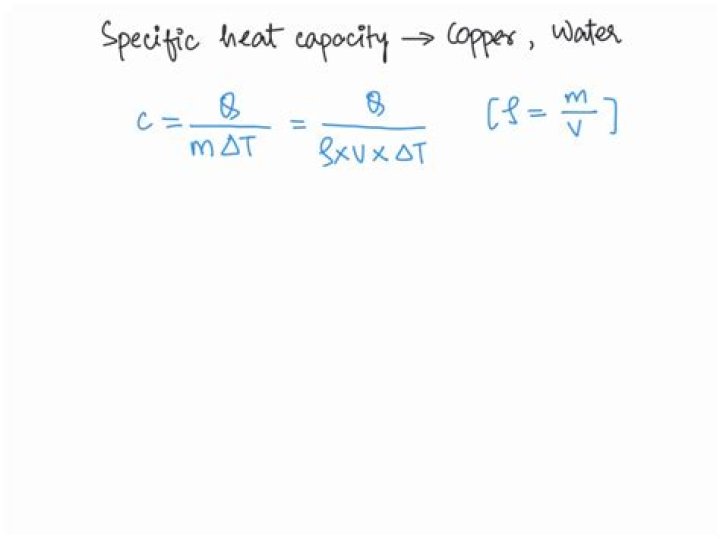
Which substance has a greater specific heat capacity copper or water
 Christopher Martinez
Christopher Martinez There is a greater increase in temperature in copper when water is hot because of the lower specific heat capacity.
What has a greater heat capacity water or copper?
Water has a higher specific heat capacity because it has less of a tendency to change in temperature. … Copper with the temperature of 60 degrees Celsius, and the same mass as that of the water in which it was placed, cooled off to 23.4 degrees Celsius.
What has a higher specific heat than water?
Originally Answered: Which substance has a longer heat capacity than water? Among solids, liquids and gases , hydrogen has the highest specific heat (heat capacity).
Which substance has a greater specific heat capacity?
Water has the highest specific heat capacity of any liquid. Specific heat is defined as the amount of heat one gram of a substance must absorb or lose to change its temperature by one degree Celsius.Does copper have a high specific heat capacity?
A substance with a high specific heat capacity can absorb a large quantity of heat before it will raise in temperature (water has a high specific heat). A substance with a low specific heat requires relatively little heat to raise its temperature (copper has a low specific heat).
Which material has more specific heat?
Liquid water has one of the highest specific heat capacities among common substances, about 4184 J⋅kg−1⋅K−1 at 20 °C; but that of ice, just below 0 °C, is only 2093 J⋅kg−1⋅K−1. The specific heat capacities of iron, granite, and hydrogen gas are about 449 J⋅kg−1⋅K−1, 790 J⋅kg−1⋅K−1, and 14300 J⋅kg−1⋅K−1, respectively.
Why does copper have a low specific heat capacity?
– they don’t. Metal atoms in pure metal are very close together and are able to transfer heat easily via conduction from one atom exciting the other atoms next to it. … So the amount of energy it takes to heat a metal is relatively small to that of water for example.
What has highest heat capacity?
Mercury =0.033 g−1 ∘C−1. Name a liquid which has the highest specific heat capacity.Does water have a high heat capacity?
Water has a high specific heat capacity—it absorbs a lot of heat before it begins to get hot.
Which metal has highest specific heat capacity?Specific Heat Capacity of Metals Table ChartMetalBtu/(lb-°F)J/(g-°C)Aluminum0.2200.921096Antimony0.0500.20934Barium0.0480.2009664
Article first time published onWhich substance has the lowest specific heat capacity?
Mercury has a low specific heat capacity . Therefore it does not take much heat from the body whose temperature is to be measured. Otherwise it will lower the temperature of the body.
Which liquid has highest specific heat capacity?
Water has the highest specific heat capacity.
Why is specific heat of water higher than metals?
Water atoms are attracted to each other due to the attraction between the more negative oxygen atoms and the more positive hydrogen atoms due to the distribution of electrons in the molecule (called H-bonding). This H-bonding is why water has a surprisingly high specifics heat capacity.
