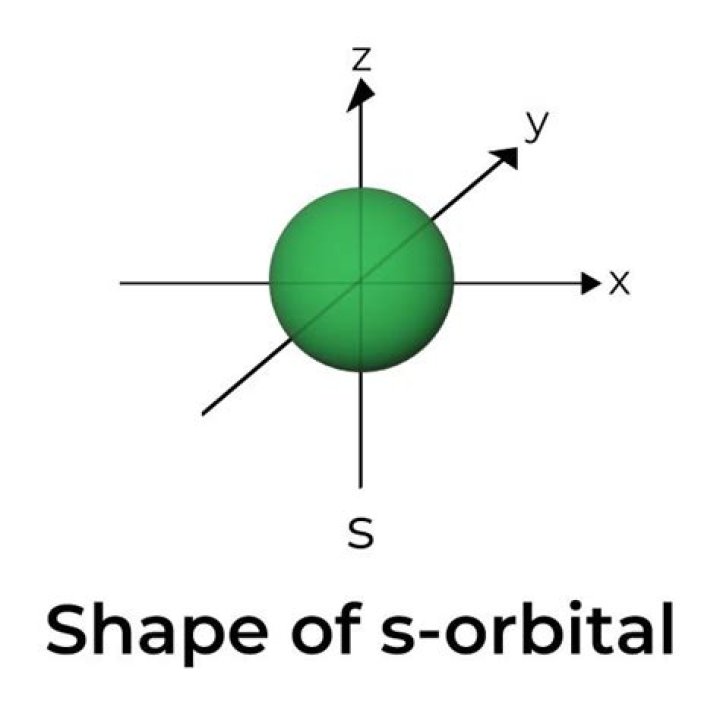
What is the shape of the S orbital
 Rachel Young
Rachel Young The s orbital is a spherical shape. The p orbital is a dumbbell shape. There are three p orbitals that differ in orientation along a three-dimensional axis.
What is the shape of an s orbital quizlet?
All s orbitals are spherical shape.
What is the shape of S Sharp orbital?
An s orbital is spherical with its centre at the nucleus. Thus a 1s electron is almost entirely confined to a spherical region close to the nucleus; a 2s electron is restricted to a somewhat larger sphere.
What shape is the 1s orbital?
The closest orbital to the nucleus, called the 1s orbital, can hold up to two electrons. This orbital is equivalent to the innermost electron shell of the Bohr model of the atom. It is called the 1s orbital because it is spherical around the nucleus.What are the shapes of s and p orbitals quizlet?
S orbitals have no angular nodes and are spherical. P orbitals have a single angular node across the nucleus and are shaped like dumbbells.
Why is s orbital spherical?
The s orbital has an angular momentum quantum number, l of zero. This means that all s orbitals have zero angular nodes, i.e. there is no angle taken from the nucleus at which there is zero electron density. This results in a spherical shape.
What is the shape of ap orbital *?
p orbitals are said to be “dumbbell shaped”. There are three p orbitals, each with two regions of probability on opposite sides of the nucleus along the x, y, or z axis. A p orbital has the approximate shape of a pair of lobes on opposite sides of the nucleus, or a somewhat dumbbell shape.
What is the shape of 2s orbital?
Thus all s orbitals such as the 1s, 2s are spherical. An important point is that only a limited number of orbital shapes is possible for each value of n.How many s orbitals are there?
The s sublevel has just one orbital, so can contain 2 electrons max. The p sublevel has 3 orbitals, so can contain 6 electrons max.
What does s orbital stand for?The s orbital is a sphere around the atomic nucleus. Within the sphere there are shells in which an electron is more likely to be found at any given time. … As with the s orbital, the p orbital describes a region in space around the nucleus in which an electron may be found with the highest probability.
Article first time published onHow many electrons are in s orbital?
This means that the s orbital can contain up to two electrons, the p orbital can contain up to six electrons, the d orbital can contain up to 10 electrons, and the f orbital can contain up to 14 electrons.
Which orbital is the daisy shaped orbital?
The general shape of the d-orbitals can be described as “daisy-like” or “four leaf clover” with the exception of the the dz2 orbital which looks like the donut with a lobe above and below.
How many shapes does af orbital have?
