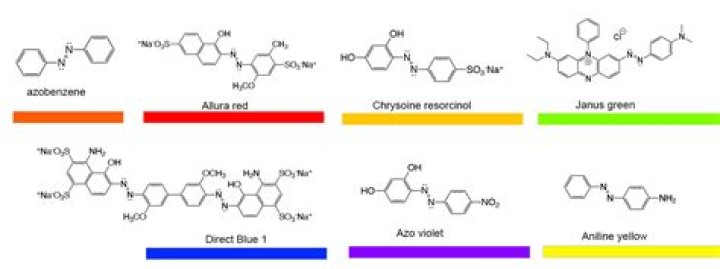
Why do different dye molecules make different colors
 Christopher Lucas
Christopher Lucas Each different shape of dye molecule absorbs light differently. That’s what makes the different colors! If you put these two molecules, the dye and the cotton, together, nothing will happen, unless you can get some of the atoms on the surfaces to come unstuck.
Why do dyes have different Colours?
Unlike most organic compounds, dyes possess colour because they 1) absorb light in the visible spectrum (400–700 nm), 2) have at least one chromophore (colour-bearing group), 3) have a conjugated system, i.e. a structure with alternating double and single bonds, and 4) exhibit resonance of electrons, which is a …
Why do different fabrics dye differently?
It was hypothesized that there would be significant variation across the different fabrics in terms of absorption of the dye, that the natural fabrics would absorb the dye more effectively than the synthetic fibers, and that the cotton would bond the best with the dye overall.
Why do molecules have different colors?
That is because for a single atom the electrons have to absorb and emit the same light. In molecules, where two or more atoms share some of their electrons, the molecules can absorb light of one color and emit another color. This works whether the atoms are the same (eg two Nitrogen atoms) or different elements.How do dyes work chemically?
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. … Both dyes and pigments are colored, because they absorb only some wavelengths of visible light.
How does dyeing occur at the molecular level?
The dyeing process must place dye molecules within the microstructure of the fibre. The dye molecules can be anchored securely through the formation of covalent bonds that result from chemical reactions between substituents on the molecules of the dye and the fibre.
What is a dye molecule?
Dye molecules have large molecular weights and contain extended systems of conjugated double bonds. These molecules can be dissolved in an adequate organic solvent (such as ethanol, methanol, ethanol/water, and methanol/water) or incorporated into a solid matrix (organic, inorganic, or hybrid).
How do atoms and molecules behave differently?
When two or more atoms form chemical bonds together, that association is called a molecule. Molecules usually behave differently than the atoms that they are composed of. … When a substance is made of molecules rather than a single element we call that substance a compound.Can molecules change color?
Molecular Basis for the Indicator Color Change Color changes in molecules can be caused by changes in electron confinement. More confinement makes the light absorbed more blue, and less makes it more red.
What causes color change in chemical reactions?When two or more substances combine, they create one or more new substances, which sometimes have different molecular structures from the original substances, meaning they absorb and radiate light in different ways, leading to a color change.
Article first time published onWhat does it mean for a dye to run in chemistry?
A dye will run if it has a weak affinity for the material it is attached to, or a much stronger affinity for a non-aqueous solvent. Detergents may cause running because they help to stabilise the hydrophobic regions of dye molecules due to their ability to form micelles.
Why the indigo dye did not adhere to the different fibers?
It does not attach to the fiber by means of hydrogen bonds. Unlike most dyes, indigo has a neutral charge. … When cotton or another natural fiber is dipped into a solution of reduced indigo, some of the dissolved indigo penetrates inside the fibers, between the cellulose or protein molecules (depending on the fiber).
Why the indigo dye did not adhere to the different fibers in your test fabric?
Indigo does not form a chemical bond with the fabric, as other dyes. Instead it forms a physical bond, bonding molecularly to the fabric when reduced. … This makes indigo very impervious to sunlight, but indigo will fade with rubbing.
What is a dye molecule describe the theory of dyeing?
Dyeing is the application of dyes or pigments on textile materials such as fibers, yarns, and fabrics with the goal of achieving color with desired color fastness. … Dye molecules are fixed to the fiber by absorption, diffusion, or bonding with temperature and time being key controlling factors.
