Monsanto
.
Also to know is, who discovered acetic acid?
The first person to extract acetic acid from vinegar was the Muslim alchemist Jabir ibn Hayyan Geber (c. 721–815). The pure compound was not produced, however, for another ten centuries when the German chemist Georg Ernst Stahl (1660–1734) extracted acetic acid from vinegar in 1700 by distillation.
Also Know, how is acetic acid manufactured? Industrially, acetic acid is produced both synthetically and by bacterial fermentation. Approximately 75% of acetic acid used in the chemical industry is made by the carbonylation of methanol. Most acetic acid is made by methanol carbonylation, where methanol and carbon monoxide react to produce acetic acid.
Also to know, where does acetic acid come from?
Acetic acid is produced and excreted by acetic acid bacteria, notably the genus Acetobacter and Clostridium acetobutylicum. These bacteria are found universally in foodstuffs, water, and soil, and acetic acid is produced naturally as fruits and other foods spoil.
Is used for the production of acetic acid?
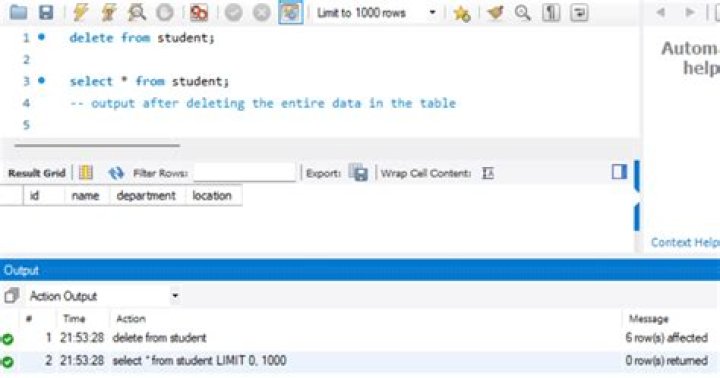
The most widely accepted route for the chemical synthesis is methanol carbonylation also called Monsanto process. Methanol in the presence of a rhodium-based carbonyl catalyst reacts with carbon monoxide producing acetic acid in the temperature and pressure ranges of 150–200 °C and 30–50 bar, respectively (12.
Related Question Answers
Is Vinegar an oxidizer?
Vinegar is an acid (in fact, it's known as acetic acid). The hypochlorous acid then reacts with the rest of the vinegar, Dr. Biberdorf continues. Because hypochlorous acid is something that's called an oxidizing agent, it takes electrons from whatever it comes into contact with.What is the Colour of acetic acid?
colourless
Does acetic acid dissolve plastic?
acetic acid) dissolve high-density polyethylene bottles? The other answers are fine. The exception is hydrofluoric acid that attacks glass; for it, we use plastic of some kind, probably a fluorinated plastic. However, HDPE is chemically resistant to most acids, except strong oxidizers like nitric acid.What is the common name for acetic acid?
methanecarboxylic acid
Is acetic acid miscible in water?
Acetic acid, as a small, polar molecule capable of hydrogen bonding with water is very soluble in water. You might reasonably expect it to be insolube in nonpolar solvents. However, a subtle phenomenon causes acetic acid to be soluble in nonpolar solvents as well.Is acetic acid poisonous?
Acetic Acid Hazards. Acetic acid can be a hazardous chemical if not used in a safe and appropriate manner. This liquid is highly corrosive to the skin and eyes and, because of this, must be handled with extreme care. Acetic acid can also be damaging to the internal organs if ingested or in the case of vapor inhalation.What foods contain acetic acid?
It has found use in a variety of products, including condiments such as ketchup, mustard, mayonnaise, and relish, salad dressings, marinades for meat, poultry, and fish, bakery products, soups, and cheeses.Is vinegar acetic acid?
Vinegar is an aqueous solution of acetic acid and trace chemicals that may include flavorings. Usually the acetic acid is produced by the fermentation of ethanol or sugars by acetic acid bacteria.What is the pH of blood?
The pH scale, ranges from 0 (strongly acidic) to 14 (strongly basic or alkaline). A pH of 7.0, in the middle of this scale, is neutral. Blood is normally slightly basic, with a normal pH range of about 7.35 to 7.45. Usually the body maintains the pH of blood close to 7.40.Is acetic acid strong or weak?
Acetic acid is a weak acid because it is not a strong acid which has a specific definition in chemistry: Strong acids completely dissociate in aqeous solution, that is, all their H+ come off in water. H+ is also called a proton because hydrogen without an electron is essentially a proton.What is the chemical formula of vinegar?
CH3COOH
Is acetic acid flammable?
Acetic acid is a colourless liquid; with a strong vinegar-like odour. It is flammable, and at temperatures warmer than 39°C, explosive vapour/air mixtures may be formed.How do you name acids?
In simple binary acids, one ion is attached to hydrogen. Names for such acids consist of the prefix “hydro-“, the first syllable of the anion, and the suffix “-ic”. Complex acid compounds have oxygen in them. For an acid with a polyatomic ion, the suffix “-ate” from the ion is replaced with “-ic.”How do you calculate acetic acid?
CH3COOH
Why is acetic acid good for cleaning?
Vinegar's acidity is what makes it such a good cleaner. Because vinegar is so acidic, it can counteract some icky buildups. It can dissolve away soap scum, brines left by hard water, and glue left behind by stickers. A paste of vinegar and baking soda can dissolve set red wine stains.Where is acetic acid found naturally?
Ingredient Results - Acetic Acid. A naturally occurring acid found in a variety of plants and fruits such as apples, grapes, oranges, pineapples, and strawberries. It is an organic acid that gives vinegar its sour taste and distinct smell. It Is produced through a fermentation process.What is pure acetic acid?
Acetic acid, also known as ethanoic acid, is an organic chemical compound best recognized for giving vinegar its sour taste and pungent smell. Pure water-free acetic acid (glacial acetic acid) is a colorless hygroscopic liquid and freezes below 16.7 °C (62 °F) to a colourless crystalline solid.How do you make acetic acid from ethanol?
Ethanol is oxidised to ethanoic acid by adding cold acidified potassium manganate (VII). Cold potassium manganate is a moderately strong oxidising agent, and it is reduced to manganese(IV) oxide, which then reacts with the sulphuric acid in solution to produce a brown solution.Is ammonia an acid or base?
Ammonia is a base because it is accepting hydrogen ions from the water. The ammonium ion is its conjugate acid - it can release that hydrogen ion again to reform the ammonia. The water is acting as an acid, and its conjugate base is the hydroxide ion. The hydroxide ion can accept a hydrogen ion to reform the water. 
 Emily Sparks
Emily Sparks