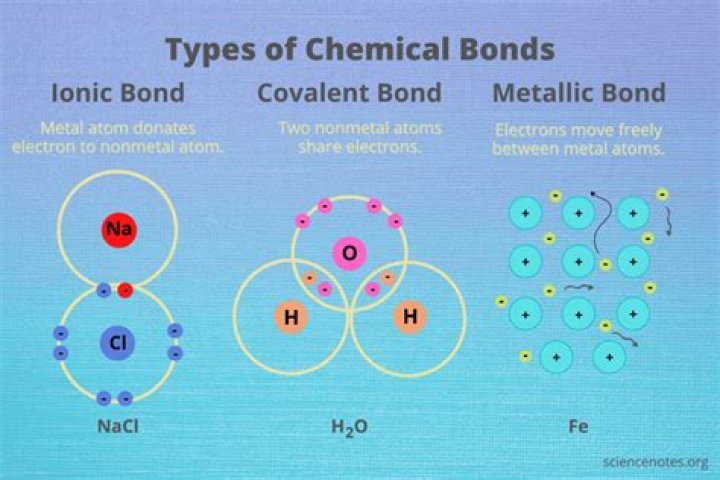
What kinds of electrons form the covalent bonds in molecules
 Isabella Bartlett
Isabella Bartlett Covalent bonds are a class of chemical bonds where valence electrons are shared between two atoms, typically two nonmetals. The formation of a covalent bond allows the nonmetals to obey the octet rule and thus become more stable. For example: A fluorine atom has seven valence electrons.
What kinds of atoms will form covalent bonds?
Covalent bonds usually occur between nonmetals. For example, in water (H2O) each hydrogen (H) and oxygen (O) share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom. In general, ionic bonds occur between elements that are far apart on the periodic table.
What atoms can form 4 covalent bonds?
Carbon can form four covalent bonds to create an organic molecule.
What are molecular covalent bonds made of?
A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei.How do you find bonding electrons?
- 6 (S)+2⋅6 (2O)=18.
- 8 (S)+2⋅8 (2O)=24.
- 24−18=6, i.e. three bonding electron pairs.
- 18−6=12 i.e. six lone pairs.
Which electrons are involved in bonding?
The electrons on the outermost energy level of the atom are called valence electrons. The valence electrons are involved in bonding one atom to another. The attraction of each atom’s nucleus for the valence electrons of the other atom pulls the atoms together.
How do you form a covalent bond?
A covalent bond consists of the mutual sharing of one or more pairs of electrons between two atoms. These electrons are simultaneously attracted by the two atomic nuclei. A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions.
Can form four covalent bonds?
A: Carbon can form four covalent bonds. Covalent bonds are chemical bonds that form between nonmetals. In a covalent bond, two atoms share a pair of electrons. By forming four covalent bonds, carbon shares four pairs of electrons, thus filling its outer energy level and achieving stability.What are the electrons in a covalent bond doing?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.
What kind of particle is formed after covalent bonding?Particles formed from the covalent bonding of atoms are called ions. Polar molecules share their electrons equally.
Article first time published onHow many bonds nitrogen can form?
Nitrogen atoms will form three covalent bonds (also called triple covalent) between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell.
How do you find the number of covalent bonds in a molecule?
A covalent bond is formed between two atoms by sharing electrons. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet.
How many electrons are in covalent bond?
A covalent bond can be thought of as a ‘shared pair’ of electrons, so there are 2 electrons in each bond.
How many bonds can bromine form?
So fluorine and bromine are halogens (Group VII) and when you work out the Lewis dot structure you’ll see that they each have one unpaired electron, therefore these unpaired electrons join together to form one bond.
Do covalent bonds form molecules?
Covalent bonds and molecules When atoms combine by forming covalent bonds, the resulting collection of atoms is called a molecule. We can therefore say that a molecule is the simplest unit of a covalent compound.
Which particles are involved in bonding?
The subatomic particle involved in chemical bonding is the electron. Electrons are the smallest of all subatomic particles and orbit the nucleus in…
Why do bonds form between atoms?
Atoms form chemical bonds to make their outer electron shells more stable. … An ionic bond, where one atom essentially donates an electron to another, forms when one atom becomes stable by losing its outer electrons and the other atoms become stable (usually by filling its valence shell) by gaining the electrons.
Do lone pair electrons form covalent bonds?
Summary. Covalent bonds form when electrons in two atoms form overlapping orbitals. Lone pair electrons in an atom are not shared with another atom.
Why does carbon form covalent?
Carbon has 4 electrons in its outermost shell. … Therefore, carbon completes its octet by sharing its 4 electrons with other carbon atoms or with atoms of other elements and forms covalent bond. It forms strong covalent bonds because of its small size.
Why do multiple bonds form in covalent compounds?
Covalent bonding is the sharing of one or more electron pairs. In many covalent bonding situations, multiple chemical bonds exist — more than one electron pair is shared. … A nitrogen atom can fill its octet by sharing three electrons with another nitrogen atom, forming three covalent bonds, a so-called triple bond.
How do atoms form covalent double bonds?
A double covalent bond is where two pairs of electrons are shared between the atoms rather than just one pair. Two oxygen atoms can both achieve stable structures by sharing two pairs of electrons as in the diagram. The double bond is shown conventionally by two lines joining the atoms.
Does nitrogen form covalent bonds?
Nitrogen atoms can form three covalent bonds, so a triple bond forms between them.
What elements form nonpolar covalent bonds?
Thus ethane, ethylene, and acetylene have nonpolar covalent bonds, and the compounds are nonpolar. Bonds between carbon and other elements such as oxygen and nitrogen are polar. The polarity of a bond depends on the electronegativities of the bonded atoms.
Why nitrogen can form 4 bonds?
If you look at the above image you can see that when nitrogen has a positive charge (one less electron), it can form four covalent bonds. Either with single, double, or triple bonds. It is similar to phosphorus in this regard because they both have five valence electrons (four when they have a positive charge).
How many covalent bonds are there in molecule of ethane?
The total number of covalent bonds that are present in the ethane molecule is seven. So, the correct answer is B. Seven covalent bonds are present in ethane.
Does each bond have 2 electrons?
Electrons can be shared between atoms, and the sharing is more equal if the atoms are similar or the same, and more unequal if the atoms are different. Two shared electrons make one chemical bond.
Do bonds count as 2 electrons?
A covalent bond is pictured as two electrons shared by two different atomic nuclei, which means that each of the two nuclei has two electrons.
What are binary covalent bonds?
A binary covalent compound is composed of two different elements (usually nonmetals). For example, a molecule of chlorine trifluoride, ClF3 contains 1 atom of chlorine and 3 atoms of fluorine.
What type of bond will form between bromine and bromine?
To do that, a bromine atom forms a covalent bond with another bromine atom. Each atom contributes one electron to the bond, and share these bonding electrons equally. Hence, the bromine molecule, Br2 , is formed.
What covalent bonds can fluorine form?
Fluorine and the other halogens in group 7A (17) have seven valence electrons and can obtain an octet by forming one covalent bond. Typically, the atoms of group 4A form 4 covalent bonds; group 5A form 3 bonds; group 6A form 2 bonds; and group 7A form one bond.