Are phospholipid tails polar or nonpolar
 Isabella Bartlett
Isabella Bartlett Phospholipids consist of a glycerol molecule, two fatty acids, and a phosphate group that is modified by an alcohol. The phosphate group is the negatively-charged polar head, which is hydrophilic. The fatty acid chains are the uncharged, nonpolar tails, which are hydrophobic.
Are phospholipid tails polar?
Phospholipids are amphiphilic. They have a polar head and two hydrocarbon tails, which are nonpolar.
Do phospholipids have a polar and nonpolar end?
Phospholipids have both a polar, hydrophilic end, and a nonpolar, hydrophobic end. Phospholipids are partially soluble in water, meaning that part of the molecule is attracted to water, and part of it is not.
Are the phospholipid tails hydrophilic?
1: A phospholipid consists of a head and a tail. The “head” of the molecule contains the phosphate group and is hydrophilic, meaning that it will dissolve in water. The “tail” of the molecule is made up of two fatty acids, which are hydrophobic and do not dissolve in water.Why is the tail of a phospholipid nonpolar?
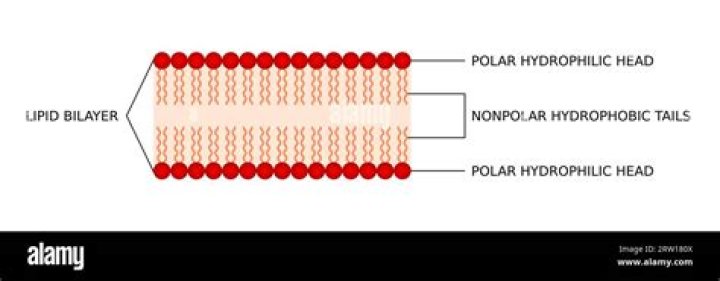
Thus, both surfaces of the plasma membrane are hydrophilic. In contrast, the interior of the membrane, between its two surfaces, is a hydrophobic or nonpolar region because of the fatty acid tails. This region has no attraction for water or other polar molecules (we will discuss this further in the next page).
Is plasma membrane polar or nonpolar?
The main component of the cell membrane is a phospholipid bi-layer or sandwich. The heads (the phospho part) are polar while the tails (the lipid part) are non-polar.
Are phospholipids hydrophilic or hydrophobic?
Phospholipids. Phospholipids consist of two hydrophobic “tails,” which are fatty acid chains, and one hydrophilic “head,” which is phosphate group. They connect with glycerol and the “head” is typically found at the sn-3 position.
Why are the tails of a phospholipid hydrophobic?
The tail of the phospholipid is hydrophobic because it is composed of carbon and hydrogen atoms.Is a phospholipid soluble in water?
2 Phospholipids. … Phospholipids are soluble in both water and oil (amphiphilic) because the hydrocarbon tails of two fatty acids are still hydrophobic, but the phosphate group end is hydrophilic.
How do polar and nonpolar phospholipids interact?They interact with other non-polar molecules in chemical reactions, but generally do not interact with polar molecules. … The hydrophilic regions of the phospholipids tend to form hydrogen bonds with water and other polar molecules on both the exterior and interior of the cell.
Article first time published onWhy is phospholipid head polar?
A single phospholipid molecule has a phosphate group on one end, called the “head,” and two side-by-side chains of fatty acids that make up the lipid “tails. ” The phosphate group is negatively charged, making the head polar and hydrophilic, or “water loving.” The phosphate heads are thus attracted to the water …
What is the tail of a phospholipid made of?
The phospholipids have a hydrophilic (water attracting) heads and two hydrophobic (water repelling) tails. The head of a phospholipid is made of an alcohol and glycerol group, while the tails are chains of fatty acids.
What is a phospholipid molecule?
In general, phospholipids are composed of a phosphate group, two alcohols, and one or two fatty acids. … On one end of the molecule are the phosphate group and one alcohol; this end is polar, i.e., has an electric charge, and is attracted to water (hydrophilic).
Is the hydrophobic tail polar or nonpolar?
Both stearic acid (a fatty acid) and phosphatidylcholine (a phospholipid) are composed of chemical groups that form polar “heads” and nonpolar “tails.” The polar heads are hydrophilic, or soluble in water, whereas the nonpolar tails are hydrophobic, or insoluble in water.
What are the nonpolar parts of phospholipids quizlet?
