What temperature does water evaporate in a vacuum
 Emily Sparks
Emily Sparks While water boils at 100°C at atmospheric pressure, its boiling point under vacuum of below 1 mm goes down to 15°C.
Does water evaporate in vacuum?
Water boils and evaporates irrespective of the presence of air and irrespective of its humidity. The evaporation increases the gasseous volume and the vacuum pump removes this extra vapour while fighting to keep the pressure at 0.1 Atmospheres.
What happens if water is kept in vacuum?
Originally Answered: What happen with water in vacuum? It will boil, freeze, and sublime. Once the air pressure is reduced low enough, water will boil at room temperature. That process causes it to become colder, until it literally boils and freezes simultaneously (this is known as the triple point).
How long does it take to evaporate water under vacuum?
Key Results: We found that it took ~150 minutes to evaporate 25 ml of ~20°C water with a pressure of ≥2.86 Torr during the experiment. When 25 grams of 100-170 μm glass beads were added to 25 ml of water, we found it took ~300- 360 minutes for the water to evaporate.Does temperature drop in a vacuum?
Vacuum flasks work because they reduce the transfer of heat by conduction and convection. … The NBMR that permeates the universe could be said to raise the temp above absolute zero other than that the perfect vacuum has no Kinetic energy and therefore zero temperature.
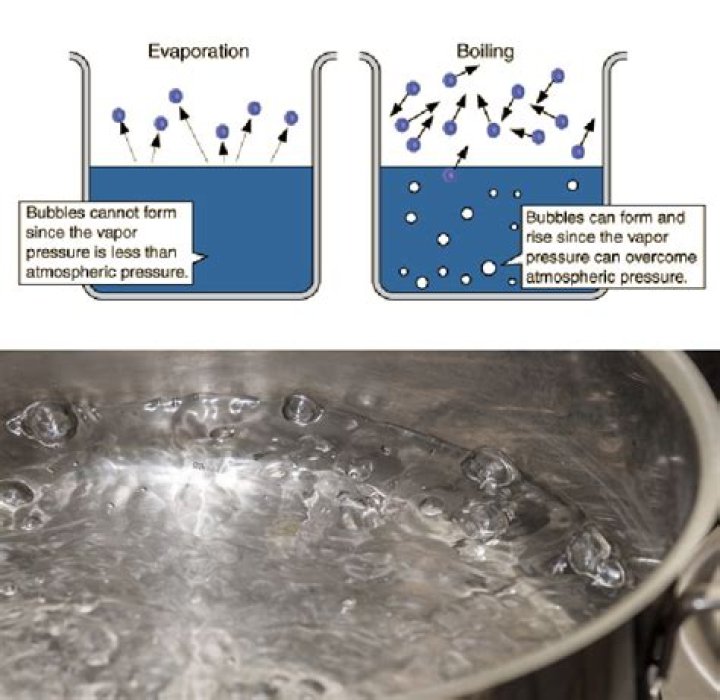
Can water boil in a vacuum?
In a vacuum, there is no pressure. This is critical for most liquids to remain in a liquid state because with no pressure, the temperature at which they start to boil drops. … This means that in the vacuum of space, a cup of water would boil into a vapor before it can freeze.
At what temperature does water freeze in a vacuum?
Water freezes at its freezing point. In a vacuum in contrast to standard temp and pressure it will freeze at about 0.01C.
Can liquid water exist in a vacuum?
Liquids will boil whenever the surrounding pressure is less than the vapor pressure of that material. All materials have a vapor pressure, and in a perfect vacuum, the surrounding pressure would be zero. This means that no liquids could remain a liquid in a perfect vacuum.Does water evaporate in 100 humidity?
Relative humidity greatly affects evaporation rates. When it is high, relative humidity slows evaporation; relative humidity reduces it to zero (no evaporation at all) when it reaches 100 percent. Relative humidity is the amount of moisture in the air compared to what the air can “hold” at that temperature.
Why does water evaporate in a vacuum?Vacuum evaporation is the process of causing the pressure in a liquid-filled container to be reduced below the vapor pressure of the liquid, causing the liquid to evaporate at a lower temperature than normal.
Article first time published onDoes blood boil in a vacuum?
