Why is the half life of a first order reaction constant
 Isabella Bartlett
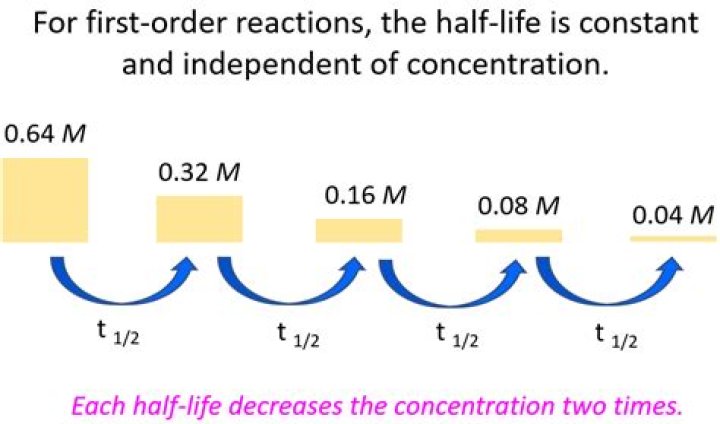
Isabella Bartlett In other words, the initial concentration of the reactant has no influence on the half-life of the reaction, i.e. the half-life is constant regardless of the concentration of the reactant.
Why do first order reactions have constant half-life?
The best way to determine rate constant k in half-life of first order is to determine half-life by experimental data. The reason is half-life in first order order doesn’t depend on initial concentration.
Is a first order reaction constant?
Because the units of the reaction rate are always moles per liter per second, the units of a first-order rate constant are reciprocal seconds (s−1).
Does the half-life of a first order reaction stay constant?
The half-life of a reaction is the time required for the reactant concentration to decrease to one-half its initial value. The half-life of a first-order reaction is a constant that is related to the rate constant for the reaction: t1/2 = 0.693/k.What is the half-life expression for a first order reaction?
The half-life equation for a first-order reaction is t12=ln(2)k t 1 2 = ln ( 2 ) k .
What is half-life period of reaction?
The half life (t1/2) of a reaction is the time in which the concentration of reactant is reduced to one half of its initial concentration [R]0 .
What is the half-life of first-order reaction if time is required to reduce concentration?
Concentration is reduced to 25%. It means it takes two half-lives to decrease the concentration of reactant from 0.8 M to 0.2 M in first-order reaction. Hence, half-life of the reaction is 12/2 = 6 hours.
Is half-life affected by temperature?
Half-lives can be calculated from measurements on the change in mass of a nuclide and the time it takes to occur. … Although chemical changes are sped up or slowed down by changing factors such as temperature and concentration, these factors have no effect on half-life.How do the half lives of first-order and second order reactions differ?
If we plot the concentration of a reactant versus time, we can see the differences in half lives for reactions of different orders in the graphs. … For a 1st order reaction (Half life is constant.) For a second order reaction (Half life increases with decreasing concentration.)
What is a first order rate constant?The elimination rate constant (usually a first-order rate constant) represents the fraction of xenobiotics that is eliminated from the body during a given period of time.
Article first time published onWhat is 1st order reaction?
Definition of first-order reaction : a chemical reaction in which the rate of reaction is directly proportional to the concentration of the reacting substance — compare order of a reaction.
Why is a reaction first order?
A first-order reaction can be defined as a chemical reaction in which the reaction rate is linearly dependent on the concentration of only one reactant. In other words, a first-order reaction is a chemical reaction in which the rate varies based on the changes in the concentration of only one of the reactants.
What will be the half-life of the first-order reaction for which the value of rate constant is 200?
Half life period (t1/2)=0.693k=0.693200s-1=3.465×10-3s.
What will be the half-life of the first-order reaction for which the value of rate constant is 200 2nd?
46×10−2s.
What is the time for a first order reaction to be 99 complete?
