What are the properties of elements in the periodic table
 Emma Terry
Emma Terry These properties include color, density, melting point, boiling point, and thermal and electrical conductivity. While some of these properties are due chiefly to the electronic structure of the element, others are more closely related to properties of the nucleus, e.g., mass number.
What are the properties of the elements?
All elements have properties. Those properties include, but are not limited to, conductivity, magnetism, melting point, boiling point, color, state of matter, and others. Elements with similar properties are grouped together in different areas of the periodic table of elements.
What are the 10 properties of elements?
- Atomic number. The atomic number indicates the number of protons within the core of an atom. …
- Atomic mass. The name indicates the mass of an atom, expressed in atomic mass units (amu). …
- Electronegativity according to Pauling. …
- Density. …
- Melting point. …
- Boiling point. …
- Vanderwaals radius. …
- Ionic radius.
What are the 5 properties of the periodic table?
Major periodic trends include electronegativity, ionization energy, electron affinity, atomic radii, ionic radius, metallic character, and chemical reactivity.What is periodic table and its properties?
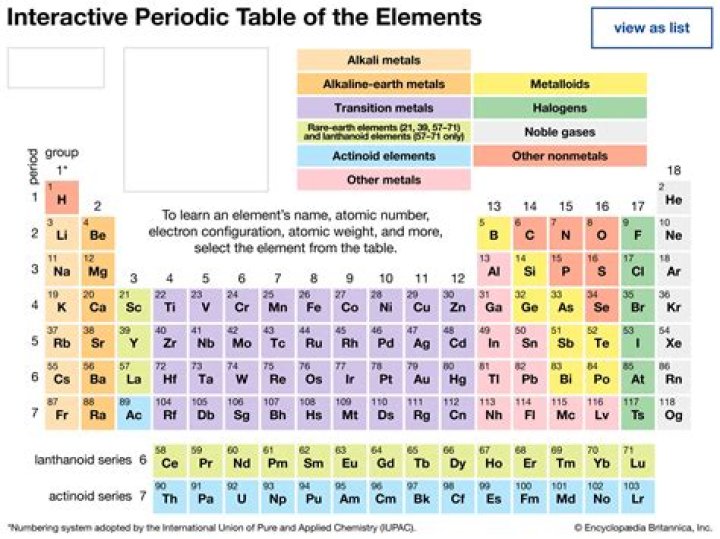
The periodic table is a tabular display of various chemical elements that are arranged according to their electronic configuration, atomic number, and common chemical properties. There are certain trends that are common throughout all groups and periods.
What are the properties of Group 8 elements?
- They are inert gases located on the right of the periodic table.
- They have a full-set of valence electrons, so they’re stable.
- They are colorless, odorless and tasteless.
- They have low melting and low boiling points.
What are the 4 types of elements?
Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals.
What are the properties of an element and compound?
An element is a material that consists of a single type of atom. Each atom type contains the same number of protons. Chemical bonds link elements together to form more complex molecules called compounds. A compound consists of two or more types of elements held together by covalent or ionic bonds.What is periodic property class 11?
The basic law governing modern periodic table states that the properties of elements are periodic functions of their atomic number. These properties reappear at regular intervals or follow a particular trend at regular intervals. This phenomenon is known as the periodicity of elements.
What are the 3 types of elements in the periodic table?The elements can be classified as metals, nonmetals, or metalloids. Metals are good conductors of heat and electricity, and are malleable (they can be hammered into sheets) and ductile (they can be drawn into wire).
Article first time published onHow many types of elements are there in periodic table?
This list contains the 118 elements of chemistry. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. The first chemical element is Hydrogen and the last is Ununoctium.
What are the 5 types of elements?
Everything in nature is made up of five basic elements: earth, water, fire, air, and space.
What are the properties of Group 1?
Group one elements share common characteristics. They are all soft, silver metals. Due to their low ionization energy, these metals have low melting points and are highly reactive. The reactivity of this family increases as you move down the table.
What are the properties of Group 1 on the periodic table?
ElementMelting pointLithium, Li180°CSodium, Na98°CPotassium, K63°CRubidium, Rb39°C
What are the properties of Group 0?
Group 0 Elements the outer shell, apart from Helium which has 2 – stable full outer shell. Unreactive. They are very unreactive. This means they don’t form molecules easily, so elements are found as single atoms.
On which property of an element the periodic table depends on?
An element’s location in the periodic table is largely dependent on its electrons; the number of valence shell electrons determines its group, and the type of orbital in which the valence electrons lie in determines the element’s block.
What are the properties of compound?
PropertyIonic CompoundsMolecular CompoundsPhysical state at room temperatureSolidGas, liquid, or solidWater solubilityUsually highVariableMelting and boiling temperaturesGenerally highGenerally lowElectrical conductivityGood when molten or in solutionPoor
What are the three properties of compounds?
- They have a variable composition.
- They have no definite properties.
- They can be separated by physical methods.
What are matter properties?
Any characteristic that can be measured, such as an object’s density, color, mass, volume, length, malleability, melting point, hardness, odor, temperature, and more, are considered properties of matter.
What are properties of metals?
- high melting points.
- good conductors of electricity.
- good conductors of heat.
- high density.
- malleable.
- ductile.
How are elements arranged in the periodic table?
In the modern periodic table, the elements are listed in order of increasing atomic number. The atomic number is the number of protons in the nucleus of an atom. … In a periodic table arranged in order of increasing atomic number, elements having similar chemical properties naturally line up in the same column (group).
What are elements name the 3 types of elements write any one property of elements?
The elements are arranged in the periodic table and split as either metallic or non-metallic based on their groups. Metallic is further categorized into Main Group Metals, Transition Metals, and f-block metals. These are again further divided, depending upon their properties.
How many metals are in the periodic table?
According to the Royal Society of Chemistry, there are 94 metals listed on the Periodic Table.
What are the 7 elements?
The 7 elements of design consider space, line, form, light, color, texture and pattern. A balance of these elements is vital to every scheme.
What are the properties of Group 2 in the periodic table?
The Group 2 alkaline earth metals include Beryllium, Magnesium, Calcium, Barium, Strontium and Radium and are soft, silver metals that are less metallic in character than the Group 1 Alkali Metals.
What are the properties of Group 7 elements?
- At room temperature (20 °C), the physical state of the halogens changes as you go down the group. Fluorine and chlorine are gases, bromine is a liquid and iodine is crumbly solid.
- The colours of the halogens also change as you descend the group – they become darker.
What are the chemical properties of Group 7 elements?
- Fluorine is a pale yellow gas.
- Chlorine is a poisonous green gas.
- Bromine is a toxic red-brown liquid.
- Iodine is a dark grey solid which gives off a purple vapour when heated.
- Astatine is a black solid.
What are 2 properties of alkali metals?
- shiny.
- soft.
- silvery.
- highly reactive at standard temperature and pressure.
- readily lose their outermost electron to form cations with a charge of +1.
What is Group 2 on the periodic table called?
Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). … In most cases, the alkaline earth metals are ionized to form a 2+ charge.