A phospholipid is made of a polar head (which includes the phosphate group and the glycerol molecules) and 2 nonpolar fatty acid tails. The head is hydrophilic and the tails are hydrophobic. Phospholipids make up cell membranes and form a bilayer.
Which portion of a phospholipid is hydrophilic?
The hydrophilic, or “water-loving,” portion of a phospholipid is its head, which contains a negatively charged phosphate group as well as an additional small group (of varying identity, “R” in the diagram at left), which may also or be charged or polar.
Is methyl hydrophobic or hydrophilic?
The only hydrophobic group below is the methyl (CH 3start subscript, 3, end subscript) group, which is nonpolar. The remaining six functional groups in the table all have varying degrees of hydrophilic character.
How do you know the phospholipid tails are water fearing and the heads are water loving?
A phospholipid is made up of a polar, phosphorus-containing head, and two long fatty acid (hydrocarbon), non-polar “tails.” That is, the head of the molecule is hydrophilic (water-loving), and the tail is hydrophobic (water-fearing). … The polar head group and fatty acid chains are attached by a 3-carbon glycerol unit.
What substances are polar?
- Water – H2O.
- Ammonia – NH. …
- Sulfur dioxide – SO. …
- Hydrogen sulfide – H2S.
- Ethanol – C2H6O.
Why do phospholipids surrounding the cell form a bilayer?
Why do the phospholipids surrounding the cell form a bilayer? because the properties of polar heads and nonpolar tails cause the phospholipids to arrange themselves in layers. … Some proteins extend through one or both phospholipid layers and help materials cross the membrane.
Why do the phospholipids form a bilayer?
Why do Phospholipids form bilayers? … -Phospholipids are amphipathic with a hydrophilic phosphate group and one or two hydrophobic hydrocarbon tails. – They form bilayers because the hydrophobic hydrocarbon tails will be shielded from interacting with water and will form noncovalent interactions.
Is a phospholipid a solid or liquid?
Because of their amphiphilic character, phospholipids exhibit various thermotropic and lyotropic phase structures, from solid-like lamellar to liquid phases. Most of the PLs exhibit a 3-D lamellar crystalline structure at low temperature and/or hydration level.
What type of molecules interact with water polar or nonpolar?
Water interacts best with polar molecules.
What is the function of phospholipids in plasma membrane?
Phospholipid bilayers are critical components of cell membranes. The lipid bilayer acts as a barrier to the passage of molecules and ions into and out of the cell. … These proteins form channels through which certain specific ions and molecules are able to move.
Why do phospholipids orient themselves tail to tail in a bilayer?
The polar head region in the phosphate group of the molecule is hydrophillic (attracted to water), while the fatty acid tail is hydrophobic (repelled by water). When placed in water, phospholipids will orient themselves into a bilayer in which the non-polar tail region faces the inner area of the bilayer.
Why are the tails hydrophobic quizlet?
Phospholipids have both hydrophobic and hydrophilic regions in a single molecule. The phosphate head group is hydrophilic because it is polar, enabling it to form hydrogen bonds with water. IN CONTRAST, the two long fatty acid tails are hydrophobic because they are nonpolar and do not form hydrogen bonds with water.
What is the hydrophobic tail made of?
The hydrophilic head is made up of Gycerol and a phosphate group – it is the phosphate group which makes the head hydrophilic. The hydrophobic tail is made up of 2 fatty acid chains, one of which usually contains a Cis double bond (C=C).
What does hydrophobic tail do?
Hydrophobic tails face inward and hydrophilic heads face outward. If you get these two ends mixed up, think of the root word “phobia” which means “fear.” Hydrophobic tails fear the water, so they will always try to be as far as possible from the water solutions in and out of the cell.
How does the polar head of a phospholipid react to water?
If you were to add small amounts of phospholipid molecules to water, they would float to the surface and align so that the polar head groups awere in the water, and the non-polar fatty acid tails would stick up from the surface of the water and form an oily film.
What do hydrophilic tails do?
A single phospholipid has a hydrophilic head and a hydrophobic tail. A phospholipid bilayer contains two connected layers of phospholipids with the tails of each layer meeting. … Hydrophilic means water loving and attracts water molecules while hydrophobic means water fearing and pushes water molecules away.
Which part of a phospholipid is polar and why?
Explanation: The phosphate heads are polar, which allows them to mix with water. This is why they are on the external and internal part of the cell membrane (because there’s a lot of water that cells come into contact with externally, and there’s a lot of water internally too).