What is the uses of fractional distillation?
 Isabella Bartlett
Isabella Bartlett .
Similarly, it is asked, what is fractional distillation used for?
Fractional distillation is a process by which components in a chemical mixture are separated into different parts (called fractions) according to their different boiling points. Fractional distillation is used to purify chemicals and to separate mixtures to obtain their components.
Similarly, what are the advantages of fractional distillation? If so, fractional distillation achieves a greater degree of separation between products by taking advantage of multiple vaporisation and condensation processes in the column. So, purity and separation of products are the main advantages.
Similarly, it is asked, what is fractional distillation in simple words?
Fractional distillation is a process of separating a mixture of chemical compounds. This means that each part (called a "fraction") of the mixture can be kept apart from the other chemicals. Different chemicals have different boiling points.
Why do we use fractional distillation?
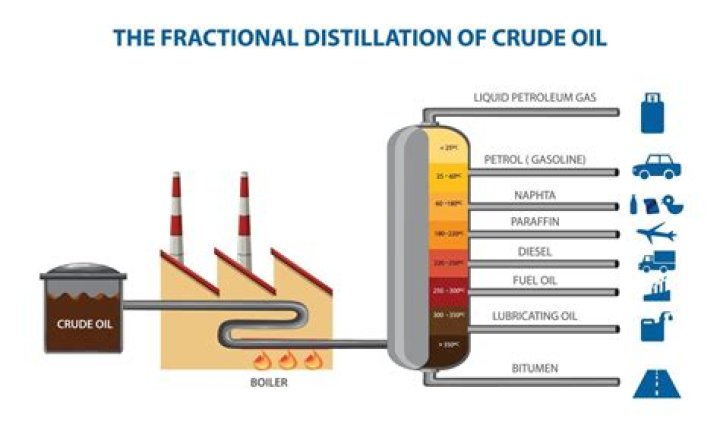
For example, fractional distillation is used in oil refineries to separate crude oil into useful substances (or fractions) having different hydrocarbons of different boiling points. The crude oil fractions with higher boiling points: have more carbon atoms.
Related Question AnswersWhat are the examples of fractional distillation?
Fractional distillation is used to purify chemicals and also to separate mixtures to obtain their components. crude oil being separated into numerous chemicals, such as gasoline, diesel fuel and acetone are examples are examples of fractional distillation.What are the advantages and disadvantages of simple distillation vs fractional distillation?
Simple distillation Fractional distillation Advantages ? simpler setup than fractional ? faster distillation times ? consumes less energy than fractional distillation ? much better separation between liquids than simple distillation ? can more readily purify complex mixtures than simple distillation Disadvantages ?What is fractional distillation explain with diagram?
Fractional Distillation: It is a process of separation of a chemical mixture whose components have different boiling points. It is an special type of distillation in which two miscible liquids having different boiling points but closed to each other are separated by fractionating column.Is fractional distillation bad for the environment?
Environment polluted, the most toxic substances will go to the human body. So protect the environment is very important. Extraction and fractional distillation need use energy. Fractional distillation need heat, it can let crude oil become to gas then crude oil can fractional distillation to many different products.Is fractional distillation a chemical or physical process?
Is fractional distillation a chemical or physical reaction? Fractional distillation is a strictly physical process. The idea is that part of the mixed solution boils off leaving the higher boiling point component behind.What is difference between simple and fractional distillation?
1, Simple distillation is the method used to separate substances in mixtures with significantly different boiling points, while fractional distillation is used for mixtures containing chemicals with boiling points close to each other.What is the importance of distillation?
The distillation process is used to separate components based on their boiling points. Chemicals like common gas, diesel, and jet fuel achieve their boiling points at different temperatures. Distillation columns are used to separate mixed feed streams into their own distinct products.What is the principle of distillation?
Distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. To separate a mixture of liquids, the liquid can be heated to force components, which have different boiling points, into the gas phase.What is the method of fractional distillation?
Fractional distillation is a process by which individual components can be separated using heat from a given mixture. The boiling points of each component in the mixture determine the order of separation. Common uses of fractional distillation include the production of distilled water, and gasoline from crude oil.What main advantage and disadvantage can you predict about fractional distillation?
Fractional distillation can make better separation than simple distillation. A distillation can be performed by fractional distillation even the boiling points of each components are close. The disadvantage is the column has too large surface area that liquid vapor too fast.Why simple distillation is not effective?
We have already said that the simple distillation is less efficient at separating liquids, because there is a smaller surface area inside the column, but it is usually much faster. This means that the fractions obtained are impure, and often contain traces of the other liquids found in the original mixture.Why is simple distillation effective?
Simple distillation is effective only when separating a volatile liquid from a nonvolatile substance or when separating two liquids that differ in boiling point by 50 degrees or more. As vapors continue to rise through the column, the liquid that has condensed will revaporize.How do we use distillation in everyday life?
Practical Uses of Distillation- Scientific Uses. One practical use of distillation is in the laboratory.
- Water Purification. Water from natural sources contains a variety of minerals and other impurities, many of which can be removed by distillation.
- Alcoholic Beverages.
- Petroleum Products.
- Perfume.
- Food Flavorings.