Why can water exist in three states of matter
 Rachel Young
Rachel Young Water is unique because the properties of water allow it to exist in all three states of matter! Water is usually a liquid, but when it reaches to 32° Fahrenheit (F), it freezes into ice. (Ice is the solid state of water.) … When it begins to boil, some of the water turns into steam.
How can water exist in three states of matter?
Water can occur in three states: solid (ice), liquid, or gas (vapor). Solid water—ice is frozen water. When water freezes, its molecules move farther apart, making ice less dense than water. … When you boil water, the water changes from a liquid to a gas or water vapor.
Why does the three states of matter exist?
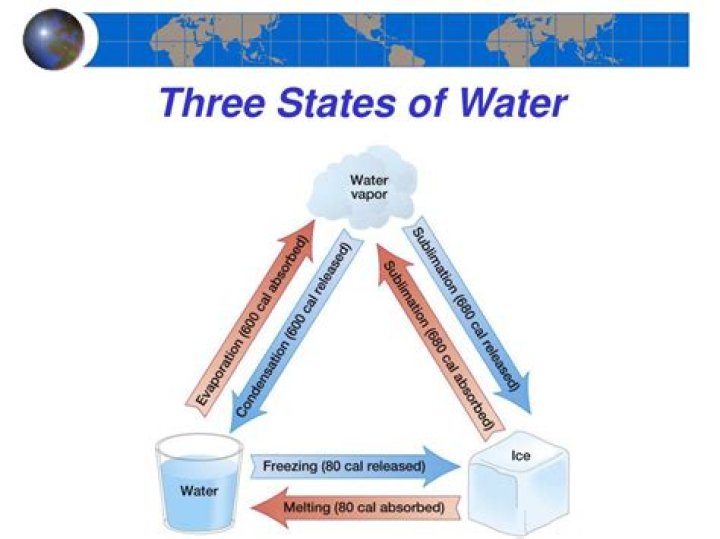
Basically the existence of different states of matter has to do with Inter-molecular forces, Temperature of its surroundings and itself and the Density of the substance. This image below shows you how the transition between each states occur (called Phase transitions).
Why does water exist as a solid liquid and gas?
Zero degrees celsius is defined by the triple point of water which is 273.16K at 611.2 Pa. … At a certain energy molecules will have enough energy to evaporate, even if the water temperature is 0 degrees C. Because of these two effects it is possible for the water to exist as solid, liquid and gas at the same time.Can water exist in all three forms at the same time?
At low pressure, steam, water and ice can all occupy the same container simultaneously. The combination of temperature and pressure is called the “triple point” of a substance. In this state, all three forms of water are in thermodynamic equilibrium — the amounts of each phase remain unchanged.
What three states can water exist in?
One of the most basic things we are taught in school science classes is that water can exist in three different states, either as solid ice, liquid water, or vapour gas.
Why is water still water when it changes states of matter?
That’s because state of matter is a physical property. As a result, when matter changes state, it doesn’t become a different kind of substance. For example, water is still water whether it exists as ice, liquid water, or water vapor.
Is water a matter why?
States of Matter Not only is water the most common substance on earth, but it is also the only substance that commonly appears as a solid, a liquid, and a gas within the normal range of earth’s temperatures. This makes water a good model for discussing the solid, liquid, and gas states of matter.Why is water a liquid state?
At room temperature (anywhere from zero degree centigrade to 100 degrees centigrade), water is found in a liquid state. This is because of the tiny, weak hydrogen bonds which, in their billions, hold water molecules together for small fractions of a second. Water molecules are constantly on the move.
What are the three states of water how can water be changed from one state to the other?If ice (solid) is heated, it changes to water (liquid). This change is called melting. Water (liquid) can change to water vapour (gas). This is called evaporation.
Article first time published onAre all liquids miscible with water?
The term is most often applied to liquids but also applies to solids and gases. For example, water and ethanol are miscible because they mix in all proportions. By contrast, substances are said to be immiscible if there are certain proportions in which the mixture does not form a solution.
