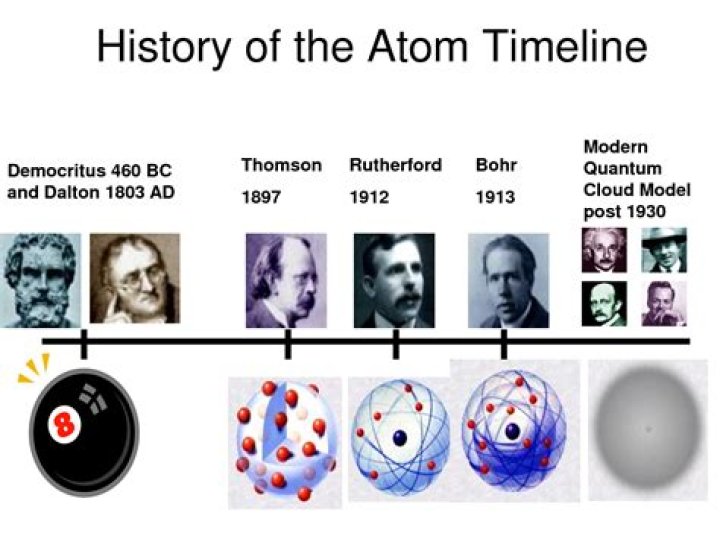
Who discovered the atom timeline
 Andrew Campbell
Andrew Campbell In the early 1800s, the English Chemist John Dalton performed a number of experiments that eventually led to the acceptance of the idea of atoms. He formulated the first atomic theory since the “death of chemistry” that occurred during the prior 2000 years. Dalton theorized that all matter is made of atoms.
Who discovered the atoms in order?
English chemist John Dalton subsequently made on the Greek notion of atoms in 1808. He postulated that matter is made of atoms, which are small indivisible particles. He also proposed that while all atoms of one element are identical, they are totally different from those that make up other elements.
When was the atom discovered and by who?
Lesson Summary. Around 450 B.C., the Greek philosopher Democritus introduced the idea of the atom. However, the idea was essentially forgotten for more than 2000 years. In 1800, John Dalton re-introduced the atom.
Who discovered atom first time?
Although the concept of the atom dates back to the ideas of Democritus, the English meteorologist and chemist John Dalton formulated the first modern description of it as the fundamental building block of chemical structures.Who are the 5 scientists who discovered atoms?
- Oct 21, 1803. JOHN DALTON by google images. …
- Apr 30, 1897. J.J THOMSON by google images. …
- Dec 14, 1900. MAX PLANCK by google images. …
- Apr 30, 1905. ALBERT EINSTEIN by google images. …
- Jul 10, 1913. NEILS BOHR by google images. …
- Jan 1, 1917. ERNEST RUTHERFORD by google images. …
- Jan 28, 1932. JAMES CHADWICK by google images. …
- Dec 2, 1942.
Who discovered the proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
Who named the atom?
But when it comes to the word atom, we have to go to ancient Greece of 400 B.C. And there was a brilliant philosopher named Democritus, and he proposed the Greek word atomos, which means uncuttable. And so as he explained, all matter was eventually reducible to discrete, small particles or atomos.
Who discovered atom Class 9?
The recorded history of the atom begins around 450 B.C. with a Greek philosopher know as Democritus.Who discovered molecules Einstein?
By 1908, Jean Perrin used Einstein’s paper to make the first estimate of the number of molecules in a mole of any substance. Within a decade, 6.02 x 1023 atoms per gram-mole was on its way to becoming one of the most widely known of the fundamental constants.
What is the history of an atom?Early History of the Atom Matter is composed of indivisible building blocks. This idea was recorded as early as the fifth century BCE by Leucippus and Democritus. The Greeks called these particles atomos, meaning indivisible, and the modern word “atom” is derived from this term.
Article first time published onWho discovered neutron?
By 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron.
What did Albert Einstein discover about the atom?
Einstein also in 1905 mathematically proved the existence of atoms, and thus helped revolutionize all the sciences through the use of statistics and probability. Atomic theory says that any liquid is made up of molecules (invisible in 1905). Furthermore, these molecules are always in random, ceaseless motion.
What did James Chadwick discover?
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge.
Who said atoms Cannot be created or destroyed?
