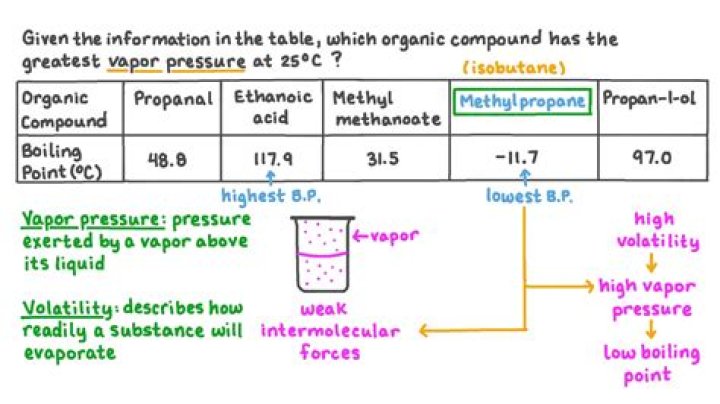
Which compound would have the highest vapor pressure at a given temperature
 Emily Sparks
Emily Sparks Thus at room temperature, the substance with the lowest boiling point will have the highest vapor pressure (easiest to get into the gas phase). The substance with the highest boiling point will have the lowest vapor pressure.
What substances have higher vapor pressures?
Thus at room temperature, the substance with the lowest boiling point will have the highest vapor pressure (easiest to get into the gas phase). The substance with the highest boiling point will have the lowest vapor pressure.
Do ionic compounds have high vapor pressure?
Ionic solid have high vapor pressures because they have very strong intermolecular forces holding the molecules together in a crystalline structure.
Does higher temperature mean higher vapor pressure?
As the temperature of a liquid increases, the kinetic energy of its molecules also increases. As the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure.Which would you expect to have the highest vapor pressure at a given temperature quizlet?
Which would you expect to have the highest vapor pressure at a given temperature? To evaporate or change phase to a gas, liquids must overcome their IMFs. Therefore, substances with smaller IMFs tend to have higher vapor pressures.
Is vapor pressure proportional to temperature?
The vapor pressure of a liquid varies with its temperature, as the following graph shows for water. … As the temperature of a liquid or solid increases its vapor pressure also increases. Conversely, vapor pressure decreases as the temperature decreases.
Do larger molecules have higher vapor pressure?
The bigger the molecule is and the more electrons it has, the bigger the London forces are. Bigger molecules usually have larger molecular weights; hence the correlation of vapor pressure with molecular weight.
How do you find the vapor pressure of a compound?
In chemistry, vapor pressure is the pressure that is exerted on the walls of a sealed container when a substance in it evaporates (converts to a gas). To find the vapor pressure at a given temperature, use the Clausius-Clapeyron equation: ln(P1/P2) = (ΔHvap/R)((1/T2) – (1/T1)).What is a high vapor pressure?
Vapor pressure is a property of a liquid based on the strength of its intermolecular forces. A liquid with weak intermolecular forces evaporates more easily and has a high vapor pressure. A liquid with stronger intermolecular forces does not evaporate easily and thus has a lower vapor pressure.
What factors affect vapor pressure?Three common factors that influence vapor press are surface area, intermolecular forces and temperature. The vapor pressure of a molecule differs at different temperatures.
Article first time published onWhich has the highest vapor pressure quizlet?
The compound with the weakest intermolecular forces will have the highest vapor pressure. CH3CH2CH2F is the largest and only polar compound and therefore has the greatest intermolecular forces (both dipole-dipole and the greatest London Dispersion forces) and is eliminated.
Do ionic compounds have lower vapor pressure?
One property of ionic compounds is very high melting and boiling points. Compared to covalent compounds they are much less volatile. … The high electronegativity of ionic compounds leads to lower vapor pressures.
Which has higher vapor pressure water or ethanol?
Water is much smaller than either of the previous substances and exhibits weaker dispersion forces, but its extensive hydrogen bonding provides stronger intermolecular attractions, fewer molecules escaping the liquid, and a lower vapor pressure than for either diethyl ether or ethanol.
