What is ferrocene soluble in
 Andrew White
Andrew White Ferrocene is an air-stable orange solid with a camphor-like odor. As expected for a symmetric, uncharged species, ferrocene is soluble in normal organic solvents, such as benzene, but is insoluble in water. It is stable to temperatures as high as 400 °C.
Is ferrocene soluble in ethanol?
It is soluble in many organic solvents such as methanol, ethanol, ethyl ether, petroleum ether, gasoline, kerosene, diesel oil, methylene chloride, benzene, toluene, and xylene, Because of the large polarity of ethanol polarity, it is usually recommended to dissolve it with toluene.
Why is ferrocene soluble in organic solvents?
In ferrocene one iron atom is sandwiched between two cyclopentadiene rings. Means ferrocene contains one iron atom and two cyclopentadiene rings. … – But ferrocene is soluble in organic solvents like benzene and highly acidic compounds like nitric acid and concentrated sulphuric acid and etc.
Is ferrocene soluble in hexane?
The introduction of the hydroxyalkyl group to ferrocene strongly influences the solubility, causing a decrease in the solubility in hexane, an increase in the solubility in aromatic solvents, and a large increase in the solubility in alcohol, ether, and ketone.Is ferrocene organic or aqueous?
Ferrocene (Fc) is one of the very limited organic catholyte options for aqueous organic flow batteries (AOFBs), a potential electrochemical energy storage solution to the intermittency of renewable electricity.
What is acetyl ferrocene soluble?
CAS Min %94.0Quantity25gSolubility InformationSolubility in water: insoluble. Other solubilities: soluble in ethanolFormula Weight228.07Physical FormNeedle-Like Crystalline Powder
Is ferrocene soluble in water?
Ferrocene is an air-stable orange solid with a camphor-like odor. As expected for a symmetric, uncharged species, ferrocene is soluble in normal organic solvents, such as benzene, but is insoluble in water. It is stable to temperatures as high as 400 °C.
Is ferrocene toxic?
May be harmful if swallowed. May cause liver damage. May cause blood abnormalities. May cause eye, skin, and respiratory tract irritation.What is the melting point of Acetylferrocene?
Compound FormulaC12H12FeOMolecular Weight228.07AppearanceOrange to brown powder, crystals, or chunksMelting Point81-83 °CBoiling Point160-163°C /4mm
Is ferrocene paramagnetic or diamagnetic?Nickelocene and ferrocene have similar molecular structure and they are isostructural at room temperature. However, their thermal behaviors are quite different. From a magnetic viewpoint, nickelocene is paramagnetic with the electron spin S=1 (the ground state configuration 3A2g), while ferrocene is diamagnetic.
Article first time published onWhat kind of compound is ferrocene?
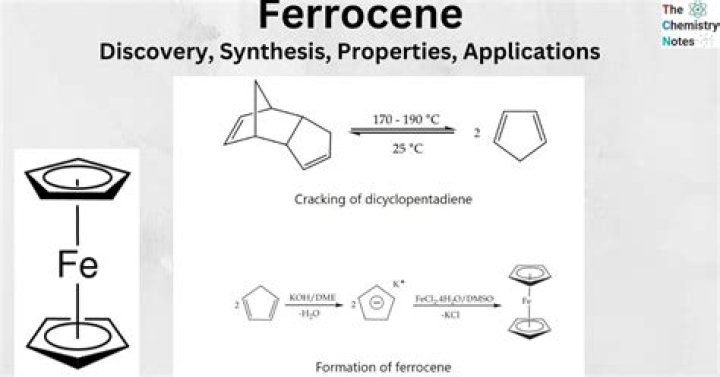
Ferrocene is an organometallic compound of the general class metallocene with the molecular formula Fe(η5-C5H5)2. In this molecule, iron is sandwiched between two cyclopentadienyl rings in staggered conformation, as shown in Figure 6.
How is ferrocene prepared in the laboratory?
Ferrocene can be prepared on a laboratory or large-scale basis by the reaction of ferrous chloride and sodium cyclopentadienide in an amine solvent. The basicity of the latter is an important factor in the yield of ferrocene obtained.
Is ferrocene polar or nonpolar?
Answer: Diacetylferrocene is most polar; ferrocene is least polar.
Why is ferrocene easily oxidised?
Ferrocene has mp 173o and, although stable to sulfuric acid, it is readily oxidized by nitric acid to the less stable ferricinium ion: … In cobalticinium ion, the metal has the 18 outer-shell electrons characteristic of krypton. Many other unsaturated organic compounds can form π complexes with transition metals.