The most complex set of orbitals are the f orbitals. When l = 3, ml values can be −3, −2, −1, 0, +1, +2, +3 for a total of seven different orbital shapes. Again, note the specific orientations of the different f orbitals.
What is the shape of a p orbital how many p orbitals are there in a sublevel?
Each principal energy level above the first contains one s orbital and three p orbitals. A set of three p orbitals, called the p sublevel, can hold a maximum of six electrons. Therefore, the second level can contain a maximum of eight electrons – that is, two in the s orbital and 6 in the three p orbitals.
How are s orbitals different from p orbitals quizlet?
The s orbitals are spherical, and the p orbitals are dumbbell shaped.
Why s orbital is non directional?
s orbitals are non-directional because they have spherical symmetry which means that the probability of finding an electron at a particular distance from the nucleus is same in all directions as compared with a p orbital which has somewhat a dumbbell like shape.
What are the 4 types of orbitals?
There are four basic types of orbitals: s, p, d, and f. An s orbital has a spherical shape and can hold two electrons.
How many orbitals are in the S sub shell?
Thus the s subshell has only one orbital, the p subshell has three orbitals, and so on.
What orbital is shaped like dumbbells?
Named for their energy sublevels, there are four types of orbitals: s, p, d, and f. Each orbital type has a unique shape based on the energy of its electrons. The s orbital is a spherical shape. The p orbital is a dumbbell shape.
How many orbitals are in 1s subshell?
Johnson Z. There is 1 orbital in the 1s sublevel.
What is the difference between 1s and 2s orbitals?
The 2s orbital is larger than 1s orbital. Hence, its radius is larger than that of the 1s orbital. … Its energy is higher than 1s orbital but is lower than other orbitals in an atom. 2s orbital also can be filled only with one or two electrons.
What is the shape of 2s and 2p orbital?
A 2 s orbital is spherical, and there is only one of them. A 2 p orbital is dumbbell-shaped, and there are three of them oriented on the x, y, and z axes. The 2 p orbitals have higher energy than the 2 s orbital.
Where is s orbital?
s ORBITALS An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.
Why is s shell called sharp?
The sharp series has given the letter s to the s atomic orbital or subshell. … The limit for the series corresponds to electron emission, where the electron has so much energy it escapes the atom. Even though the series is called sharp, the lines may not be sharp.
Why does the s orbital fill before the p orbitals?
Notice that the s orbital always has a slightly lower energy than the p orbitals at the same energy level, so the s orbital always fills with electrons before the corresponding p orbitals do.
What is the difference between s orbital and p orbital?
Main Difference – S Orbital vs P Orbital An atom is composed of electrons that are in continuous movement in any direction around the nucleus. … The main difference between s orbital and p orbital is that s orbitals are spherical shaped whereas p orbitals are dumbbell shaped.
What is 1s 2s 2p 3s 3p?
In the question 1s 2s 2p 3s 3p represents electron orbital energy levels. … The sequence of orbital energy levels is as always-1s < 2s = 2p < 3s = 3p = 3d <4s = 4p = 4d= 4f. The orbital having the same energy is called a degenerate orbital.
What are the 7 orbitals?
The shape of the seven 7f orbitals (cubic set). From left to right: (top row) 7fy 3, 7fz 3, 7fx 3, (middle row) 7fy(z 2-x 2), 7fz(x 2-y 2), and 7fx(z 2-y 2) (bottom row) 7fxyz. For each, the green zones are where the wave functions have positive values and the white zones denote negative values.
What is the shape of orbital with L 2 and L 3?
The orbitals with l= 2 are the d orbitals which have complex shapes with at least two nodal surfaces. The orbitals with l= 3 are called the f orbitals that are more complex.
Why is p orbital dumbbell shaped?
The p orbital is a dumbbell shape because the electron is pushed out twice during the rotation to the 3p subshell when an opposite-spin proton aligns gluons with two same-spin protons.
What does sublevel s look like?
The s-sublevel is made up of a singular orbital holding a maximum of 2 electrons. It is a spherical shape. The 1s is the closest to the nucleus and is smaller that the 2s, which is smaller than the 3s and so on. … The d-sublevel is made up of a 5 different orbitals and the sublevel holds a maximum of 10 electrons.





