Why does water adhesion happen
 Emily Sparks
Emily Sparks Water is very adhesive; it sticks well to a variety of different substances. Water sticks to other things for the same reason it sticks to itself – because it is polar so it is attracted to substances that have charges.
What causes adhesion in water?
Adhesion is caused by the polarity of water. Water molecules have an uneven sharing of electrons due to covalent bonding. This creates a negative and positive end of each water molecule. This results in water being attracted to other molecules.
What causes adhesion to occur?
Adhesions Causes Adhesions develop as the body attempts to repair itself. This normal response can occur after surgery, infection, trauma, or radiation. Repair cells within the body cannot tell the difference between one organ and another.
What causes adhesion and cohesion in water?
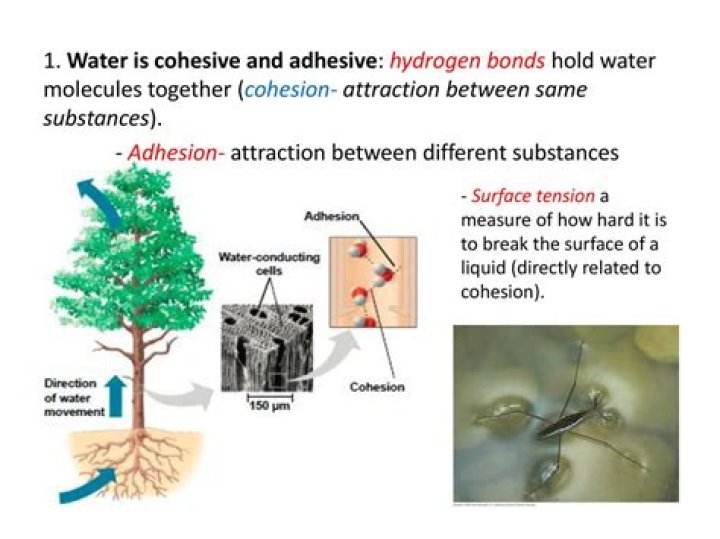
The very strong intermolecular bonds (called Hydrogen bonding) cause the cohesion molecules of water being attracted to one another. … The attraction of the polar water molecules for other molecules besides water account for the the properties of adhesion.What does adhesion mean in water?
Adhesion: Water is attracted to other substances. Adhesion and cohesion are water properties that affect every water molecule on Earth and also the interaction of water molecules with molecules of other substances.
Why does cohesion and adhesion occur?
Cohesion occurs when molecules are attracted to other molecules of the same type. Adhesion is when molecules are attracted to different substances.
Why does water stick to surfaces?
So water has a property called “cohesion” meaning the molecules like to stick to each other due to hydrogen bonding. This cohesion accompanied by surface tension causes water to run along surfaces, such as a glass tipped at an angle or your arms when washing your hands.
Why do hydrophobic molecules repel water?
Hydrophobic molecules and surfaces repel water. … Without opposite electrical charges on the molecules, water cannot form hydrogen bonds with the molecules. The water molecules then form more hydrogen bonds with themselves and the nonpolar molecules clump together.What causes adhesion and cohesion?
Adhesion is caused by electrostatic or mechanical forces that exist among two kinds of different substances. Cohesion is caused by hydrogen bonding and Van der Waals forces.
Why does water form a meniscus?Why a meniscus occurs Adhesion is responsible for a meniscus and this has to do in part with water’s fairly high surface tension. … And since water molecules like to stick together, when the molecules touching the glass cling to it, other water molecules cling to the molecules touching the glass, forming the meniscus.
Article first time published onWhat is a adhesion?
Adhesions are bands of scar-like tissue that form between two surfaces inside the body and cause them to stick together.
Why does ice float on liquid water?
Believe it or not, ice is actually about 9% less dense than water. Since the water is heavier, it displaces the lighter ice, causing the ice to float to the top.
What effect does adhesion produce?
Adhesion produces an effect called Capillary action when you place a straw in a liquid.
What is adhesion example?
Adhesion may refer to the joining of two different substances due to attractive forces that hold them. For instance, cohesion causes water to form drops and adhesion keeps the water drops on the surfaces of leaves and flowers in place. … This binding of the cells is referred to as cell adhesion.
How does adhesion affect flow?
