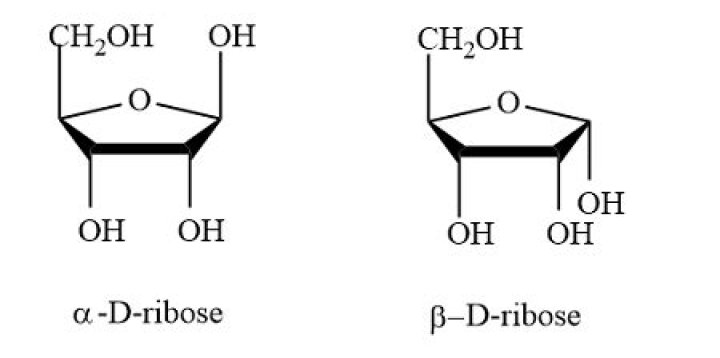
What is the structure of D Ribose?
 Emily Sparks
Emily Sparks What is the structure of D Ribose?
C5H10O5
Ribose/Formula
What is Haworth projection formula?
A Haworth projection is a common way of writing a structural formula to represent the cyclic structure of monosaccharides with a simple three-dimensional perspective. Organic chemistry and especially biochemistry are the areas of chemistry that use the Haworth projection the most.
Which is the correct representation of ribose?
Ribose is a simple sugar and carbohydrate with molecular formula C5H10O5 and the linear-form composition H−(C=O)−(CHOH)4−H.
Is ribose optically active?
Ribose exists as optically active D-ribose and L-ribose and as an inactive racemate. The melting point for D-ribose is 86°–87°C. Ribose characteristically has a high (8.5 percent) acyclic (aldehyde) content in solution. D-ribose is found in all living organisms.
What does ribose look like?
Ribose is an organic compound classified as a monosaccharide, or simple sugar. Ribose is composed of five carbon atoms, ten hydrogen atoms, and five oxygen atoms that have been bonded together. This means that the five carbons that form the majority of the structure give the molecule a pentagon shape.
Is ribose ad or L Stereoisomer?
There is a quartet of five-carbon aldehyde sugars (aldopentoses): ribose, arabinose, xylose, and lyxose, each existing as a pair of enantiomers (D- and L- ). The most familiar name on that list should be ribose, which is the sugar backbone of ribonucleic acid (RNA).
What are Fischer and Haworth projection?
Fischer and Haworth projections are two types of illustration which are used to represent the 3D arrangement of atoms in carbohydrates. They are also used to compare different carbohydrates.
Is ribose and D-Ribose the same?
Also known as D-ribose, it is sold as a nutritional supplement to reduce fatigue and improve athletic performance. The synthetic form of this supplement is called N-ribose.
What is the difference between ribose and D-Ribose?
Ribose and deoxyribose are monosaccharides or simple sugars….Deoxyribose vs Ribose.
| Deoxyribose | Ribose |
|---|---|
| 2-deoxy-D-ribose | (2S,3R,4S,5R)-5-(hydroxymethyl)oxolane-2,3,4-triol |
| Structure | |
| It has a hydroxyl (OH) group at position 2 | It has a hydrogen (H) atom at position 2 |
| Molar mass |
Is ribose a Furanose?
Ribose and Deoxyribose. Ribose is the most common pentose (5 carbon sugar). However most of the time the sugar forms a five-atom ring structure called a furanose (left image).
Are L and D isomers mirror images?
L isomer is the mirror image of the D isomer. Physical properties are the same for two mirror images because the molecular masses are the same. Chemical properties are also similar because the same functional groups are present in both molecules.





