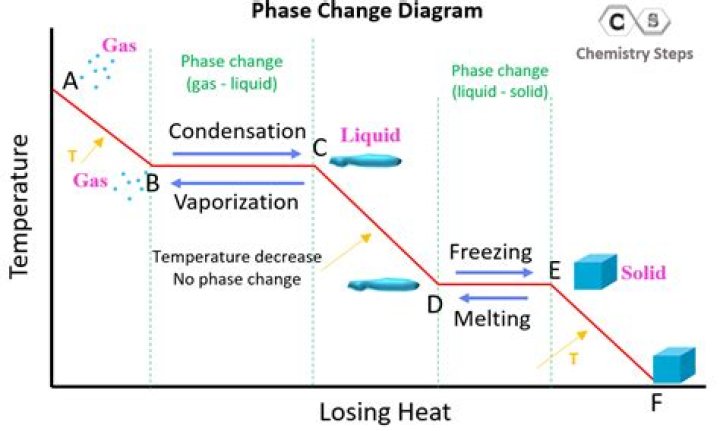
Why does the temperature remain constant during a phase change from E to F
 Isabella Bartlett
Isabella Bartlett Temperature remains constant during phase change because at that point, any energy added to the substance is used to change its phase rather than to change the temperature. In other words, during phase change, the distance between the molecules changes, but not the translational velocity of such molecules or particles.
Why does the temperature remain constant during the phase changes?
During a change of the state of matter, the supplied energy is not used to increase the kinetic energy of the molecules, but to change the binding energies. Therefore, the temperature remains constant.
Why does the temperature not change during phase changes?
But there is no temperature change until a phase change is complete. i.e. during phase change, the energy supplied is used only to separate the molecules ; no part of it is used to increase the kinetic energy of the molecules. So its temperature will not rise, since kinetic energy of molecules remains the same.
Why does the temperature of a substance remain constant during a phase change use a phase change as an example to explain what is happening on a particulate level?
During the phase change, when solid melts into liquid, its temperature remains constant as the heat energy is stored as potential energy. Likewise, as heat is added to a liquid, its temperature increases as the molecules, once again, move faster.Why does the temperature remain constant during its melting point or boiling point?
Answer: The temperature of a substance remains constant at its melting and boiling points until all the substance melts or boils because, the heat supplied is continuously used up in changing the state of the substance by overcoming the forces of attraction between the particles.
Why does the temperature remain constant during the melting of ice even though heat is supplied constantly?
The temperature remains constant during the melting of ice even though heat is supplied continuously because the temperature is supplied for changing the state of matter not for increasing the temperature.
Why does the temperature of a substance stay constant during a phase change such as vaporization?
Temperature remains constant during phase change because at that point, any energy added to the substance is used to change its phase rather than to change the temperature. In other words, during phase change, the distance between the molecules changes, but not the translational velocity of such molecules or particles.
How does the temperature affect the phases changes of matter?
Temperature affects phase change by slowing down the movement in between the atoms, thus causing a change in kinetic energy, which in turn causes the atoms to undergo forms of combining or a type of disepersion.Does temperature change when a phase change occurs?
During a phase change energy my be added or subtracted from a system, but the temperature will not change. The temperature will change only when the phase change has completed.
Why does the temperature remain constant during boiling though heat is constantly supplied?During the boiling of water the temperature stays constant while heat is supplied continuously. It is because the heat provided by the water particles is consumed, and this heat increases their kinetic energy. … Therefore, the temperature stays constant only though heat is continually supplied to the water.
Article first time published onWhy does the temperature of a substance remain constant during its melting or boiling point even when heat is being supplied to it continuously?
The temperature of a substances remains constant during melting and boiling even when heat is being supplied to it continuously because the heat that is supplied is being used up in changing a state of matter by overcoming intermolecular force of attraction. … therefore the temperature remains same.
Why the temperature of the melting ice does not change even though energy is being transferred as heat to ice?
No temperature change occurs from heat transfer if ice melts and becomes liquid water (i.e., during a phase change). … Similarly, energy is needed to vaporize a liquid, because molecules in a liquid interact with each other via attractive forces. There is no temperature change until a phase change is complete.
Why is there no change in temperature during a phase change even if energy is absorbed by the system?
Since the energy involved in a phase changes is used to break bonds, there is no increase in the kinetic energies of the particles, and therefore no rise in temperature.
What happens during phase change?
A phase change is a physical process in which a substance goes from one phase to another. Usually the change occurs when adding or removing heat at a particular temperature, known as the melting point or the boiling point of the substance. … Removing heat from a substance changes a gas to a liquid or a liquid to a solid.
What is the effect of temperature?
The rate of a chemical reaction can be changed by altering the temperature. If the temperature is increased: the reactant particles move more quickly. they have more energy.
How does the temperature affect the phase of water hypothesis?
2. Hypothesis: How does the temperature affect the phase of water? … [Students should note that as temperatures rise, solids turn into liquids, and liquids turn into gases.]
Why the temperature of boiling water does not rise even when heat is continuously supplied to it Class 9?
The heat which we supply to water during boiling is used to overcome these forces of attraction between the particles so that they become totally free and change into a gas. This latent heat does not increase the kinetic energy of water particles and hence no rise in temperature takes place during the boiling of water.
Why the temperature of boiling water does not rise even when heat is continuously supplied to it?
The temperature of boiling water does not rise even though heat is being supplied continuously because the heat which is supplied is used as latent heat of vaporization to convert boiling water into steam and it does not rises the temperature of boiling water .
Why does the temperature of water does not change till all of it has changed into Vapours?
All the heat that is supplied is utilized by the molecules of water to overcome the force of attraction between them and change into vapour. So there will be no change in the temperature.
Why does the temperature remain unchanged until the entire solid changes into liquid even if we are heating the solid?
This happens because the latent heat of fusion or melting gets hidden on the surface of the solid. … This also results in an unchanged temperature even though the solid melts into its liquid state.
Why there is no change in temperature during the melting of a solid?
When solid melts, the temperature remains the same, this is because the absorbed heat is utilized in breaking the forces of attraction between the particles and hence solid changes to a liquid state without changing in temperature.
Why does temp not change during freezing?
When freezing takes place, latent heat of fusion is discarded. When latent heat of fusion is discarded, material gets solidified. This process takes place at constant temperature, because latent heat is a constant quantity.
Why do phase changes occur?
Why Do Phase Changes Occur? Phase changes typically occur when the temperature or pressure of a system is altered. When temperature or pressure increases, molecules interact more with each other. When pressure increases or temperature decreases, it’s easier for atoms and molecules to settle into a more rigid structure.
What causes phase changes?
Phase changes require either the addition of heat energy (melting, evaporation, and sublimation) or subtraction of heat energy (condensation and freezing). … Changing the amount of heat energy usually causes a temperature change.