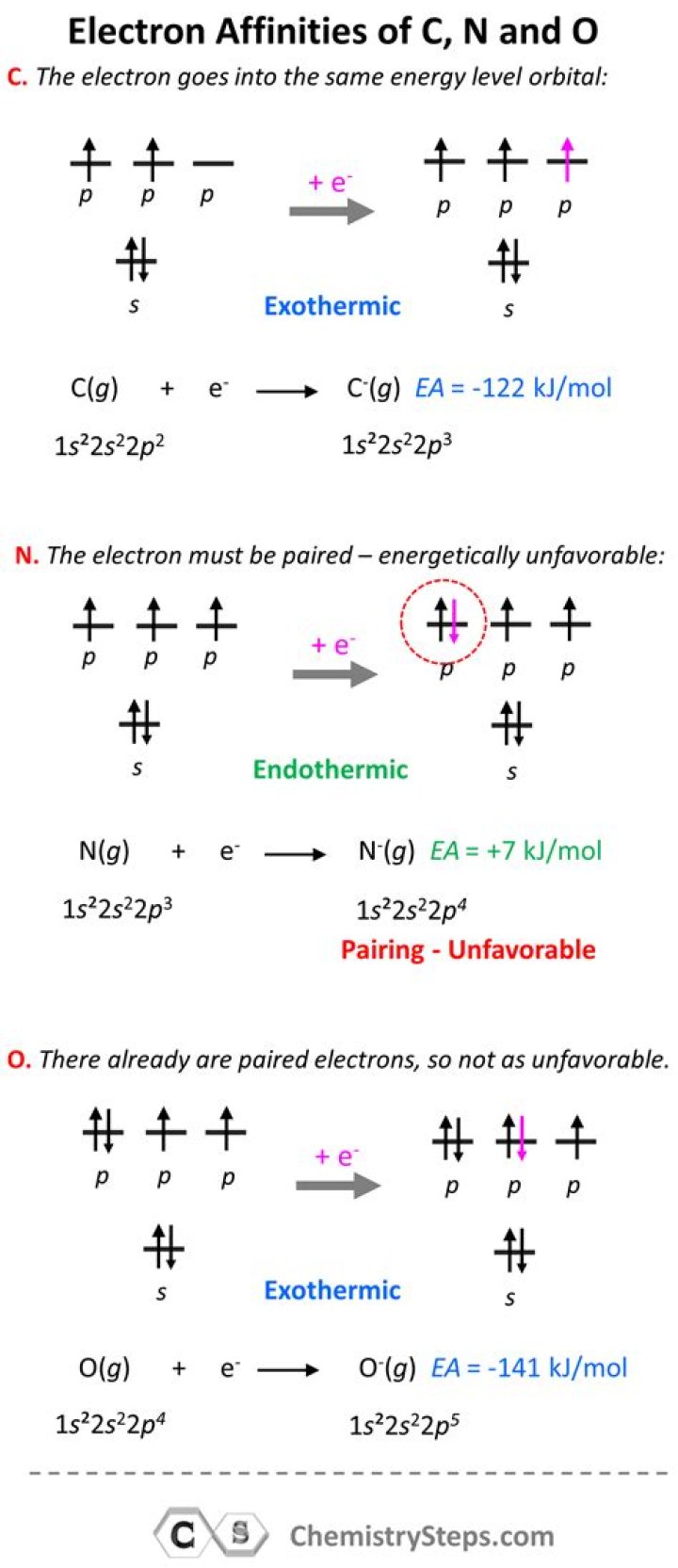
Which has more electron affinity carbon or nitrogen
 Andrew Campbell
Andrew Campbell Since a half-filled “p” subshell is more stable, carbon has a greater affinity for an electron than nitrogen.
Why is the electron affinity of nitrogen less than carbon?
Nitrogen has a half-filled p orbital. The number of electrons in the p orbital in nitrogen is three. Nitrogen, therefore, does not accept more electrons as it has a stable configuration. … Therefore, nitrogen has a lower electron affinity than carbon and oxygen.
Which has more electron affinity N or O?
Answer: Oxygen has more electron affinity because Nitrogen gains more stability by attaining partial configuration.
Does carbon have a high affinity for electrons?
Carbon has a higher electron affinity than Oxygen because it is easier to place an electron in the ground state C atom than a ground state O atom.What is the electron affinity of nitrogen?
