How is biodiesel synthesized
 Emily Sparks
Emily Sparks Biodiesel is produced
How is Biodiesel produced from biomass?
Bio-oil. Biomass serves as a suitable feedstock for the production of bio-oil. It is considered green energy with respect to reducing environmental pollution. Two processes are used for the production of bio-oil from biomass: fast pyrolysis and hydrothermal liquefaction.
What is the process of transesterification?
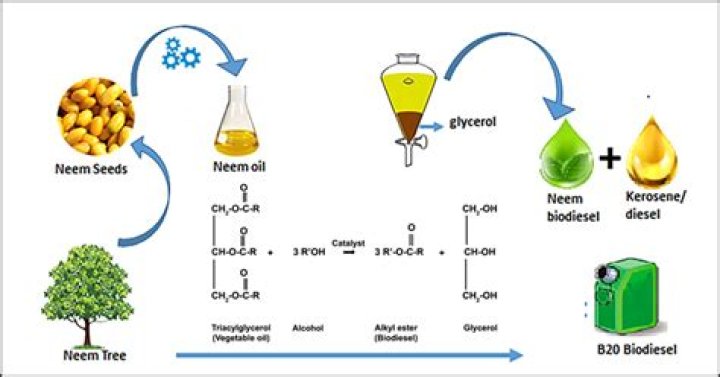
Transesterification is the process in which fat or oil reacts with an alcohol to form esters and glycerol. A catalyst is used to improve the reaction rate and yield. Because the reaction is reversible, excess alcohol is used to shift the equilibrium to the product side.
Which catalyst is used in synthesis of biodiesel?
Strong alkali catalysts such as NaOH, KOH, CH3ONa and CH3OK (potassium methoxide) are used for biodiesel production.How are biofuels formed?
There are various ways of making biofuels, but they generally use chemical reactions, fermentation, and heat to break down the starches, sugars, and other molecules in plants. The resulting products are then refined to produce a fuel that cars or other vehicles can use.
What is the cause of soap formation during biodiesel production?
During production of biodiesel, the free fatty acids in the oil react with the sodium or potassium catalyst to form soaps. After the biodiesel and by‐product glycerin are separated, trace amounts of glycerin remain in the biodiesel.
What is the process of production of biofuel?
Biodiesel production is the process of producing the biofuel, biodiesel, through the chemical reactions of transesterification and esterification. This involves vegetable or animal fats and oils being reacted with short-chain alcohols (typically methanol or ethanol). The alcohols used should be of low molecular weight.
How can biodiesel be made through transesterification process?
The transesterification of vegetable oils, animal fats or waste cooking oils is the process behind conventional biodiesel. In the transesterification process a glyceride reacts with an alcohol (typically methanol or ethanol) in the presence of a catalyst forming fatty acid alkyl esters and an alcohol.How do you convert an oil reaction to biodiesel?
Biodiesel is an alternative fuel for diesel engines that is produced by chemically reacting a vegetable oil or animal fat with an alcohol such as methanol or ethanol. In words, the reaction is: Oil + alcohol → biodiesel + glycerin. The photo shows a bottle of biodiesel and glycerin (also called glycerol).
What is transesterification in biodiesel process?Transesterification is a chemical reaction used for the conversion of triglycerides (fats) contained in oils, (Feedstocks) into usable biodiesel. Biodiesel produced by the process of transesterification has a much lower viscosity, making it capable of replacing petroleum diesel in diesel engines.
Article first time published onHow transesterification is helpful in Biodiesel Production?
Transesterification is an imperative process for biodiesel production, as it can reduce the viscosity of the feedstock/vegetable oils to a level closer to the conventional fossil-based diesel oil [20].
Where are biofuels produced?
CharacteristicProduction in petajoulesUnited States1,347.3Brazil883.7Indonesia283Germany146.3
How are biofuels made from algae?
It’s a fairly simple process: Algae are cultivated in large pools or farms. The micro-organisms convert sunlight to energy, and store the energy as oil. The oil is extracted using a mechanical process such as pressing or using sound waves, or with chemical solvents that break down the cell walls and release the oil.
How is biomass made?
Biomass contains stored chemical energy from the sun. Plants produce biomass through photosynthesis. Biomass can be burned directly for heat or converted to renewable liquid and gaseous fuels through various processes.
Can you make biodiesel without methanol?
