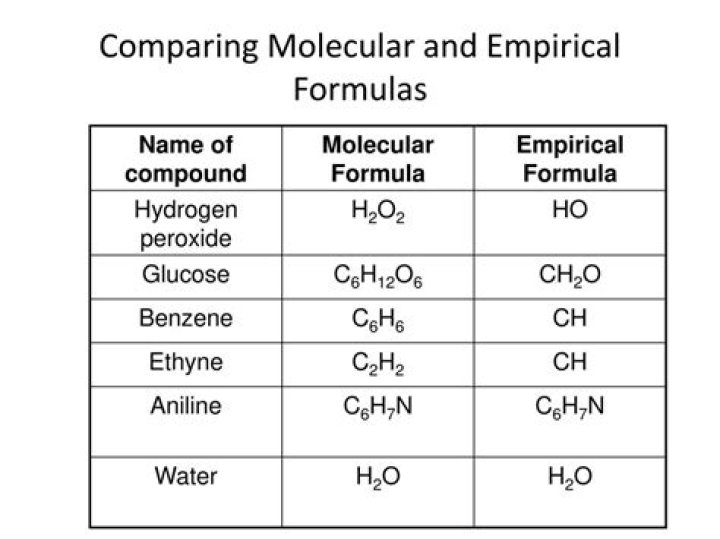
The Empirical formula is the lowest whole number ratio of the elements in a compound. In (section 2.10), we discovered that benzene and acetylene have the same mass percent composition, and thus it is logical that they have the same ratio of elements to each other, that is, they have the same empirical formula.
Which of the following is not empirical formula?
Explanation: An empirical formula represents the simplest whole number ratio of elements in a compound. Since the subscripts of the elements in C6H12O6 can be divided by 6 to get the simplest whole number ratio of elements, it is not an empirical formula.
How are molecules different from ionic compounds?
Molecular compounds are pure substances formed when atoms are linked together by sharing of electrons while ionic compounds are formed due to the transfer of electrons. … Molecular compounds are made due to covalent bonding while ionic compounds are made due to ionic bonding.
Why are molecular formulas not used in ionic compounds?
Because an ionic compound is not made up of single, discrete molecules, it may not be properly symbolized using a molecular formula. Instead, ionic compounds must be symbolized by a formula indicating the relative numbers of its constituent ions.
Which compound has the same empirical and molecular formula?
Empirical formulaCH2O (40.0% C; 6.7% H; 53.3% O)CompoundformaldehydeMolecular formulaCH2OBoiling point, °C-21
What are the subscripts in the empirical formula of this compound?
Subscripts after element symbols represent the number of atoms. If there is no subscript, it means one atom is present in the compound. … The empirical formula is the ratio of elements present in the compound. The subscripts in the formula are the numbers of atoms, leading to a whole number ratio between them.
What is empirical formula with example?
In chemistry, the empirical formula of a chemical compound is the simplest whole number ratio of atoms present in a compound. A simple example of this concept is that the empirical formula of sulfur monoxide, or SO, would simply be SO, as is the empirical formula of disulfur dioxide, S2O2.
What is required to calculate the empirical formula of a compound?
In order to determine the Empirical formula for a compound or molecule, we need to know the mass percentages of the the elements in the compound. Once we have this information we can convert it to moles to determine the ratios between the elements. Start with the number of grams of each element, given in the problem.
Are empirical and molecular formulas the same?
Empirical formulas show the simplest whole-number ratio of atoms in a compound, molecular formulas show the number of each type of atom in a molecule, and structural formulas show how the atoms in a molecule are bonded to each other.
How do you tell if a formula is ionic or covalent?
- If a compound is made from a metal and a non-metal, its bonding will be ionic.
- If a compound is made from two non-metals, its bonding will be covalent.
Why are empirical formulas used for ionic compounds?
The formula for ionic compounds is always an empirical formula because it shows the smallest, whole-number ratio between the cations and anions. The actual number of ions in a sample of an ionic compound will be very large and will vary from sample to sample. Figure 2.6.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.
What is a chemical formula for an ionic compound called?
Chemical formulas for ionic compounds are called ionic formulas. A proper ionic formula has a cation and an anion in it; an ionic compound is never formed between two cations only or two anions only. The key to writing proper ionic formulas is simple: the total positive charge must balance the total negative charge.
When writing an ionic formula What is the total?
Chemical formulas for ionic compounds are called ionic formulas. A proper ionic formula has a cation and an anion in it; an ionic compound is never formed between two cations only or two anions only. The key to writing proper ionic formulas is simple: the total positive charge must balance the total negative charge.
How do you know the ionic charge of a compound?
- the number of charges on an ion formed by a metal is equal to the group number of the metal.
- the number of charges on an ion formed by a non-metal is equal to the group number minus eight.
- hydrogen forms H + ions.
What do ionic compounds consist of?
27-31. Ionic compounds are compounds composed of ions, charged particles that form when an atom (or group of atoms) gains or loses electrons. (A cation is a positively charged ion; an anion is a negatively charged ion.)
Are all ionic compounds crystalline?
Ionic compounds usually form crystalline structures when solid. Ionic compounds containing basic ions hydroxide (OH−) or oxide (O2−) are classified as bases. … Ionic compounds typically have high melting and boiling points, and are hard and brittle.
Is NaCl molecular or empirical?
Sodium Chloride is an ionic compound with the chemical formula NaCl. Since sodium chloride is not a molecule it has an empirical formula, not a molecular formula.
Is ch3 an empirical formula?
Therefore, CH3 is the empirical formula. CH3 molar mass = 1C + 3H = (12.01) + 3(1.01) = 15.04 g/mol.
How do you tell if two compounds have the same empirical formula but different molecular formula they must have?
If two compounds have the same empirical formula but different molecular formula, they must have Different molecular weights .
What is empirical formula give an example for a compound whose empirical and molecular formula are the same?
The compound that have same molecular as well as empirical formula is carbon monoxide i.e. CO. In case of carbon monoxide, the ratio of carbon and oxygen atoms is 1:1 thus it is already as simple as it can be, hence the empirical formula is CO same as the molecular formula.