Z7ElementNNameNitrogenElectron affinity (eV)-0.07Electron affinity (kJ/mol)-6.8
Which has more electron affinity?
Electron affinity follows the trend of electronegativity: fluorine (F) has a higher electron affinity than oxygen (O), and so on.
Which has maximum electron affinity?
Noble gases have maximum electron affinity.
Why is electron gain enthalpy of carbon more than nitrogen?
Carbon has lowest electron gain enthalpy. than nitrogen , oxygen , sulpher. because carbon has teo electron in p orbital if it gain one electron then it will become partially stable than other. Electron affinity of nitrogen is lower(less negative ) than carbon.Why does nitrogen have a high electron affinity?
Nitrogen has a half-filled 2p subshell, so that there is one electron in each orbital. This creates an unusually stable atom because of half-shell stability.
Is carbon electron affinity low?Not all elements have a negative electron affinity. They do not always go through the exothermic process. Some stable elements also have positive electron affinity. Carbon has a negative electron affinity and Nitrogen has a positive electron affinity.
Article first time published onWhat has the lowest electron affinity?
What is the lowest electron affinity? Metals are more likely to lose electrons than they are to gain them. Of the metals, mercury has the lowest electron affinity.
Which has more electron affinity carbon or silicon?
Electron affinity decreases down the group because the size of atom increases and the added electron releases less amount of energy. … But, out of carbon and silicon, the electron affinity of silicon is greater than carbon because silicon has empty 3d orbital.
Which has more electron affinity nitrogen or phosphorus?
Nitrogen has greater electron affinity than phosphorus .
What is the maximum electron affinity of nitrogen?
ElementNitrogenElectron Configuration[He] 2s2 2p3Possible Oxidation States+1,2,3,4,5/-1,2,3Electron Affinity [kJ/mol]7Electronegativity [Pauling scale]3.04
Why is nitrogen smaller than carbon?
Electronic orbitals take up space. The atomic size decreases when we move right in a period Due to increase in effective nuclear charge. Hence, Nitrogen is smaller than carbon.
What is electron affinity of carbon?
Electron Affinity of Carbon is 153.9 kJ/mol.
Which has higher electron affinity fluorine or neon?
In the modern periodic table, electron affinity increases on moving left to right in a period while it decreases on moving down the group. … Thus, noble gases have the least electron affinity in a period. Hence, we can conclude that in a period, fluorine (halogen) has higher electron affinity than neon (noble gas).
Is carbon more negative than nitrogen?
As along the period, from left to right, the no. of electrons increase in shells, the power/attraction of the nucleus also increases, making each element more electronegative than the previous one. … From all this, we conclude that Nitrogen is more electronegative than carbon.
Why Carbon has more electron gain enthalpy than silicon?
Greater the size of atom, less will be the effective nuclear charge and so less readily it will accept an electron to form ion. … C is smaller in size than Si, therefore C has higher effective nuclear charge and so higher electron gain enthalpy.
Does nitrogen has positive electron gain enthalpy?
Nitrogen has positive electron gain enthalpy whereas oxygen has negative. However, oxygen has lower ionization enthalpy than nitrogen.
Why does nitrogen not have a measurable electron affinity?
Nitrogen has a zero value of electron affinity because of the stability of the half filled 2psubshell (that is, N has little tendency to accept another electron)
Which of the following has electron affinity less than zero?
ElectronegativityElectron affinityIt is the property of a bonded atomIt’s the property of an isolated atom
Does francium have the highest electron affinity?
Electron Affinity: Electron affinity is defined as the energy change that accompanies the addition of an electron to an atom. Some atoms readily attract electrons, and the electron affinity has a negative value, meaning that energy is released. … Chlorine has the highest affinity for electrons and francium is the lowest.
Is nitrogen more electronegative than carbon?
Electronegativity varies predictably across the periodic table. Electronegativity increases from bottom to top in groups, and increases from left to right across periods. … Carbon has an electronegativity of 2.55 while Nitrogen has an electronegativity of 3.04.
Why electron affinity of nitrogen is less than phosphorus?
There is less repulsion for the added electron and the process requires less energy. … So, the reason that the change in energy for both electron affinities is lower for atoms of phosphorus than for atoms of nitrogen is answer choice (B).
Why does fluorine have the highest electron affinity?
The incoming electron is going to be closer to the nucleus in fluorine than in any other of these elements, so you would expect a high value of electron affinity.





