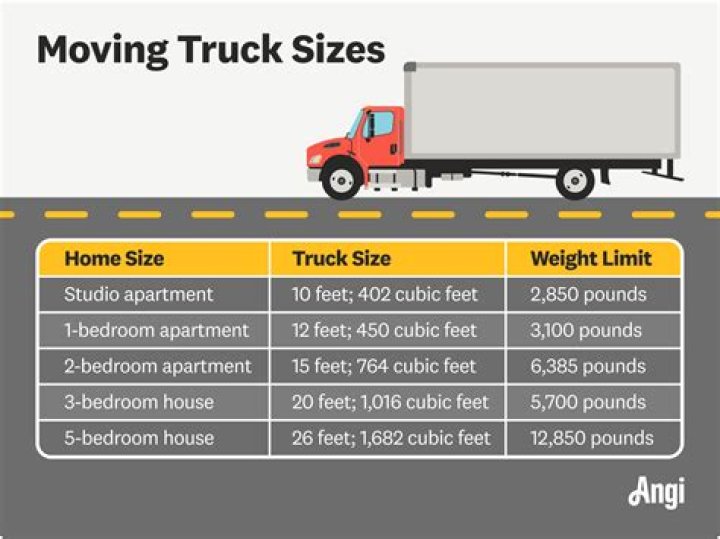
Will phosphoric acid clean copper
 William Burgess
William Burgess Hydrochloric and phosphoric acid don’t oxidize metals well and won’t dissolve copper.
What is the best chemical to clean copper?
- Citric acid- Citric acid is an organic acid which is used to clean copper. …
- Acetic acid- This is perhaps one of the most commonly used materials to clean copper. …
- Acetone- Acetone is an organic compound which is commonly used as a solvent.
What acid will dissolve copper?
Copper metal “dissolves” in nitric acid (HNO3). Actually, the nitrate ion oxidizes the copper metal to copper (II) ion while itself being transformed to NO2 gas in the process; the copper (II) ion then binds to six water molecules.
Does phosphoric acid etch copper?
The etching rate of aluminium etching based on H3PO4/HNO3 mixtures is strongly temperature-depend- ent and doubles every few degrees of temperature rise. … Copper is etched by this Al-etchant much more greatly than aluminium, nickel is comparatively weakly etched.How do you clean badly tarnished copper?
Vinegar and Salt Rub a mixture of 1 tablespoon of table salt and 1 cup of white vinegar onto the copper with a soft cloth and rinse. Or, immerse the tarnished copper into a pot of 3 cups of water and the salt-vinegar mixture, bring to a boil and boil until the grime and tarnish comes off.
Will Sulphuric acid clean copper?
Take some concentrated sulfuric acid, add about 5% by weight or potassium dichromate or chromic acid and stir till dissolved. This will give a thick dark red/brown solution that will immediately eat up what ever it touches, but it will usually clean copper quite beautifully.
What will make copper shiny?
- Mix together one-quarter cup salt and one-quarter cup flour, then add enough vinegar to make a thick paste.
- Rub that paste on the copper surface and gently buff it using a microfiber cloth until it shines; wash and dry thoroughly.
What does hydrochloric acid do to copper?
There will be no reaction. Copper is a very unreactive metal, and it does not react with hydrochloric acid. It is above copper in a metal reactivity series, so copper cannot replace the hydrogen in HCl to form CuCl2 .What does copper and phosphoric acid make?
Both are oxyacids of phosphorus. Yes, if you dissolve them both in the same solution and the concentrations are high enough, the copper ions from the copper chloride and phosphate ions from the phosphoric acid will combine to form an insoluble blue solid, copper phosphate.
What will muriatic acid do to copper?Copper is often associated with noble metals for its resistance to non-oxidizing acids like muriatic (hydrochloric) acid. … The catalyst in this case, is oxygen which bonds with copper to form copper oxide. Once bonded, the presence of oxygen enables muriatic acid to attack copper and over time dissolve it.
Article first time published onHow do you dissolve copper quickly?
Pour hydrogen peroxide in a glass. Hydrogen peroxide will help to oxidize the copper. Unfortunately, it can degrade pretty quickly, making your solution weaker. Start off with about 30 mL of hydrogen peroxide in a beaker.
Does vinegar break down copper?
Most pennies that have been around for a while have dark spots of a compound called copper oxide. … The combination of vinegar (a weak solution of acetic acid), and table salt (sodium chloride) helps to dissolve the copper oxide, and also forms the blue copper(II) ion, which is soluble in water.
How does acid clean copper?
