Why water potential of a cell is always negative
 William Burgess
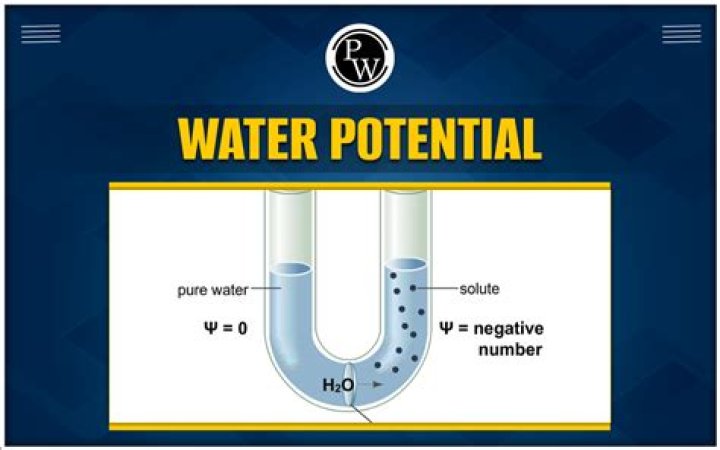
William Burgess Water pontential is negative when some solute is dissolved in pure water. So solution has fewer free water and the concemtration of water decreases reducing it’s water potential. The magnitude of this lowering is due to dissolution of solute called solute potential which is always negative.
Is water potential is always negative?
Water potential of pure water is taken as zero at a standard temperature so solute potential ( s) is always negative. The more the solute concentration, the lower or more negative is the solute potential.
Which potential is always negative for a cell?
Ψs is always negative. The more the solute molecules, the lower (more negative) is the solute potential Ψs water potential of a cell is affected by both solute and pressure potential.
What does a negative water potential mean?
If the flow is not restricted, water will move from an area of higher water potential to an area that is lower potential. A common example is water with dissolved salts, such as seawater or the fluid in a living cell. These solutions have negative water potential, relative to the pure water reference.Why is solute potential always negative explain?
– Solute potential is always negative because, increase in solute concentration lowers the solute potential which further decreases the overall water potential of the solution. – Distilled water or pure water’s solute potential is always zero because they don’t contain any free solute in their solution.
Which one of the following has more negative value of water potential?
The osmotic potential has important implications for many living organisms. The water potential of this solution will be negative. So, the correct answer is an option (C) – Distilled water.
Can water potential be greater than zero?
It can also be described as a measure of how freely water molecules can move in a particular environment or system. It is measured in kilopascals (kPa) and is represented by the Greek letter Psi (Ψ). Water potential is never positive but has a maximum value of zero, which is that of pure water at atmospheric pressure.
Where is water potential most negative in a plant?
The highest water potential we find in plants is zero, and water will always moves into areas of more negative water potential. The most negative areas of a plant are at the top where evaporation is occurring, and the least negative are in the roots.Is higher water potential more negative?
Water always moves from the system with a higher water potential to the system with a lower water potential. … The internal water potential of a plant cell is more negative than pure water; this causes water to move from the soil into plant roots via osmosis..
Why does dehydration decrease water potential?If the potato is allowed to dehydrate by sitting out in the open air the water potential would decrease (be more negative) because the concentration of solutes within the cells would increase as potato cells dehydrate. Therefore, the osmotic pressure and water potential both decrease.
Article first time published onWhen can water potential be positive?
posItIve adjacent to cells with a water potential less than zero would be (I) if the xylem sap were separated from the living cells by a water impermeable barrier, or (2) if there were an active pumping of water into the xylem.
Why does water potential affect animal cells differently than plant cells?
Osmosis affects plant and animal cells differently because plant and animal cells can tolerate different concentrations of water. In a hypotonic solution, an animal cell will fill with too much water and lyse, or burst open.
Can solute potential be negative?
Solute potential (Ψs), also called osmotic potential, is negative in a plant cell and zero in distilled water.
What is water potential of pure water?
The water potential of pure water is zero. If it is converted into a solution of 0.1 M concentration, the water potential.
Why is solute potential negative in osmotic pressure?
Because of the presence of solute particles, osmotic potential is the free energy of water in a system. This osmotic potential value is always negative since the presence of solute always produces a solution of less water than the same amount of pure water (means no solute particles are present).
What happens when water potential is 0?
In order for water to displace another object, water must be moving. The largest water potential any volume of water can have, if only standard atmospheric pressure is being applied to that volume of water, is defined as 0. This is the water potential for distilled water.
What factors affect water potential?
- Ψs will decline.
- Ψtotal will decline.
- the Δ between the cell and the surrounding tissue will decline.
- water will move into the cell by osmosis.
- Ψp will increase.
Which of the following does not affect water potential of water?
Atmospheric pressure .
Why is the water potential of sugar solution more negative?
Answer: A solution will have a lower and hence more negative water potential than that of pure water. Furthermore, the more solute molecules present, the more negative the solute potential is. Osmotic potential has important implications for many living organisms.
Why does pressure increase water potential?
Because pressure is an expression of energy, the higher the pressure, the more potential energy in a system, and vice versa. Therefore, a positive Ψp (compression) increases Ψtotal, and a negative Ψp (tension) decreases Ψtotal.
How does pressure potential affect water potential?
When the amount of solutes increases, osmotic potential decreases, and total water potential decreases. When the pressure increases, water potential increases. Both of these can be used to decrease water potential in specific areas, forcing the movement of high potential water into various cells of the plants.
What area in the tree has the highest water least negative potential?
Mesophyll of the upper most leaves.
How does water potential affect osmosis?
Factors Affecting the Rate of Osmosis Difference in Water Potential – The higher the difference in water potential, the faster the osmosis; for the lesser water molecules are in the region of low concentration, more water molecules from the region of higher concentration can enter faster and easier.
How does temperature affect water potential?
By increasing the temperature from 5 to 20 °C, the value of base water potential starts to decrease, but by increasing the temperature from 20 °C to 40 °C, base water potential start to increase (Table 3). The lowest water potential that enables the fulfillment of germination is known as the base water potential.
What is the relationship between water potential and solute concentration inverse or proportional?
Solute potential: All solutions have lower water potential than their pure form. Thus the water potential is inversely proportional to solute concentration, because free energy of water decreases.
Why are water potentials in plants soil air etc always negative numbers?
The internal water potential of a plant cell is more negative than pure water because of the cytoplasm’s high solute content. Because of this difference in water potential, water will move from the soil into a plant’s root cells via the process of osmosis.
Why don t plant cells burst when water enters them?
Tapwater and pure water are hypotonic. A single animal cell ( like a red blood cell) placed in a hypotonic solution will fill up with water and then burst. … Plant cells have a cell wall around the outside than stops them from bursting, so a plant cell will swell up in a hypotonic solution, but will not burst.
Why does water move out of the cell in a hypertonic solution?
Water moves into and out of cells by osmosis. If a cell is in a hypertonic solution, the solution has a lower water concentration than the cell cytosol, and water moves out of the cell until both solutions are isotonic.
Is the water potential on the right side of tube a positive or negative?
Water moves in response to the difference in water potential between two systems (the left and right sides of the tube). Positive water potential is placed on the left side of the tube by increasing Ψp such that the water level rises on the right side.
How does ionization constant affect water potential?
i Ionization constant Greater ionization decreases water potential/increases water movement, OR Decrease in ionization increases water potential/decreases water movement.
Do all solutions have higher water potential than pure water?
When some solutes are dissolved in water, the solution has less water molecules. This results in a decrease in the water potential of the solution. Hence, all solutions have lower water potential than pure water.





