The presence of an alcohol can be determined with test reagents that react with the -OH group. The initial test to identify alcohols is to take the neutral liquid, free of water and add solid phosphorus(V) chloride. A a burst of acidic steamy hydrogen chloride fumes indicate the presence of an alcohol..
Correspondingly, how do you test for primary and secondary alcohols?
You would then add a few drops of the alcohol to a test tube containing potassium dichromate(VI) solution acidified with dilute sulphuric acid. The tube would be warmed in a hot water bath. In the case of a primary or secondary alcohol, the orange solution turns green. With a tertiary alcohol there is no colour change.
Also, what is Jones test for? A Jones II dye test is used to determine the presence or absence of anatomical obstruction of the nasolacrimal outflow system. A positive Jones II dye test (colored fluid from the nose) indicates a patent system anatomically.
Moreover, how do you test for tertiary alcohols?
A positive test for aldehydes and primary or secondary alcohols consists in the production of an opaque suspension with a green to blue color. Tertiary alcohols give no visible reaction within 2 seconds, the solution remaining orange in color. Disregard any changes after 15 seconds. Enols may give a positive test.
Does a urine test show alcohol use?
The EtG test is quite sensitive and can detect even low levels of alcohol. In fact, the test can detect alcohol in the urine up to five days after consumption. In studies of participants without alcohol-use disorders, EtG has been detected in urine samples for up to 80 hours (3.3 days) after heavy alcohol exposure.
Related Question Answers
How do you test for primary alcohol?
A few drops of the alcohol are added to a test tube containing potassium dichromate(VI) solution acidified with dilute sulfuric acid. The tube is warmed in a hot water bath. In the case of a primary or secondary alcohols, the orange solution turns green.What happens when alcohol is oxidised?
Primary alcohols can be oxidized to either aldehydes or carboxylic acids, depending on the reaction conditions. In the case of the formation of carboxylic acids, the alcohol is first oxidized to an aldehyde, which is then oxidized further to the acid.How do you test for aldehydes?
To carry out the test, you add a few drops of the aldehyde or ketone to the freshly prepared reagent, and warm gently in a hot water bath for a few minutes. No change in the colourless solution. The colourless solution produces a grey precipitate of silver, or a silver mirror on the test tube.How does Lucas reagent work?
Lucas' reagent is a solution of anhydrous zinc chloride in concentrated hydrochloric acid. This solution is used to classify alcohols of low molecular weight. The reaction is a substitution in which the chloride replaces a hydroxyl group.How can you tell the difference between a ketone and alcohol?
Aldehydes and ketones react with 2,4-dinitrophenylhydrazine reagent to form yellow, orange, or reddish-orange precipitates, whereas alcohols do not react. Formation of a precipitate therefore indicates the presence of an aldehyde or ketone. The precipitate from this test also serves as a solid derivative.How do you test for ethanol?
FOR ETHANOL. To determine if ethanol is in the gas: On a test tube or olive bottle six or seven inches long, make a permanent line about two inches from the bottom. Fill with water to this line, then fill the tube to the top with gasoline.Is 1 butanol a primary alcohol?
1-Butanol, or butyl alcohol, is a four-carbon chain, with the OH group on an end carbon. It is used as a solvent and a paint thinner, and has some potential use as a biofuel. Butyl alcohol is a primary (1º) alcohol, and is easily oxidized. sec-Butyl alcohol is a secondary (2º) alcohol, and is easily oxidized.Is ethanol a secondary alcohol?
A primary alcohol is an alcohol which has the hydroxyl group connected to a primary carbon atom. In contrast, a secondary alcohol has a formula “–CHROH” and a tertiary alcohol has a formula “–CR2OH”, where “R” indicates a carbon-containing group. Examples of primary alcohols include ethanol and butanol.What happens when a secondary alcohol is oxidized?
Oxidation of secondary alcohols to ketones. The oxidation of secondary alcohols to ketones is an important oxidation reaction in organic chemistry. Where a secondary alcohol is oxidised, it is converted to a ketone. The hydrogen from the hydroxyl group is lost along with the hydrogen bonded to the second carbon.Is cyclohexanol a primary alcohol?