Instead, you would face another gruesome fate first: your blood, your bile, your eyeballs –will boil furiously, since the low pressure of the vacuum massively reduces the boiling point of water. It is only then that you would freeze.
How does vacuum evaporation work?
Vacuum evaporation is a technique which is characterized by transforming liquid effluent into two flows, one of high quality water and the other comprising a concentrated waste. The water obtained is of sufficiently high quality to be re-used, whereas the waste can be concentrated, even reaching almost total dryness.
Does water expand in a vacuum?
If your vacuum chamber is thermally isolated, then the temperature of the water decreases because it is expanding adiabatically into the gas state. … This tells us that the water cools down as it expands.
What is the maximum temperature water can reach?
Liquid water can be hotter than 100 °C (212 °F) and colder than 0 °C (32 °F). Heating water above its boiling point without boiling is called superheating. If water is superheated, it can exceed its boiling point without boiling.
Does water freeze or boil in a vacuum?
Water immediately boils in space or any vacuum. Space does not have a temperature because temperature is a measure of molecule movement. … After water vaporizes in a vacuum, the vapor could condense into ice or it could remain a gas.
Does ice boil in a vacuum?
Yes, but also no. Water ice does not melt into liquid in a vacuum. It sublimates—goes directly from solid to gas. In fact, liquid water in a vacuum will simultaneously freeze and boil; you can turn water into ice at room temperature just by exposing it to hard vacuum.
Does moisture freeze in a vacuum?
Neil’s video proved that pulling a deep vacuum quickly can result in freezing with even a small amount of liquid water present.
Does water heat faster in a vacuum?
Water Boiling in a Vacuum at Room Temperature This is the same reason it is easier to boil water in the mountains than it is at sea level: you need to add less energy to get the water boiling when there is less air pressure on it.
Does mercury boil in a vacuum?
Yes. Mercury is largely boiled inside fluorescent light tubes, where there is a pretty good vacuum (when new).
Does water evaporate at 50 degrees?
So, water in sub-cooled state at 50 degree C will continue to evaporate until the vapour pressure is less than saturation pressure or say RH is not 100 % but it will not boil at 50 degree C at 1 atm pressure.
What temperature does water evaporate outside?
At 100 degreeC, water converts to steam (at atmospheric pressure) and temperature remains steady until all the water is converted to steam. Originally Answered: At what temperature does water evaporate? It will evaporate at any temperature from 0 to 100 centigrade.
What rate does water evaporate?
Scientists there found that the rate of evaporation can be below 76 centimeters (30 inches) per year at the low end, to 305 centimeters (120 inches) per year on the high end.
How cold is it in space?
Hot things move quickly, cold things very slowly. If atoms come to a complete stop, they are at absolute zero. Space is just above that, at an average temperature of 2.7 Kelvin (about minus 455 degrees Fahrenheit).
Would your blood boil in space?
In space, there is no pressure. So the boiling point could easily drop to your body temperature. That means your saliva would boil off your tongue and the liquids in your blood would start to boil. All that bubbly boiling blood could block blood flow to vital organs.
What temperature does water boil at in space?
But astronauts on the International Space Station (ISS) have shown that something else entirely is happening in a microgravity environment. They found that vapour condensed into a liquid, even when the temperature was 160 Kelvin above the substance’s normal boiling point – a new phenomenon that’s stumped scientists.
What happens if you bleed in space?
In space, blood can splatter even more than it usually does on Earth, unconstrained by gravity. Or it can pool into a kind of dome around a wound or incision, making it hard to see the actual trauma. (Fun fact: If you are bleeding more than 100 milliliters per minute, you are probably doomed.
Has anyone ever tried to breathe in space?
No, there is no way to stand out in space and try to breath! Why? because the water and other liquids in your body are going to evaporate instantly (and you’re gonna die, off course). As we know, the water evaporating temperature is a function of the pressure.
Would your skin freeze in space?
Acute exposure to the vacuum of space: No, you won’t freeze (or explode) … The absence of normal atmospheric pressure (the air pressure found at Earth’s surface) is probably of greater concern than temperature to an individual exposed to the vacuum of space [1].
Are vacuum chambers cold?
As pointed out by the comments, vacuum itself does not have temperature, since temperature is defined in terms of the kinetic energy of the particles. However, if you put an object (let’s say an idealized blackbody) inside the vacuum, its temperature is not simply zero.