Why can you dissolve more sugar than salt in water
 Rachel Young
Rachel Young The reason for this is because the sugar molecules are bigger than the ions of dissolved salt. This allows for more water molecules to surround a single particle, pulling it into solution faster.
Does sugar dissolve more than salt?
For example, sugar is much more soluble in water than is salt. But even sugar has an upper limit on how much can dissolve.
Why does sugar dissolves faster than salt?
The reason for this is because the sugar molecules are bigger than the ions of dissolved salt. This allows for more water molecules to surround a single particle, pulling it into solution faster.
Why does more sugar dissolve in water?
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come into contact with the sugar more often, causing it to dissolve faster.Is sugar water more dense than salt water?
In tap water the egg sinks because the egg is denser than the water; the egg in salt water and sugar water floats because it is less dense than the water (salt and sugar increase the density of the water thus allowing the egg to float). Procedure: Collect all materials.
Why sugar does not dissolve in water?
Sugar has individual molecules in them joined together by comparatively weak forces. When sugar is put to water, these weak bonds get broken gradually and the individual molecules get released.
Why does sugar not fully dissolve in water?
Sugar Dissolving in Different Liquids Sugar dissolves well in water because water is very polar and interacts with the polar areas of sucrose. Sugar does not dissolve very well in alcohol because alcohol has a large part that is pretty non-polar. Sugar hardly dissolves at all in oil because oil is very non-polar.
What sugar dissolves the fastest?
A packet of granulated sugar would dissolve faster than a cube of sugar. The rate of dissolving would be increased by stirring, or agitating the solution. Finally, the sugar would dissolve faster in hot tea than it would in cold tea.Is sugar bigger than salt?
Here’s why: Salt is about 25% more dense than sugar. Therefore a teaspoon of salt weighs more than a teaspoon of sugar by almost 25%. Your dissolving test would not be accurate because you would be starting out with a larger mass of salt than sugar.
How much sugar can water dissolve?The maximum amount of sugar that will dissolve in a liter of 20 °C water is 2000 grams. A sugar-water solution that contains 1 liter of water and 2000 grams of sugar is said to be saturated.
Article first time published onWhy does sugar have higher solubility in hot water as compared to salt?
Most solids, including sugar and salt, become more soluble with increasing temperature. This is because heat increases molecular movement, causing more collisions between the water molecules and the solid. But the opposite is true for gases, which tend to become less soluble as a solvent’s temperature increases.
Is sugar solution heavier than water?
As you add sugar to the water, more and more sugar molecules take up the available space between water molecules, making the solution denser.
Does sugar weigh more than water?
An important example is water. … Thus 2 ml of water has a mass of 2 g etc.. Table sugar is more dense than water by about 60 percent.
Does sugar affect buoyancy?
Archimedes also found that some liquids have greater buoyancy than others. … Since salt or sugar water weighs more than an equal volume of fresh water, it has greater buoyancy. In other words, it can push up with greater force.
Is sugar completely dissolved in water?
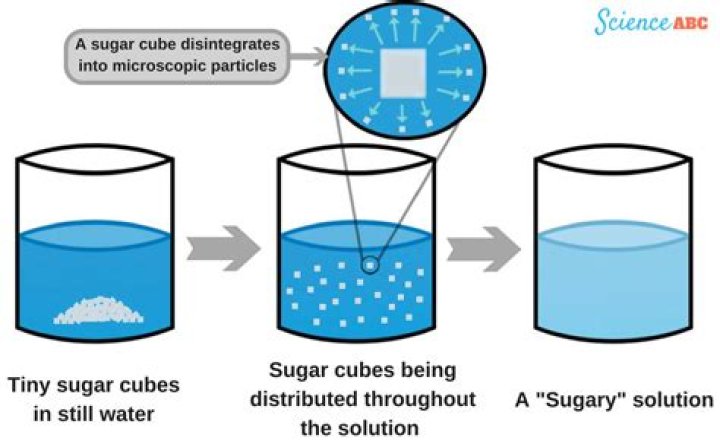
Sugar is and example of a solid that breaks down into tiny pieces, and dissolves in water. When the sugar is spread completely throughout the water, we call this a solution. Not everything dissolves, however. Rocks and sand do not dissolve in water—they sink to the bottom.
Why does some sugar not dissolve?
Note that if you add too many sugar cubes to the cup of water, they might not dissolve completely because the water may become saturated with the solute. In this case, some of the sugar will dissolve and the rest will gather in a solid state at the bottom of the cup.
What happens when sugar dissolves in water?
Solid sugar consists of individual sugar molecules held together by intermolecular attractive forces. When water dissolves sugar, it separates the individual sugar molecules by disrupting the attractive forces, but does not break the covalent bonds between the carbon, hydrogen, and oxygen atoms.
How sugar and water can be separated?
The sugar can be separated by removing the water from the solution. Therefore, distillation is the process by which sugar can be separated from the sugar solution. The solution is heated. The water in the sugar solution boils at the 1000C.
Why does salt dissolve in water?
When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. … Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.
What's the difference between salt and sugar?
They are also very different chemically. Salt is made up of sodium and chloride and is ionically bonded. Sugar, on the other hand, is composed of carbon, oxygen, and hydrogen and has covalent bonds. A salt molecule is made up of one sodium atom and one chlorine atom.
How does sugar dissolve in water faster?
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come in contact with the sugar more often, causing it to dissolve faster.
Does sugar dissolve or melt?
Students consider that sugar melts when it dissolves in water. Often melting is considered to be substances turning into water. This is further reinforced because children see heat being involved in both processes – they know if you want to dissolve more sugar you warm up the water.
Which sugar dissolves fastest in water?
Not only did granulated sugar dissolve the fastest, it dissolved the fastest in cold water as well.
What is sugar dissolved in water called?
Answer: When Sugar is dissolved in water, Sugar is the solute, water is the solvent and sweet water is solution respectively.
Why do some salts not dissolve in water?
Insoluble salts are salt compounds that are insoluble in water at room temperature. These are insoluble in water because water molecules cannot attract the ions in the salt compound. Therefore, there are no intermolecular interactions between water molecules and insoluble salt compounds.
How much salt can you dissolve in water?
The saturation level is only nominally dependent on the temperature of the water. At 20 °C one liter of water can dissolve about 357 grams of salt, a concentration of 26.3% w/w. At boiling (100 °C) the amount that can be dissolved in one liter of water increases to about 391 grams, a concentration of 28.1% w/w.
Why does powder dissolve faster in hot water?
Hot water has more energy in it than cold water does. That means that the molecules in the water move faster. The molecules attack and break down the powder faster in hot water than cold.
Is salt heavier than water?
Salt water is more dense than fresh water That stuff would be the salt, which makes the mass of water greater, without adding much to its volume. Density = mass/volume. Increasing the mass by adding salt increases the density. Seawater is a little bit more dense than fresh water so it sinks beneath freshwater.
Is salt healthier than sugar?
A study, published by US researchers in online journal Open Heart suggests that sugar is in fact worse than salt for raising our blood pressure levels and heart disease risk.
Why is sugar more dense than water?
When you mix sugar with water, the sugar molecules occupy the space in between the water molecules, making the solution more tightly packed (denser).
Does dissolved sugar increase weight?
The solid sugar crystals break apart in water as the sugar dissolves, but the individual sugar particles or molecules are still present and do not change as a result of dissolving in the water. The combined mass of the sugar and water shouldn’t change.