The oxygen in the air and the copper in the pennies form an oxide that coats the pennies and makes them look dirty. The acids (usually vinegar – acetic acid) break the copper oxide free from the penny.
How do you make copper shiny again?
- Squeeze a little bit of ketchup on a rag, then sprinkle salt on top.
- Rub the mixture on your copper item in circular motions and repeat until the entire surface is tarnish-free.
- Use a damp cloth to wipe the ketchup and salt mixture off your item and use another clean cloth to dry your item.
How do you clean copper without removing patina?
A simple cleaning with mild soap and water will remove the dirt and fingerprints without removing tarnish or patinas that have formed over time.
How do you stop copper from oxidizing?
Paint Sealer A quick and easy way to prevent copper from tarnishing is to spray or paint it with a special type of sealer. This sealer is usually readily available in the market and do a very good job of keeping an airtight seal on copper pieces.
How do you clean copper with Coke?
Copper Cleaner Restore that bright sheen by soaking the copper bottom in a pan filled with Coke for about an hour. The acid in the Coke makes the copper look shiny and new—without scrubbing. This cleaning trick works on plenty of other household copper accents as well.
How do I get the green off my copper pipe?
Wet a rag with acetone. Wipe the green section to remove the patina from the copper pipes. Acetone counteracts the patina and restores the copper coloring. Wear eye protection, gloves and a respirator to avoid acetone contact with skin and membranes.
How do you clean copper molds?
- Put the stopper in sink. Add two to three drops of dish soap. …
- Submerge the copper Jell-O mold. Any remaining Jell-O will melt off as it warms. …
- Rinse off the mold with clean, hot water.
- Pour 1/8 cup of vinegar into a bowl. …
- Pour some of the paste onto a soft cloth. …
- Hold the mold under running water to remove the paste.
Will muriatic acid eat brass?
Despite your best attempts to clean it, it just won’t return to its former brightness. Using hydrochloric acid, also commonly referred to as muriatic acid, can help restore your brass. The acid eats away at the outer dull layer, exposing a fresh, bright layer underneath.
How do you clean an old copper vessel?
Create a mixture of one tablespoon of salt and one cup of white vinegar. After mixing it well add some flour and make it a paste. Apply this paste on the affected vessel leave it for fifteen minutes. Then rinse it with warm water and polish.
Does Brasso clean copper?
Brasso Brass , Copper and Metal Polish – Wipes away tarnish on most metals quickly and easily, leaving a brilliant shine. … Cleans, polishes and protects brass, pewter, chrome, copper, and stainless steel. Leaves a smooth, polished surface and a brilliant shine.
Does phosphoric acid react with copper sulfate?
Copper sulfate also reacts with phosphoric acid and forms sulfuric acid, but it’s hardly accessible in prison, I reckon.
Does copper corrode in acid?
Acid does not corrode copper.
Does citric acid dissolve copper?
Citric acid is commonly found in most citrus fruits such as pineapples and oranges, and is most concentrated in lemons. Citric acid cannot dissolve copper or most other metals. However, it does react with and dissolve copper oxide.
Why is copper not react with hydrochloric acid?
Copper is a less reactive metal, hence does not react with any acid. … But Cu does not react with HCl because the reduction potential of Cu is higher than that of hydrogen. Only metals which have their reduction potential less than that of hydrogen react with non – oxidising acids.
Can you acid wash copper?
Soak in muriatic acid (hydrochloric) approximately 1 hour. Sometimes toilet bowl cleaners contain this acid. This soaking removes all the green/brown oxidation from copper, plus any calcite is dissolved. Rinse well and brush the copper and rock again to remove any further loose rock.
How do you make copper green fast?
Soaking copper in white vinegar and salt will create a blue or green patina. Other ways of doing this are to bury the copper in sawdust or crushed potato chips soaked in white vinegar. The longer the copper is buried, the darker the patina becomes.
Does sodium hypochlorite react with copper?
My first objective was to see what each chemical did to the copper. … I expect the chlorine bleach — sodium hypochlorite, NaClO, with caustic soda, NaOH — to react with copper to produce cupric chloride, CuCl2, and possibly cupric hydroxide, Cu(OH)2.
Does hydrogen peroxide dissolve copper?
The rate of dissolution of copper in hydrogen peroxide solution goes through a maximum then drops at higher peroxide concentrations. … The dissolution behavior of copper is hydrogen peroxide is explained by carefully examining the chemical conditions at the metal/solution interface.
Will HCl and hydrogen peroxide dissolve copper?
According to a link on the pdf, to dissolve a pound of copper, it takes 1.87 gallons of 3% H2O2 and . 32 gallons of HCl.