Is sugar dissolved in water a pure substance
 Rachel Young
Rachel Young Sugar dissolves and is spread throughout the glass of water. The sand sinks to the bottom. The sugar-water is a homogenous mixture while the sand-water is a heterogeneous mixture. Both are mixtures, but only the sugar-water can also be called a solution.
Is a solution of sugar dissolved in water a pure substance?
YES -Homogeneous mixtureNO -Heterogeneous mixture
When sugar is dissolved in water what is it called?
Answer: When Sugar is dissolved in water, Sugar is the solute, water is the solvent and sweet water is solution respectively.
Is sugar solution a pure substance or mixture?
Sugar solution is a heterogenous mixture.Is sugar a pure substance?
Sugar is a pure substance.
Why does sugar completely dissolve in water?
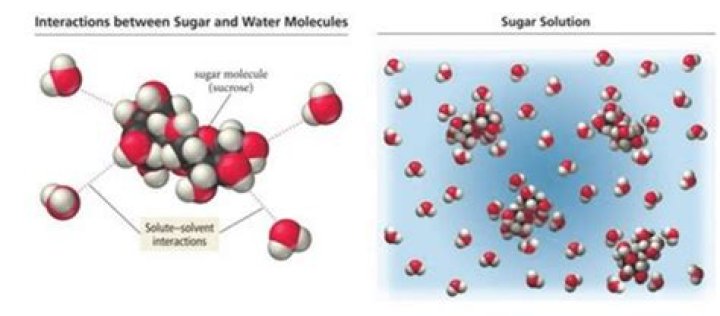
The sucrose molecules are attracted to one another by positive and negative polar areas. The polar water molecules attract the oppositely charged polar areas of the sucrose molecules and pull them away, resulting in dissolving.
Why is sugar water a solution and not a pure substance?
Sugar solution is a mixture because it can be seperated into sugar and water by evaporation. a mixture is ehen two or more different substance is mixed but not chemically combined . sugar and water are also mixed not chemically combined.
Is sugar water an element or compound?
But most definitely a compound, regardless of which sugar you choose. Water : H2O, so two hydrogen atoms (H2) and one oxygen atom (O). So both sugar and water are chemical compounds. Together they are a mix of chemical compounds.Is sugar is a mixture?
Sugar is a compound that is formed by a combination of three atoms: carbon, hydrogen and oxygen. Since these three atoms are chemically joined with each other thus they form a compound in nature.
When sugar dissolves in water the sugar?Solid sugar consists of individual sugar molecules held together by intermolecular attractive forces. When water dissolves sugar, it separates the individual sugar molecules by disrupting the attractive forces, but does not break the covalent bonds between the carbon, hydrogen, and oxygen atoms.
Article first time published onIs water is a pure substance?
Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. Although water is the most abundant substance on earth, it is rarely found naturally in its pure form. Most of the time, pure water has to be created. Pure water is called distilled water or deionized water.
Is sugar a pure carbon?
Sugar is made up of carbon, hydrogen and oxygen atoms. It’s the way these atoms are connected that makes each type of carbohydrate different. In each molecule of table sugar there are: 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms. … The black stuff itself is mainly carbon.
Does sugar dissolve in water Yes or no?
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. The weak bonds that form between the solute and the solvent compensate for the energy needed to disrupt the structure of both the pure solute and the solvent.
Is sugar dissolving in water a chemical or physical reaction?
Dissolving sugar in water is a physical change because sugar molecules are dispersed within the water but the individual sugar molecules are unchanged.
Is sugar dissolved in water a homogeneous mixture?
Sugar dissolves and is spread throughout the glass of water. … The sugar-water is a homogenous mixture while the sand-water is a heterogeneous mixture. Both are mixtures, but only the sugar-water can also be called a solution.
Is sugar in water a element?
A compound is a substance formed when two or more elements are chemically joined. Water, salt, and sugar are examples of compounds.
Is sugar a mixture or compound or element?
Sugar is a compound. It’s a chemical combination of carbon, hydrogen, and oxygen.
What is sugar water chemistry?
When you stir a spoonful of sugar into a glass of water, you are forming a solution. This type of liquid solution is composed of a solid solute, which is the sugar, and a liquid solvent, which is the water. As the sugar molecules spread evenly throughout the water, the sugar dissolves.
Is sugar a conductor?
Solutions with ions conduct electricity. Because pure water has few ions, it is a poor conductor. Uncharged molecules that dissolve in water, like sugar, do not conduct electricity. … Why do some solutions conduct electricity?
Does sugar dissolve in cold water?
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come into contact with the sugar more often, causing it to dissolve faster.
Which is the pure substance?
Pure Substance: The substances that are free from any kind of mixture and contain only one kind of particle are pure substances. Examples of pure substances include iron, aluminum, silver, and gold. Mixtures: Substances that have two or more different particles are mixtures.
What is a pure substance answer?
Pure substances are substances that are made up of only one kind of particles and has a fixed or constant structure. Pure substances are further classified as elements and compounds. An element is a substance that consists of only one type or kind of atom.
How do you know if a substance is pure?
Impure substances tend to have a slightly lower melting point than the pure substance, and a broader melting temperature range. Pure substances can be identified by comparing the melting point found in the experiment with published reference data of what the melting point should be.
What kind of substance is sugar?
The white stuff we know as sugar is sucrose, a molecule composed of 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen (C12H22O11). Like all compounds made from these three elements, sugar is a carbohydrate.
Is sugar homogeneous mixture?
Sugar is a homogeneous mixture. A homogeneous mixture means a substance which dissolve in water.
What kind of matter is sugar?
Powdered and granulated solids such as sugar or salt crystals might flow like liquids–but under a microscope they are definitely solid. The tiniest pieces of a solid are close together and don’t move around much. Solid things tend to be heavier or denser than liquids or gases.
Why does sugar not dissolve in a solid?
Once the attraction becomes large enough the water is able to pull individual sugar molecules from the bulk sugar crystals into the solution. … At some point the solution becomes saturated. This means that if you add more of the compound, it will not dissolve anymore and will remain solid instead.
Is sugar dissolved in water reversible?
Dissolution of sugar in water is a physical change because it does not result in the formation of a new substance. It is a reversible change as sugar and water can be extracted back from the solution by the process of crystallization followed by evaporation.
Is sugar burning a chemical change?
Burning a sugar cube is a chemical change. Fire activates a chemical reaction between sugar and oxygen. The oxygen in the air reacts with the sugar and the chemical bonds are broken.
Why is the dissolving of sugar in water a physical change quizlet?
Because you are not changing the object completly you are just changing the shape or size of the object. Describe what happens to the sugar molecules when they are dissolved in water. sugar dissolves, the sugar molecules become spread out between the water molecules. Do the molecules of sugar change once dissolved?