What is colorant in textile?
Definition. Textile colorants impart color to a textile material, usually with a high degree of permanency, as a result of their chemical binding or physical entrapment within or around the textile material.
What is the function of dye?
dye, substance used to impart colour to textiles, paper, leather, and other materials such that the colouring is not readily altered by washing, heat, light, or other factors to which the material is likely to be exposed.
How would you describe the movement of the dye molecules?
It may also be described as the diffusion of molecules across a membrane. … Random movements of the dye and water molecules cause them to bump into each other and mix. Thus, the dye molecules move from an area of higher concentration to an area of lower concentration.
What are the different types of dye molecules?
Acidic dyes, basic dyes, azoic dyes, nitro dyes, vat dyes, mordant dyes, and sulphur dyes, etc. are synthetic dyes.
What is dye in microbiology?
Stain or dye is the synthetic chemical which is derived from nitrobenzene or aniline. stains are used commonly in microbiology to increase the contrast between microorganisms or parts of its and the background,so that it can be easily visible.
What is role of water and dye molecule in textile processing?
Except that it is the solvent for great number of dyes, water has also the function of structural activator through the process of hidration of functional fibre group that is of a great importance of adsorption and diffucsion of dyes in the internal area of fibres.
What charge does the dye molecule have?
At the molecular level, basic dyes have amino groups, which are responsible for the molecule’s positive charge. Because of their positive charge, basic dyes react with negatively charged compounds. Therefore, the primary way in which the coloration process is achieved with basic dyes is by ionic bonding.
What happens to the cotton cellulose molecules and the dye molecules when combined?
If you put these two molecules, the dye and the cotton, together, nothing will happen, unless you can get some of the atoms on the surfaces to come unstuck. If the H comes off of the cellulose, and the Cl comes off of one end of the dye molecule, the molecules will be able to react with each other and stick together.
How does atomic structure affect color?
The arrangement of atoms in a structure affects the spacing of the allowed energy levels of the electrons in the structure, which in turn affects the possible frequencies of absorbed and emitted photons. The frequency of the photon determines the color of the photon as perceived by us.
How do firework manufacturers create fireworks of different colors?
The colors in fireworks come from a simple source: pure chemistry. They’re created by the use of metal salts. … Some of these compounds produce intense colors when they are burned, which makes them ideal for fireworks. Others, like potassium nitrate, sulfur and charcoal are often used to help the fireworks burn.
How do atoms make color?
When the atoms of a gas or vapor are excited, for instance by heating or by applying an electrical field, their electrons are able to move from their ground state to higher energy levels. … This energy corresponds to particular wavelengths of light, and so produces particular colors of light.
How do molecules of matter differ from each other?
Gases, liquids and solids are all made up of microscopic particles, but the behaviors of these particles differ in the three phases. … gas are well separated with no regular arrangement. liquid are close together with no regular arrangement. solid are tightly packed, usually in a regular pattern.
Why do molecules have different arrangements?
caffeine moleculeCaffeine is a complicated molecule, composed of many atoms bonded to each other in a specific arrangement. Molecular size varies depending on the number of atoms that make up the molecule.
Why do molecules behave differently?
The behaviour of molecules in different phases of matter represents a balance between the kinetic energies of the molecules and the attractive forces between them. All molecules are attracted to each other. … At higher temperatures, the kinetic energy of the molecules is higher.
What type of chemical reaction changes color?
Universal indicator is a chemical that changes color in the presence of acids and bases from a pH of 2 to 10. Acids turn the indicator red, pink, orange, and yellow, while bases turn it green, blue, and purple.
Why do solutions change Colour?
Whether a solution is acidic or basic can be measured on the pH scale. When universal indicator is added to a solution, the color change can indicate the approximate pH of the solution. Acids cause universal indicator solution to change from green toward red. … Acidic solutions have a pH below 7 on the pH scale.
Why does the oxidation change result in a color change?
Why does this oxidation result in a color change? … The number of electrons were changed which changes the oxidation as well. This causes the energy to change the wavelengths. When these wavelengths change the color is absorbed and changed as well.