Why does fullerene have a low melting point?
 Emily Sparks
Emily Sparks .
Besides, does fullerene have a high or low melting point?
They are made up of large molecules but do not have a giant covalent structure. Weak intermolecular forces exist between individual buckyballs. Little energy is needed to overcome these forces, so substances consisting of buckyballs are slippery and have lower melting points than graphite or diamond .
Beside above, why does iodine have a low melting point? Iodine has a low melting point because of it's large size. I2 or iodine molecule has two iodine atoms bonded together. Since the size is very large,bonds are weak and melt under low temperature.
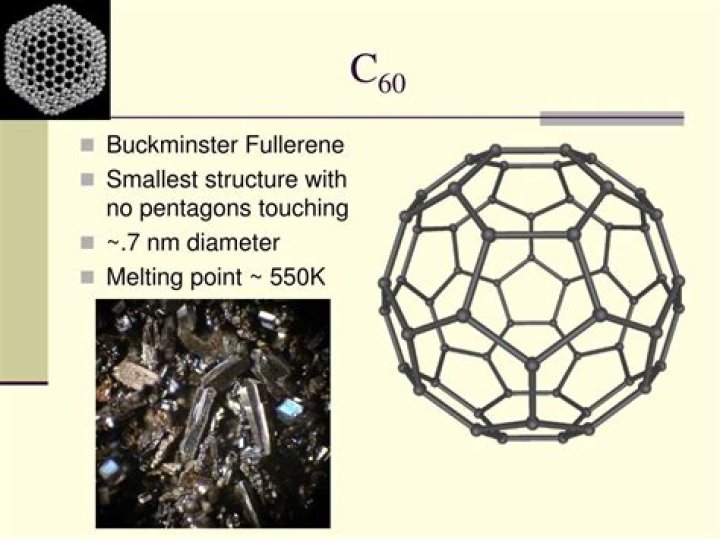
Additionally, why does c60 have a low melting point?
Its molecules are made up of 60 carbon atoms joined together by strong covalent bonds. There are weak intermolecular forces between molecules of buckminsterfullerene. These need little energy to overcome, so buckminsterfullerene is slippery and has a low melting point.
What are the properties of fullerenes?
Their unique molecular structure results in extraordinary macroscopic properties, including high tensile strength, high electrical conductivity, high ductility, high heat conductivity, and relative chemical inactivity (as it is cylindrical and "planar" — that is, it has no "exposed" atoms that can be easily displaced).
Related Question AnswersCan a diamond conduct electricity?
Diamond. Diamond is a form of carbon in which each carbon atom is joined to four other carbon atoms, forming a giant covalent structure. It does not conduct electricity as there are no delocalised electrons in the structure.Do nanotubes conduct electricity?
A metallic carbon nanotube conducts electricity when you connect different voltages to each end, just like a wire. Carbon nanotubes conduct electricity better than metals. When electrons travel through metal there is some resistance to their movement. This resistance happens when electrons bump into metal atoms.Is fullerene hard or soft?
The fullerene molecule has excellent mechanical rigidity. At the same time, the fullerite crystal is a soft material under normal conditions, but becomes harder than diamond under pressure (due to the 3-D polymerization).Can a c60 conduct electricity?
In fact C60 is diamagnetic (no unpaired electrons) and does not conduct electricity. Thus, this initial model for the bonding and electronic structure of C60 is not correct. A way out of this difficulty is to recall that C60 is not a sphere, but a truncated iscosohedron.Is graphene a metal?
The simplest way to describe graphene is that it is a single, thin layer of graphite — the soft, flaky material used in pencil lead. Despite this, graphene is also one of the strongest materials in the known universe. With a tensile strength of 130 GPa (gigapascals), it is more than 100 times stronger than steel.Does buckminsterfullerene conduct electricity?
Since each carbon atom has four bonding electrons, one electron remains unbonded in each carbon atom. These electrons form a sea of delocalised electrons similar to that found in a metal. These can move throughout the fullerene, allowing conduction of electricity.Do nanoparticles conduct electricity?
Like graphene, nanotubes are strong and conduct electricity because they have delocalised electrons. Nanotubes can be added to other materials, for example in sports equipment, to make them stronger.Why is graphene so conductive?
One of the most useful properties of graphene is that it is a zero-overlap semimetal (with both holes and electrons as charge carriers) with very high electrical conductivity. These pi orbitals overlap and help to enhance the carbon to carbon bonds in graphene.Does graphite melt or sublime?
Graphite does not sublime or melt at standard conditions, what would be the point of graphite lead in pencils, rather it forms network covalent bonds.What is the melting point of diamond?
In the absence of oxygen, diamonds can be heated to much higher temperatures. Above the temperatures listed below, diamond crystals transform into graphite. The ultimate melting point of diamond is about 4,027° Celsius (7,280° Fahrenheit).What is the melting point of buckminsterfullerene?
Buckminsterfullerene fast facts| CAS Reg. No. | 99685-96-8 |
|---|---|
| Empirical formula | C60 |
| Appearance | Shiny black needle-like crystals |
| Melting point | ≈600 ºC (subl.) |
| Water solubility | Insoluble |
What is the formula of buckminsterfullerene?
C60What are 4 properties of molecular compounds?
1 Answer- Have high melting points.
- Have high boiling points.
- Tend to be soluble in water.
- Conduct electricity in liquid form and in aqueous solution.
- Are usually hard and brittle solids.
- Have high enthalpies of fusion.
- Have high enthalpies if vaporization.