Which hybridization is found in ferrocene?
orbitals of Fe2+ as shown below in Fig. Fig.: d2sp3 hybridization of Fe2+ ion in the formation of ferrocene, Fe(C5H5)2 Page 19 19 However, the aromaticity of ferrocene implies a ready availability of π-electrons in each ring and this is inconsistent with the involvement of six electrons in bonding with the metal.
What is the solubility of aspirin?
It has a limited solubility in water, which amounts to 2–4 mg/mL and its solubility varies significantly with temperature [1]. Aspirin is more soluble in ethanol, ethyl ether, chloroform, sodium hydroxide solution, and sodium carbonate solution than in water.
What is Hapticity in organometallic chemistry?
Hapticity is the coordination of a ligand to a metal center via an uninterrupted and contiguous series of atoms. … In addition, if the ligand coordinates through multiple atoms that are not contiguous then this is considered denticity (not hapticity), and the κ-notation is used once again.
What is acetyl ferrocene used for?
Its ferrocenyl derivative has wide applications to biological and medical fields such as ferrocene-modified beta-lactam because of its physiological activity of anti-malarial, anti-tumor, bactericidal, anti-inflammatory, treatment of anemia, inhibition of enzymatic activity and so on by virtue of its unique structure …
Is Acetylferrocene more polar than 9 Fluorenone?
This makes sense with the results, since Ferrocene is nonpolar, 9fluorenone is slightly polar with its carbonyl group, and acetylferrocene is polar with its acetyl group.
What is the difference between hexane and n-hexane?
The answer is that hexane and n-hexane are related as n-hexane is an isomer of hexane and the key difference is in their structures. Hexane has a branched structure and n-hexane has an unbranched structure.
What is the difference between hexane and hexanes?
What’s the difference? Hexane (or n-hexane) is essentially pure straight-chain C6H14. Mixed hexanes are a mix which consists primarily of n-hexane and several materials (including structural isomers) which are more difficult and expensive to separate from n-hexane. Mixed hexanes are much less expensive than n-hexane.
Which substances are soluble in hexane?
Type of SubstanceSoluble in Water?Soluble in Hexane?Ionic CompoundsOftenNoMolecular Compounds with Nonpolar MoleculesNoYesMolecular Compounds with Small Polar MoleculesUsuallyOften
What is the melting point of ferrocene and Acetylferrocene?
EXPERIMENT #2: Ferrocene and Acetylferrocene The melting point should equal or exceed 171 °C ( lit., 173-174 °C).
What color is Diacetylferrocene?
Compound FormulaC14H14FeO2Molecular Weight270.1AppearanceRed to brown crystals, chunks, or powderMelting Point122-128 °CBoiling PointN/A
What is the signal word for acetone?
H225: Highly flammable liquid and vapour. H319: Causes serious eye irritation H336: May cause drowsiness or dizziness P210: Keep away from heat/sparks/open flames/hot surfaces – No smoking. P305+351+338: IF IN EYES: Rinse cautiously with water for several minutes.
What are the main hazards of hexane?
- Acute Effects:
- Acute inhalation exposure of humans to high levels of hexane causes mild CNS depression. …
- include dizziness, giddiness, slight nausea, and headache in humans. ( …
- Acute exposure to hexane vapors may cause dermatitis and irritation of the eyes and throat in humans. (
Is ferrocene volatile?
Ferrocene (dicyclopentadienyl iron; CAS No. 102-54-5) is a relatively volatile compound used as a chemical intermediate, a catalyst, and an antiknock additive in gasoline.
Does ferrocene have a dipole?
Ferrocene itself has no (when the rings thermally rotate) or only a small possible dipole moment (as a static molecule) since it is a symmetric molecule.
What is oxidation state of iron Fe in ferrocene?
The convention used is that the organic moiety (C5H5) is usually assigned a charge of -1 In ferrocence, (η5-C5H5) Fe, iron has an oxidation state of +2.
What are ferrocene discuss the method of preparation and chemical properties of ferrocene?
Ferrocene is a p-complex in which reactions between the d-orbitals of the Fe2+ metal centre with the p-orbitals of the two planar cyclopentadienyl ligands (C5H5-) form the metal-ligand bonds. Hence there is equal bonding of all the carbon atoms in the cyclopentadienyl rings to the central Fe2+ ion.
How many P bonds are present in ferrocene?
Hence there is 12 $\pi $-electrons available in the ferrocene.