Of Course Yes, Biodiesel can be produced by the reaction of vegetable oil with any alkyl source such as methanol, ethanol, dimethyl carbonate, methyl acetate, ethyl acetate…. etc.
Which plant is used for biodiesel production?
Biodiesel can be made from oils which have been extracted from plants such as palm, soybean, oilseed rape, or sunflower.
What is the byproduct of producing biodiesel?
Biodiesel production will generate about 10% (w/w) glycerol as the main byproduct. In other words, every gallon of biodiesel produced generates approximately 1.05 pounds of glycerol. This indicates a 30-million-gallon-per-year plant will generate about 11,500 tonnes of 99.9 percent pure glycerin.
Which oil is best for making biodiesel?
Biodiesel is a valuable fuel source made from soybean oil, animal fats, and other kinds of recycled cooking oil. It’s renewable, clean-burning, reduces waste, and is surprisingly easy to make.
How much biodiesel is produced from vegetable oil?
More than 80 percent of biodiesel is made from vegetable oil (the rest is mostly animal fats).
How is biodiesel extracted from vegetable oil?
TRANSESTERIFICATION PROCESS Oil or fat reacts with alcohol (methanol or ethanol). This reaction is called transesterification. The reaction requires heat and a strong catalyst (alkalis, acids, or enzymes) to achieve complete conversion of the vegetable oil into the separated esters and glycerin.
How do you separate glycerol from biodiesel?
Two-Step Purification of Glycerol as a Value Added by Product From the Biodiesel Production Process. For every ton of biodiesel produced, about 100 kg of glycerol is also generated as a by-product. The traditional method of removing glycerol is mainly by gravity separation or centrifugation.
What is the difference between transesterification and saponification?
is that transesterification is (organic chemistry) the reaction of an ester with an alcohol in order to replace the alkoxy group; it is used in the synthesis of polyesters and in the production of biodiesel while saponification is (chemistry) the hydrolysis of an ester under basic conditions to form an alcohol and the …
How do you make Methoxide?
On a large scale, potassium methoxide is produced by decomposing potassium amalgam with methanol, which is produced by the chloralkali-electrolysis of potassium chloride by the amalgam process. Impurities of the resulting potassium methoxide in methanol with metallic mercury can be eliminated by ultrafiltration.
Which is the most preferred method of biodiesel?
Due to the quality of the fuel produced, the transesterification method is the most preferred way to produce biodiesel from diverse feedstock types. Through this method, oils and fats (triglycerides) are converted to their alkyl esters with reduced viscosity to near diesel fuel levels.
Why does glycerol and biodiesel separate?
As a consequence, the non-polar methyl ester molecules making up the biodiesel do not mix with the polar glycerol molecules and the mixture of products will separate into two layers with the less dense biodiesel floating on top of the more dense glycerol layer.
How are triglycerides converted to biodiesel?
Biodiesel is a renewable fuel source. … One popular process for producing biodiesel from fats/oils is transesterification of triglyceride by methanol (methanolysis) to make methyl esters of straight-chain fatty acids. The purpose of the transesterification process is to lower the viscosity of oil.
What is biodiesel explain the method to obtained biodiesel from vegetable oil by transesterification?
Biodiesel is obtained by trans esterification (means the process of converting one ester to another ester) of vegetable oil or animal fats with methyl alcohol using catalyst sodium metal or sodium methaxide. … This is converted into methyl esters of the fatty acids.
Why does transesterification process occur?
Transesterification is one of the reversible reactions and proceeds essentially by mixing the reactants. However, the presence of a catalyst (a strong acid or base) accelerates the conversion and a little excess of alcohol is used to shift the equilibrium toward the formation of fatty acid alkyl esters and glycerol.
What is esterification and transesterification?
Esterification is any reaction (typically between an fatty acid and an alcohol) that results in the production of an ester, while transesterification is the reaction of an ester with an alcohol in order to replace the alkoxy group; it is used in the synthesis of polyesters and in the production of biodiesel.
What kind of reaction is transesterification?
Transesterification is an organic reaction in which the R group of an alcohol is exchanged with an R’ group of an ester. This is generally done via the introduction of an acid or base catalyst to the reaction mixture.
What is transesterification give one example of it?
In a transesterification reaction an alcohol molecule and an ester molecule react in either the presence of an acid or base to form a new ester. … For example, methyl ethanoate reacts with ethanol in the presence of hydrochloric acid to form ethyl ethanoate and methanol.