Western blot. A western blot is a laboratory method used to detect specific protein molecules from among a mixture of proteins. Western blots can also be used to evaluate the size of a protein of interest, and to measure the amount of protein expression..
Also know, where is Western blotting used?
Western blot is often used in research to separate and identify proteins. In this technique a mixture of proteins is separated based on molecular weight, and thus by type, through gel electrophoresis. These results are then transferred to a membrane producing a band for each protein.
Secondly, which gel is used in Western blotting? Softer and thinner gels allow efficient transfer of proteins. Nitrocellulose and polyvinylidene difluoride (PVDF) papers are the most commonly used membranes used for the western blotting approach.
Also know, what is the difference between immunoblotting and Western blotting?
Western Blotting (also called immunoblotting) is a technique used for analysis of individual proteins in a protein mixture (e.g. a cell lysate). The proteins on this immunoblot are then accessible for antibody binding for detection. Antibodies are used to detect target proteins on the western blot (immunoblot).
Why is Western blot better than Elisa?
Western Blotting is the most common method of testing to confirm positive results from ELISA test. One advantage of Western Blotting is that it's less likely to give false positive results as it can effectively distinguish between HIV antibodies and other antibodies.
Related Question Answers
Why is it called Western blot?
The name western blot is a play on the Southern blot, a technique for DNA detection named after its inventor, English biologist Edwin Southern. Similarly, detection of RNA is termed as northern blot. The term "western blot" was given by W.Can Western Blot be false negative?
A positive Western blot confirms an HIV infection. A negative Western blot test means the ELISA test was a false positive test. The Western blot test can also be unclear, in which case more testing is done. Negative tests do not rule out HIV infection.What is the Western blot test for Lyme disease?
The ELISA and Western blot are the most common tests used to diagnose Lyme disease. Both tests measure antibodies (proteins made by the immune system to fight infection) to Bb, the bacterium that causes Lyme disease.How long does a Western blot test take?
Results. A Lyme disease test detects antibodies to the Lyme disease bacteria Borrelia burgdorferi in the blood. Test results are usually available in 1 to 2 weeks.Is Western blot quantitative?
Western blot is a reliable quantitative method only if sample properties and integrity, antibody specificity to the target protein, and loading protocols are considered. With careful attention to details, you can avoid common mistakes and avoid misinterpreting Western blot data.Why is milk used in Western blotting?
Milk contains a lot of proteins which bind to the membrane to reduce background, non specific binding. Antibodies are proteins, so they can bind to the membrane inespecifically, this is, they bind to any site in the membrane (only due to being proteins) apart from binding to their antigen.When running a Western blot What is the purpose of the marker?
A protein marker (also called a protein molecular weight marker, a protein MW marker, or a protein ladder) is used to estimate the size of proteins resolved by gel electrophoresis.What is the Western Blot test used for?
The Western blot test separates the blood proteins and detects the specific proteins (called HIV antibodies) that indicate an HIV infection. The Western blot is used to confirm a positive ELISA, and the combined tests are 99.9% accurate.What information does the Western blot provide for each sample?
Western blotting is incredibly informative for determining the effect of time on a protein. For example, if each sample is a protein mixture of cells that are in different phases of the cell cycle, then western blotting will reveal how much a protein is present or absent during each phase.Is Western Blot still used?
In case of a positive result from this test, the ELISA test was previously followed by a test called a Western blot to confirm the diagnosis. However, the Western blot is no longer used, and today the ELISA test is followed by an HIV differentiation assay to confirm HIV infection.How does ECL work?
Enhanced Chemiluminescence: How Does It Work? Basically, ECL is based on antibodies that are conjugated or labeled with horseradish peroxidase (HRP). In a typical chemiluminescent assay, the light emitted is usually of low intensity and does not last long enough to make an accurate detection and analysis.What is the purpose of immunofluorescence?
Immunofluorescence is an assay which is used primarily on biological samples and is classically defined as a procedure to detect antigens in cellular contexts using antibodies. Immunofluorescence can be used on cultured cell lines, tissue sections, or individual cells.Why is immunoblotting a 2 step process?
Historically, the probing process was performed in two steps because of the relative ease of producing primary and secondary antibodies in separate processes. This gives researchers and corporations huge advantages in terms of flexibility, and adds an amplification step to the detection process.How primary antibody is prepared for Western blot?
For western blots, incubate membrane with diluted primary antibody in either 5% w/v BSA or nonfat dry milk, 1X TBS, 0.1% Tween® 20 at 4°C with gentle shaking, overnight.When was Western blot discovered?
But who deserves the real credit? Most researchers know western blotting evolved from Southern blotting (Ref 1), invented by Edwin Southern at University of Edinburgh in 1975, then northern blotting (Ref 2), invented by George Stark's Stanford group in 1977.Is SDS PAGE a Western blot?
SDS-PAGE (1D) separates protein based on molecular weight, while western blotting is done to detect the protein of interest using specific antibodies.What does Tween do in Western blot?
TWEEN should be in the blocking buffer to saturate all potential non-specific binding sites on your membrane. When all of them blocked by TWEEN the non-specific proteins in your 1st antibody solution (and milk) will not be able to bind to the membrane.How does Western blot identify proteins?
A western blot is a laboratory method used to detect specific protein molecules from among a mixture of proteins. Next, the protein molecules are separated according to their sizes using a method called gel electrophoresis. Following separation, the proteins are transferred from the gel onto a blotting membrane. 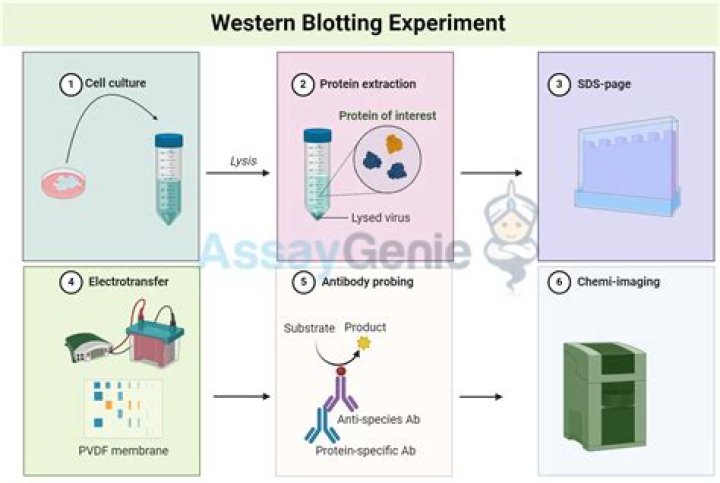
 Christopher Lucas
Christopher Lucas