For first order reaction show that time required for 99% completion is twice the time required for the completion of 90% of reaction. For the first order reaction, time required for 99% completion. Hence, time required for 99% completion is twice for the time required for the completion of 90% reaction.
What are pseudo first order reaction give one example of such reactions?
Thus, the reaction appears to be first order, but it is actually of second order that’s why known as pseudo first order reaction. So, concentration of water can be approximated as constant as its concentration doesn’t change a lot during the reaction. thus, hydrolysis of cane sugar is pseudo first order reaction.
What is elementary reaction in chemistry?
An elementary reaction is a chemical reaction in which one or more chemical species react directly to form products in a single reaction step and with a single transition state. … An example of this type of reaction is a cycloaddition reaction.
What is half-life period of a reaction show that half-life period of a first order reaction is independent of the initial concentration?
The expression for the half-life period of a first order reaction does not contain a concentration term. Thus, the half-life period for such reactions is independent of the initial concentration of the reactants. Note: The unit of rate constant for first order reaction is sec−1 or min−1.
What is the half-life for the first order decay of carbon 14?
Carbon-14 is decaying constantly with a half-life of 5720 years.
Why does the half-life for second order decrease with increasing initial concentration?
This inverse relationship suggests that as the initial concentration of reactant is increased, there is a higher probability of the two reactant molecules interacting to form product. Consequently, the reactant will be consumed in a shorter amount of time, i.e. the reaction will have a shorter half-life.
Which of the following method uses a half-life to determine the order of reaction?
time (s)[A] (M)01.200100.800200.600300.480
Does the half-life of a second order reaction increase/decrease or remain the same as the reaction proceeds How does the half-life vary as the reaction proceeds?
The quantity [A]_0 decreases in concentration over the course of the reaction. As a result, since \large{t_{1/2} = \frac {1}{k[A]_0}} in a second-order reaction, the half-life of a second-order reaction gets longer as the reaction proceeds. as the reaction proceeds.
Are Half Lives constant?
Half-life is constant over the lifetime of an exponentially decaying quantity, and it is a characteristic unit for the exponential decay equation.
Does half-life of first order reaction depend on temperature?
Although the first-order half-life depends on the rate constant k and not the initial concentration [A]0 , k is a function of temperature, and increases with increasing temperature.
Why does half-life decrease at higher temperature?
As you have studied that half life of a first order reaction is constant at a given temperature because the formula of half life of a first order reaction is ln2/rate constant…and as you know that as the temperature increases the rate constant increases according to the Arrhenius equation so as the denominator …
Why is a first order reaction never completed?
Basically in a first order reaction the rate of the reaction is directly proportional to the concentration of the reactant left . so as the reaction proceeds the concentration of the reactant goes on decreasing and rate becomes more slow as time passes hence it never comes to end.
What is half-life of zero order reaction?
Answers. The half-life is 96 seconds. Since this is a zero-order reaction, the half-life is dependent on the concentration. In this instance, the half-life is decreased when the original concentration is reduced to 1.0 M.
What is the importance of the rate constant?
Hint: Rate constant gives us an idea about the factors on which the rate of a reaction depends. Knowing the rate constant will tell us the dependency of the molar concentration of the reactants with the speed of reaction.
What are the two first order reaction rate constant in terms of temperature are given below?
For the first order reaction rate constants in terms of temperature are given below A—–>P k1 =10^15e^-1500/T B—–>Q k2= 10^14e^-1000/T If initially both reaction started with same initial concentration of a and be then at what temperature both reactions will have same initial rate.
Which of these is not true for the order of a reaction?
The order of a reaction in the sum of the powers of molar concentration of the reactants in rate law expression. Out of the given four statements , option (c) is not correct. Order of reaction is equal to the sum of power of conentration of the reactants in rate law expression.
How is rate affected when the concentration of B is tripled?
(ii) When concentration of B is tripled, rate of reaction increases by 9 times.