Dalton based his theory on two laws: the law of conservation of mass and the law of constant composition. The law of conservation of mass says that matter is not created or destroyed in a closed system.
What did Democritus name the atom?
One of the first atomic theorists was Democritus, a Greek philosopher who lived in the fifth century BC. … He called these small pieces of matter “atomos,” the Greek word for indivisible. Democritus, theorized that atoms were specific to the material which they composed.
Who is electron father?
Sir Joseph John Thomson OM PRS (18 December 1856 – 30 August 1940) was a British physicist and Nobel Laureate in Physics, credited with the discovery of the electron, the first subatomic particle to be discovered.
Who discovered electron and neutron?
02 Discovery of Electron, Proton and Neutron. Electrons were discovered by J.J. Thomson in 1897.
Who discovered nucleus of atom?
May, 1911: Rutherford and the Discovery of the Atomic Nucleus. In 1909, Ernest Rutherford’s student reported some unexpected results from an experiment Rutherford had assigned him. Rutherford called this news the most incredible event of his life.
What was Einstein's IQ?
The maximum IQ score assigned by the WAIS-IV, a commonly-used test today, is 160. A score of 135 or above puts a person in the 99th percentile of the population. News articles often put Einstein’s IQ at 160, though it’s unclear what that estimate is based upon.
Who discovered protons Ncert?
The discovery of the proton is credited to Ernest Rutherford, who proved that the nucleus of the hydrogen atom (i.e. a proton) is present in the nuclei of all other atoms in the year 1917. Based on the conclusions drawn from the gold-foil experiment, Rutherford is also credited with the discovery of the atomic nucleus.
Who discovered electron and how class 9?
Question 6 Explain the experimental set for discovery of electron? The electrons were discovered by J.J. THOMSON in 1897. 1) It consist of a long cylindrical tube sealed at both ends and is fitted with two metal electrodes.
Where do atoms come from originally?
Atoms were created after the Big Bang 13.7 billion years ago. As the hot, dense new universe cooled, conditions became suitable for quarks and electrons to form. Quarks came together to form protons and neutrons, and these particles combined into nuclei.
What was the first atom?
It took 380,000 years for electrons to be trapped in orbits around nuclei, forming the first atoms. These were mainly helium and hydrogen, which are still by far the most abundant elements in the universe.
Who invented proton Goldstein or Rutherford?
The quark content of a proton. The color assignment of individual quarks is arbitrary, but all three colors must be present. Forces between quarks are mediated by gluons.ClassificationBaryonDiscoveredObserved as H+ by Eugen Goldstein (1886). Identified in other nuclei (and named) by Ernest Rutherford (1917–1920).
What did Thomson discover?
Thomson, in full Sir Joseph John Thomson, (born December 18, 1856, Cheetham Hill, near Manchester, England—died August 30, 1940, Cambridge, Cambridgeshire), English physicist who helped revolutionize the knowledge of atomic structure by his discovery of the electron (1897).
Who discovered the photon?
December 18, 1926: Gilbert Lewis coins “photon” in letter to Nature. At the dawn of the 20th century, Max Planck and Albert Einstein turned physics on its ear by introducing the notion of quanta.
What was Heisenberg's experiment?
Heisenberg conducted a thought experiment as well. He considered trying to measure the position of an electron with a gamma ray microscope. The high-energy photon used to illuminate the electron would give it a kick, changing its momentum in an uncertain way.
What did Werner Heisenberg discover?
Werner Heisenberg discovered the uncertainty principle, which states that the position and the momentum of an object cannot both be known exactly.
What was James Chadwick theory?
James Chadwick played a vital role in the atomic theory, as he discovered the Neutron in atoms. Neutrons are located in the center of an atom, in the nucleus along with the protons. They have neither a positive nor negative charge, but contribute the the atomic weight with the same effect as a proton.
What did James Chadwick think the atom looked like?
Until 1932, the atom was believed to be composed of a positively charged nucleus surrounded by negatively charged electrons. … Chadwick interpreted this radiation as being composed of particles with a neutral electrical charge and the approximate mass of a proton. This particle became known as the neutron.
What did Niels Bohr discover?
What was Niels Bohr’s most important discovery? Niels Bohr proposed a model of the atom in which the electron was able to occupy only certain orbits around the nucleus. This atomic model was the first to use quantum theory, in that the electrons were limited to specific orbits around the nucleus.