4a) Adhesion affects flow rate because flow rate will be slower if it has more adhesion. This is because adhesion shows how strong the attraction between the particles and another substance are. The liquid will stick to the sides making it come outs lower.
Why does water stick to your hand?
First, ‘cohesion’: the attraction of water molecules to each other. This comes from the opposing electric charges on the hydrogen and oxygen atoms of neighbouring H2O molecules. And it’s this mutual attraction of water molecules in the damp layer on the skin that creates the feeling of stickiness.
Why do some water insects walk on water?
Water striders are small insects that are adapted for life on top of still water, using surface tension to their advantage so they can “walk on water.” … The attraction between water molecules creates tension and a very delicate membrane. Water striders walk on this membrane.
Why does water stick upside down?
When the angle between vertical direction and the glass wall is small, surface tension is stronger and the component of gravity perpendicular to the glass wall is small; consequently, water sticks to the outside surface of the container (or in other words, runs down the side of the container).
Why does water move from the roots to the leaves of plants?
As water molecules evaporate from plant leaves, they attract the water molecules still in the plant, helping to pull water up through the stems from the roots. The combination of transpira- tion and capillary action delivers the water from the bottom to the top of a plant.
What happens to hydrophobic molecules in water?
When a hydrophobe is dropped in an aqueous medium, hydrogen bonds between water molecules will be broken to make room for the hydrophobe; however, water molecules do not react with hydrophobe. This is considered an endothermic reaction, because when bonds are broken heat is put into the system.
What is the hydrophobic effect explain?
The hydrophobic effect describes the energetic preference of nonpolar molecular surfaces to interact with other nonpolar molecular surfaces and thereby to displace water molecules from the interacting surfaces. The hydrophobic effect is due to both enthalpic and entropic effects.
How do hydrophobic molecules react with water?
Hydrophobic molecules react with water, but with low affinity. When they are part of amphiphatic molecules, the hydrophilic regions of the molecule, of high affinity for water molecules, dictate the direction of reaction, and force the hydrophobic tails to cluster inward, producing insoluble drops (such as in fats).
Why water meniscus is concave?
A concave meniscus occurs when the particles of the liquid are more strongly attracted to the container (adhesion) than to each other (cohesion), causing the liquid to climb the walls of the container. This occurs between water and glass.
What is it called when water doesn't overflow?
When we fill the glass with water, we notice right away that it can go over the brim of the glass without spilling. This is because of surface tension. … This attraction causes the molecules to stick together and avoid spilling down the side of the glass like gravity would like them to.
Which of these is the best explanation for why the meniscus of water in the capillary tube to the right is concave?
tHis ability of water to “wet” glass makes it creep up the walls of the tube where the water surface touches the glass. … The concave shape of the meniscus shows that water’s adhesive forces toward the glass are stronger than its cohesive forces. Cohesive Forces. The intermolecular forces among the molecule of a liquid.
Are adhesions real?
One of the most common forms of pain in the body is due to fibrous adhesions. The formation of adhesions is the body’s response to injury, trauma, and overuse. These adhesions form in the soft tissue of the body, and over time, can build up to limit mobility, decrease strength, and cause pain.
What causes pelvic adhesions?
Pelvic Adhesions occur when a band of scar tissue in the pelvic region causes two pieces of tissue to become stuck together. It can result from surgeries, pelvic infections, or endometriosis and may affect organs such as the bladder, uterus, ovaries, and fallopian tubes.
How do you get rid of adhesions naturally?
Sitting in a warm bath or reclining with a hot water bottle when your pain flares up can help relax your muscles and soothe the pain from the adhesion. Your doctor may also recommend massage techniques and physical therapy to try to break up the scar tissue and lessen the pain.
What will float in water?
Objects like apples, wood, and sponges are less dense than water. They will float. Many hollow things like empty bottles, balls, and balloons will also float. That’s because air is less dense than water.
Why does water expand when it freezes?
When water freezes solid, at 32 degrees, it expands dramatically. … Each water molecule is two hydrogen atoms bonded to one oxygen atom (H2O). The H2O molecule’s slightly charged ends attract the oppositely charged ends of other water molecules. In liquid water, these “hydrogen bonds” form, break, and re-form.
Why does an object float in water?
An object floats when the weight force on the object is balanced by the upward push of the water on the object. The upwards push of the water increases with the volume of the object that is under water; it is not affected by the depth of the water or the amount of water.