Which has a higher vapor pressure br2 or i2?
Since I2 has higher molecular weight, it has stronger London dispersion forces so it has a higher boiling point than Br2.
Which compound will have the highest boiling point group of answer choices?
Among the given options, (c) ethanol has the highest boiling point as it has the strongest intermolecular force of hydrogen bonding.
Which compound would you expect to have the higher boiling point?
Compounds I and IV would be higher boiling point compounds because of additional hydrogen bonding (strong intermolecular forces). Compound IV would be the highest boiling because the hydroxy group and carboxylic acid group could BOTH participate in intermolecular hydrogen bonding.
Which has lowest Vapour pressure?
Mercury exerts the lower vapour pressure because of the strong bond among its atom.
At which temperature would you expect water to have the greatest vapor pressure?
At a pressure greater than 1 atm, water boils at a temperature greater than 100°C because the increased pressure forces vapor molecules above the surface to condense. Hence the molecules must have greater kinetic energy to escape from the surface.
Does vapor pressure increase with atmospheric pressure?
The vapor pressure is hardly influenced by the atmospheric pressure in that regard, because we ignore the intermolecular forces in ideal vapors.
How do you find the vapor pressure of water when given temperature?
Simple formula simple_pressure = e^(20.386 – (5132 / (temperature + 273)) , where vapor pressure is expressed in mmHg and temperature in kelvins.
What is the example of Vapour pressure?
Characteristics of Vapor Pressure It is important to note that when a liquid is boiling, its vapor pressure is equal to the external pressure. For example, as water boils at sea level, its vapor pressure is 1 atmosphere because the external pressure is also 1 atmosphere.
How does the vapor pressure of a substance depend on temperature and strength of intermolecular forces?
How does it depend on temperature and strength of intermolecular forces? vapor pressure is the partial pressure of a gas in dynamic equilibrium with its liquid. It increases with increasing temperature and also increases with decreasing strength of intermolecular forces.
Does CH4 or CH3CH3 have a higher vapor pressure?
The bigger molecule has more interactions and hence the higher b.p. CH3CH2OCH2CH3 is a bigger molecule than CH4 and CH3CH3, so has more dispersion forces.
Does CH3CH2CH2OH or CH3OH have a higher vapor pressure?
The main reason why CH3OH has a higher vapor pressure at a given temperature when compared to CH3CH2CH2OH is that CH3OH… The main reasons why H2CO has a higher vapor pressure at a given temperature when compared to CH3OH is that H2CO…
Which compound has the highest boiling point CH3CH2CH2OH?
The hydrogen bonding in CH3CH2OH and CH3CH2CH2OH give these molecules a higher boiling point. Of the two, CH3CH2CH2OH has more electrons and thus stronger London forces, so it is the higher-boiling compound.
Which substance has the lowest vapor pressure at room temperature?
The graph shows that propanone has the greatest vapor pressure at any given temperature compared to the other three liquids, while ethanoic acid has the lowest vapor pressure at any given temperature compared to the other three liquids.
Which has higher vapor pressure methane or carbon tetrachloride?
We know that the liquid molecules are stick together by Van Der Waal’s force. Due to the higher molecular weight of carbon tetrachloride, a large amount of energy is required to convert from liquid to vapor phase. Hence, carbon tetrachloride has a higher boiling point than methane.
Which alkane compound has the lowest vapor pressure?
The dispersion forces between the smaller pentane molecules are much weaker than the forces between the larger heptane molecules. Therefore pentane has the largest vapor pressure and heptane has the smallest vapor pressure.
Which has higher vapor pressure pentane or butanol?
At 20°C, pentane (MW = 72.2 g/mol) has a vapor pressure of 434mmHg while butanol (MW = 74.1g/mol) has a vapor pressure of 6mmHg. … So, for butanol intermolecular forces are stronger then the vapour pressure is lower.
Does methanol or ethanol have a higher vapor pressure at room temperature?
Methanol has the higher vapor pressure because its molecular weight is less than ethanol and so its intermolecular forces are less than ethanol’s.