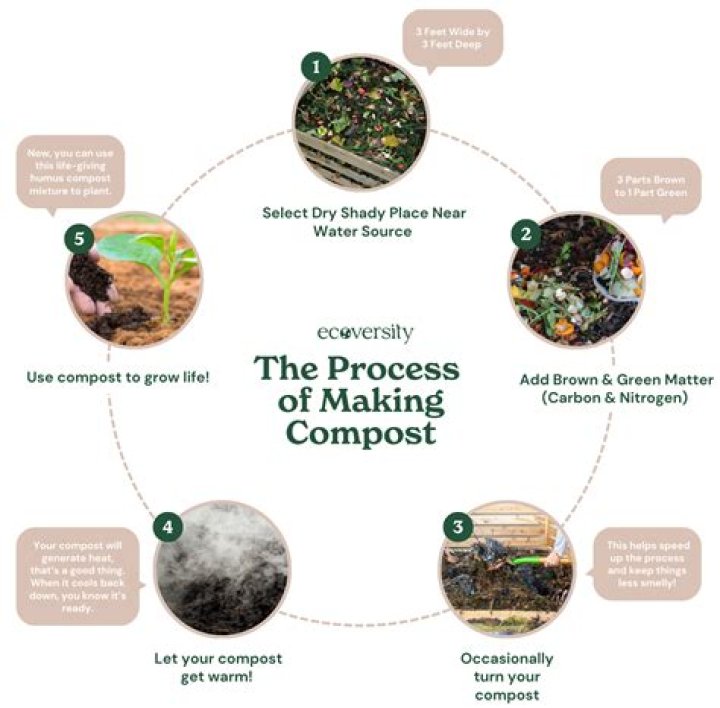
What makes an acid Pogil
 Isabella Bartlett
Isabella Bartlett Acids are covalently bonded molecules, but when they are put into water they produce ions. … Because of their special classification, acids have a naming system different from ionic or other molecular (covalent) compounds.
What makes an acid unique and how are acids named Pogil?
Acids are covalently bonded molecules, but when they are put into water they produce ions. … Because of their special classification, acids have a naming system different from ionic or other molecular (covalent) compounds.
What is a ternary acid?
Ternary Acids (Oxy-acids) Ternary acids are also known as “Oxy-acids. Contain hydrogen and oxygen and one other (usually) nonmetallic element. These acids contain both hydrogen and oxygen but their names make no reference to either hydrogen or oxygen.
How do you tell if an acid is binary or ternary?
A binary acid is an acidic compound that always has hydrogen boned to another chemical elements, most of the times a nonmetal. Ternary acids are acidic compounds that contain hydrogen and oxygen combined with another element. Binary acid has one two types of chemical elements (hydrogen bonded to a nonmetal).Is Hydrosulfuric acid a binary acid?
Binary acids like HCl and H₂S have two names: The “hydrogen name” is “hydrogen” + element stem + “ide”. For the pure compounds, this gives names like hydrogen chloride and hydrogen sulfide. … This gives names like hydrochloric acid and hydrosulfuric acid.
What are binary acids composed of?
A binary acid consists of hydrogen and one other element. Oxyacids contain hydrogen, oxygen, and one other element. The name of the acid is based on the anion attached to the hydrogen.
What is the formula for Hydrosulfuric acid?
Hydrosulfuric AcidNameHydrosulfuric AcidAlso Known asHydrogen Sulfide and SulfaneAppearanceColourless GasChemical FormulaH2S
Which substance is a binary acid?
Binary acids are certain molecular compounds in which hydrogen is combined with a second nonmetallic element; these acids include HF, HCl, HBr, and HI. HCl, HBr, and HI are all strong acids, whereas HF is a weak acid.Is carbonic acid binary or ternary?
FormulaNameHIO3iodic acidHNO3nitric acidH2CO3carbonic acidH2SO4sulfuric acid
How are ternary acids formed?Ternary compounds are those compounds containing three elements. Ionic ternary compounds are formed by the combination of a monatomic cation with a polyatomic (containing several atoms) anion, as in sodium nitrate, NaNO3. A polyatomic anion is derived from a ternary acid.
Article first time published onWhich substance is a ternary compound?
In inorganic chemistry, a ternary compound is a compound containing three different elements. An example is sodium phosphate, Na3PO4. The sodium ion has a charge of 1+ and the phosphate ion has a charge of 3-. Therefore, three sodium ions are needed to balance the charge of one phosphate ion.