Can all matter exists in 3 states?
Water is the only substance where all three states can be readily observed in everyday life: solid water as ice, liquid water in a water fountain, and gaseous water as steam. Carbon dioxide is a good example of a substance which can be a liquid, but only when it is under pressure.
Is water the liquid if not name 5 more?
They all have mass and take up space (volume). Water is not the only liquid. Others are milk, gasoline, alcohol, oil, Pepsi, tea, blood, spit, and many more.
How or when does water exist in three phases?
At the pressure and temperature of the triple point, all three phases (solid, liquid and gas) exist in equilibrium. The triple point for water occurs at a pressure of 4.6 torr and 0.01oC.
Is water a liquid or a solid?
Under standard atmospheric conditions, water exists as a liquid. But if we lower the temperature below 0 degrees Celsius, or 32 degrees Fahrenheit, water changes its phase into a solid called ice.
Does water attract water?
Just remember… Cohesion: Water is attracted to water, and Adhesion: Water is attracted to other substances.
How is water different from other liquids?
The key difference between water and liquid is that water refers to a chemical compound in its liquid stage whereas liquid is a physical state of matter. … The liquid is, in fact, a state of matter, the other two states being solid and gas.
What happens when water changes state?
When water changes state in the water cycle, the total number of water particles remains the same. … The water particles in each state behave as energy is absorbed or released. . Condensation, deposition, and freezing are processes that occur as a result of a decrease in the heat energy of water particles.
How can water be changed from one from to another?
Answer: Water changes form from liquid to gas by the process of evaporation. Water vapor can also change back to liquid by the process of condensation. Condensation and Evaporation are two processes that are extremely important for the water cycle to occur.
Is milk soluble in water?
Milk and water dissolve in each other and form a homogeneous substance. The liquids that do not mix into each other are known as immiscible liquids. … Miscible liquids form a homogeneous substance. Thus, milk and water are miscible liquids.
Is Sand soluble in water?
Salt is soluble in water whereas sand is insoluble (not dissolvable ) in water.
What liquids are insoluble in water?
A common example is oil and water. Oil contains molecules that are non-polar, thus they do not dissolve in water.
Is water the only substance that exists in 3 states naturally?
For many centuries, it was considered that there were only three states of matter: solid, liquid and gas (the three that are present and stable in our world). And water is the substance that best represents them, because it is the only one that exists naturally in the three states.
Can all matter be liquid?
There are three common states of matter on Earth; solids, liquids and gases. Far from it: many substances can be found in more than three states of matter, while others have fewer than three. All the chemical elements can be induced to form solids, liquids or gases.
Can all liquids become gases?
When you are a liquid and want to become a gas, you need to find a lot of energy. Once you can direct that energy into your molecules, they will start to vibrate. If they vibrate enough, they can escape the limitations of the liquid environment and become a gas.
What is the hardest state of matter?
As the temperature continues to drop, the matter forms a solid. Due to the solid’s low kinetic energy, particles have no “time” to move around, the particles have more “time” to be attracted. Therefore, solids have the strongest intramolecular forces (because they have the strongest attraction).
Why is water cohesive?
Cohesion refers to the attraction of molecules for other molecules of the same kind, and water molecules have strong cohesive forces thanks to their ability to form hydrogen bonds with one another. … Thus, the water molecules at the surface form stronger interactions with the neighbors they do have.
What are the 3 states of matter with examples?
- Solids – relatively rigid, definite volume and shape. In a solid, the atoms and molecules are attached to each other. …
- Liquids – definite volume but able to change shape by flowing. In a liquid, the atoms and molecules are loosely bonded. …
- Gases – no definite volume or shape.
What are the 3 processes where water enters the atmosphere?
Water enters the atmosphere through evaporation, transpiration, excretion and sublimation: Transpiration is the loss of water from plants (via their leaves).