Why does water have a high specific heat than metal?
This happens because the specific heat capacity of the metal spoon is much lower than that of the water in the soup. Water has the highest specific heat capacity of any liquid.
What is the specific heat capacity of water?
Figure 1. Water has a specific heat capacity of 4.186 J/g°C, meaning that it requires 4.186 J of energy (1 calorie) to heat a gram by one degree. is the change in temperature of the system.
Does water have a low specific heat capacity?
Temperature and Water Water has a high specific heat capacity—it absorbs a lot of heat before it begins to get hot.
Do metals have a higher heat capacity?
Specific heat is a measure of how much energy it takes to raise the temperature of a substance. It is the amount of heat energy necessary to raise one mass unit of that substance by one temperature unit. … Metals tend to have low specific heat. Water has very high specific heat.
Why does copper have a higher heat capacity than lead?
You have to consider that copper has a lower standard atomic weight thus for 1kg of copper the amount of substance is more than the for 1kg of lead (i.e. 3 times more atoms in 1kg of copper than 1kg of lead).
Why do liquids have the highest heat capacity?
liquid has a higher heat capacity than solid or gas and this is because liquid can absorb the most heat (translational and vibrational) but solids absorb only vibrational and gases absorb only translational.
Why does water have a high specific heat quizlet?
Water’s high specific heat can be traced to hydrogen bonding: … It needs energy to break the hydrogen bond. – Heat is released when hydrogen bonds form. As water cools, hydrogen bonds form and water vapor releases stored energy.
What is high specific heat?
A high specific heat capacity means it can hold a large amount of thermal energy in for a low mass or temperature change. It is also good at keeping in thermal energy, for example: Object: Mass = 3kg.
Which has higher specific heat capacity water or sand?
That’s because sand has a lower specific heat capacity—it takes less energy to raise the temperature by one degree. … The sun puts out a more or less constant rate of energy, which heats up sand more quickly and water more slowly. Sand has a much lower specific heat than water—that’s why it gets hot so fast!
Which has a higher heat capacity water or oil?
Water has higher specific heat since it has the ability to store large amount of thermal energy than oil. The specific heats of these compounds is a function of their molecular weight, density and viscosity.
What is specific heat capacity of copper?
Consider the specific heat of copper , 0.385 J/g 0C. What this means is that it takes 0.385 Joules of heat to raise 1 gram of copper 1 degree celcius.
What is the specific heat of copper?
Substancespecific heat capacity Cp,s (J/g °C)molar heat capacity Cp,m (J/mol °C)copper0.38524.47granite0.790—graphite0.7108.53helium5.193220.786
Why is the specific heat of metals lower than water?
Why do metals have low specific heat compared to water? – Quora. Metal atoms in pure metal are very close together and are able to transfer heat easily via conduction from one atom exciting the other atoms next to it. So the amount of energy it takes to heat a metal is relatively small to that of water.
Does water or alcohol have a higher heat capacity?
Specific heat capacity, or simply specific heat, is the amount of heat required to change the temperature of a substance. As water requires more time to boil than does alcohol, you might conclude that water requires more heat than alcohol to raise its temperature.
Which has the highest specific heat liquid solid and gas?
Water has the highest specific heat capacity of any liquid. “
Does water have a high heat of vaporization?
That is, water has a high heat of vaporization, the amount of energy needed to change one gram of a liquid substance to a gas at constant temperature. Water’s heat of vaporization is around 540 cal/g at 100 °C, water’s boiling point.
How does the heat capacity of water compare to the heat capacity of rock?
Water has a heat capacity that is 4-5 times higher than rock or dirt. Since earth’s surface is 3/4 water and 1/4 dirt and rock, the water covered parts heat more slowly in the sun.
Is specific heat the same as heat capacity?
Heat capacity is the amount of heat required to raise the temperature of an object by 1oC. The specific heat of a substance is the amount of energy required to raise the temperature of 1 gram of the substance by 1oC.