What polyatomic ion is mno4?
Symbols and Names of Some Common Polyatomic Ions and One MoleculeNH4+ammonium ionOH-PO33-phosphite ionMnO4-Formulas and Names for Some Common Acids (all names should have acid added)H2SO4sulfuricH3PO4
What are the acid naming rules?
Acids are named based on their anion — the ion attached to the hydrogen. In simple binary acids, one ion is attached to hydrogen. Names for such acids consist of the prefix “hydro-“, the first syllable of the anion, and the suffix “-ic”. Complex acid compounds have oxygen in them.
Is water a Hydracid?
Second word is the root of the nonmetal with a suffix “-ide”. These compounds are called hydracids because dissolve in water are acid dissolutions.
Is hydroiodic acid a binary acid?
Hydroiodic acid, hydrofluoric acid, and hydrobromic acid are binary acids because they have a hydrogen element and nonmetal element (iodine, fluorine and bromine) bonded together. In contrast to metals, which are shiny and ductile, nonmetals are dull and brittle.
What is the formula for Hydronitric acid?
Hydronitric acid has the formula HN3 (aq). The (aq) means the compound is dissolved in solution.
What type of acid is Hydrosulfuric acid?
Explanation: Hydrosulphuric acid is nothing but it is Hydrogen Sulphide. That is H2S.
Which of the following is also known as Hydrosulfuric acid?
Hydrogen sulfide is a flammable, colorless gas with a characteristic odor of rotten eggs. It is commonly known as hydrosulfuric acid, sewer gas, and stink damp. People can smell it at low levels.
What is the molar mass for Hydrosulfuric acid?
Answer. Hence, the molar mass of H2SO4is98. 08 g/mol.
When binary acids are created they are created with?
The simplest definition of an acid is a substance that produces H+ ions in a solution. A binary acid is made up of 2 types of atoms, a hydrogen atom and a non-metal atom.
What's an example of a binary compound?
Once again, a binary compound is a substance composed of exactly two different elements, which are substances that cannot be simplified further by chemical means. Examples of binary compounds include H2O, H2S, and NH3. Examples of substances that are not chemical compounds include Au, Fe, O, HCN, and HNO3.
Is CH4 a binary acid?
Binary Ionic Compounds Containing a Metal and a Nonmetal. A binary compound is a compound formed from two different elements. There may or may not be more than one of each element. … CH4 Binary, since there are only two types of atoms.
What elements make acids?
Acids are molecular compounds that release hydrogen ions. A binary acid consists of hydrogen and one other element. Oxoacids contain hydrogen, oxygen, and one other element. The name of the acid is based on the anion attached to the hydrogen.
Is hydrocyanic acid a binary acid?
All acids beginning with the prefix „hydro“ are otherwise known as binary acids. HCl, which contains the anion chloride, is called hydrochloric acid. HCN, which contains the anion cyanide, is called hydrocyanic acid.
What is the name of the ternary acid H2SO3?
Ternary acids that contain polyatomic ions whose names end with “ite” are named in a similar fashion to those whose names end with “ate.” You add “ous” to the end instead of “ic.” For example, H2SO4 is called “sulfuric acid.” But H2SO3 is called “sulfurous acid.”
Which substance is a binary acid quizlet?
Which substance is a binary acid? A. Hydrochloric acid.
What is the name for s03?
Sulfur trioxide (SO3) is generally a colorless liquid.
Is HNO2 a binary acid or an Oxyacid?
Binary acids are composed of hydrogen and a nonmetal. For example, hydrochloric acid, HCl contains hydrogen and a chlorine atom. add the suffix -ic (e.g. HCl – hydrochloric). … If the oxyanion ends in -ite, write the base name of the oxyanion and add the suffix -ous (e.g. HNO2 – nitrous).
What acid makes nitrate?
nitric acid produces nitrate salts.
How many elements are in a ternary acid?
Ternary acids also consist of three elements. A ternary acid is an acid that has the elements hydrogen and oxygen along with another element, often a nonmetal. Or, they contain hydrogen in addition to a polyatomic ion.
How do you make a ternary compound?
- Ternary compounds are composed of three or more elements.
- Ternary compounds are named by stating the cation first, followed by the anion.
- Positive and negative charges must balance.