Cyclohexanol is an alcohol that consists of cyclohexane bearing a single hydroxy substituent. The parent of the class of cyclohexanols. It has a role as a solvent. It is a secondary alcohol and a member of cyclohexanols.What type of alcohol is ethanol?
Ethyl Alcohol, or ethanol (C2H5OH), is the type used in the production of alcoholic beverages. The other three types, methyl, propyl and butyl alcohol, if consumed can result in blindness and death, even in relatively small doses. Alcohol, or ethanol, is the intoxicating agent found in beer, wine and liquor.How do you turn an alcohol into an aldehyde?
Making aldehydes Aldehydes are made by oxidising primary alcohols. There is, however, a problem. The aldehyde produced can be oxidised further to a carboxylic acid by the acidified potassium dichromate(VI) solution used as the oxidising agent. In order to stop at the aldehyde, you have to prevent this from happening.Why does acidified potassium dichromate turn green?
Acidified potassium dichromate(VI) is an oxidising agent that oxidises primary alcohols, secondary alcohols and aldehydes. During oxidation, dichromate(VI) ions are reduced and the colour changes from orange to green.What do you mean by esterification?
Esterification is a chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters are produced when acids are heated with alcohols in a process called esterification. An ester can be made by an esterification reaction of a carboxylic acid and an alcohol.Is cro3 a strong oxidizing agent?
Chromium Trioxide (CrO3) Chromium trioxide is a strong oxidizing agent that is not soluble in most organic solvents and tends to explode in the presence of organic compounds and solvents. A solution of chromium trioxide in aqueous sulfuric acid can be safely mixed with acetone (Jones Reagent).How do you test carboxylic acids?
A few drops or a few crystals of the unknown sample are dissolved in 1mL of methanol and slowly added to 1 mL of a saturated solution of sodium bicarbonate. Evolution of a carbon dioxide gas is a positive test for the presence of the carboxylic acid and certain phenols listed in the Complications section.Why is acetone used in Jones oxidation?
Chromic acid, also known as Jones reagent, is prepared by adding chromium trioxide (CrO3) to aqueous sulfuric acid. The Jones oxidation also uses acetone as a co-solvent in the reaction to prevent over-oxidation of the organic product. Ketones are not oxidized by chromic acid, so the reaction stops at the ketone stage.What is regurgitation test?
Regurgitation on pressure over the lacrimal sac (ROPLAS) is a clinical test that aids in diagnosing nasolacrimal duct obstruc- tion (NLDO) with a specificity as high as 99%. The maneuver is often resorted to by patients to empty the nasolacrimal sac and may provide relief from purulent discharge for a few hours.Is methanol a primary alcohol?
Methanol, CH3OH, is counted as a primary alcohol even though there are no alkyl groups attached to the carbon with the -OH group on it. In a secondary (2°) alcohol, the carbon with the -OH group attached is joined directly to two alkyl groups, which may be the same or different. 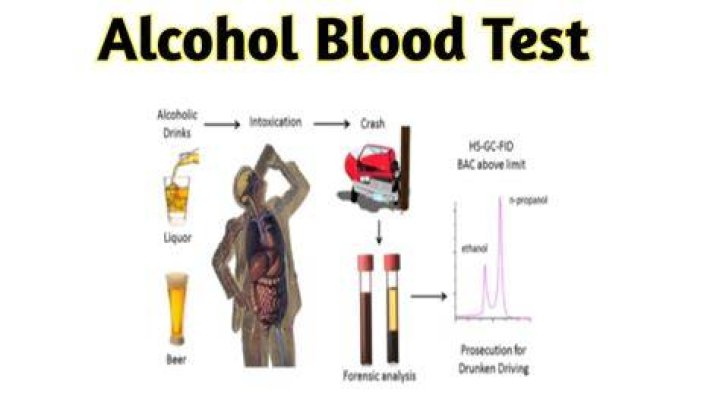
 Emily Sparks
Emily Sparks