What is the role of BIS acrylamide in SDS PAGE
 Andrew Campbell
Andrew Campbell Bis-acrylamide is used to create crosslinks between acrylamide to generate polyacrylamide gel in electrophoresis gels. The ratio of bis-acrylamide to acrylamide manipulates the porous characteristics of the polyacrylamide gel.
What is the purpose of using SDS on the acrylamide gel?
The combined use of sodium dodecyl sulfate (SDS, also known as sodium lauryl sulfate) and polyacrylamide gel allows to eliminate the influence of structure and charge, and proteins are separated solely on the basis of differences in their molecular weight.
What is the role of ammonium persulfate in SDS PAGE?
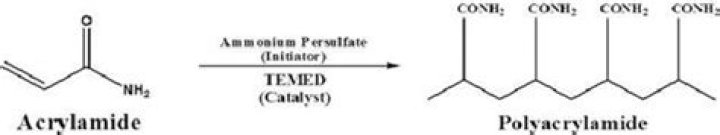
Ammonium Persulfate (APS) is an oxidizing agent that is used with TEMED to catalyze the polymerization of acrylamide and bisacrylamide. … APS is an oxidizing agent which spontaneously decomposes to form free radicals and is used with TEMED to catalyze the Polymerization of acrylamide and bisacrylamide monomers.
Why is the concentration of the acrylamide in the gel important?
Increased concentrations of acrylamide result in decreased pore size after polymerization. Polyacrylamide gel with small pores helps to examine smaller molecules better since the small molecules can enter the pores and travel through the gel while large molecules get trapped at the pore openings.What is the purpose of acrylamide and bis acrylamide?
Bis-acrylamide is used to create crosslinks between acrylamide to generate polyacrylamide gel in electrophoresis gels. The ratio of bis-acrylamide to acrylamide manipulates the porous characteristics of the polyacrylamide gel.
How does acrylamide concentration determine the resolution of SDS-PAGE gel?
Higher concentrations of acrylamide form denser gels with smaller pore sizes, which is ideal for separating low molecular weight proteins with high resolution. … Other factors to consider include gels with varying pore sizes.
What is the role of SDS in SDS-PAGE Mcq?
Sodium dodecyl sulfate (SDS) is an anionic detergent and is most commonly used in protein chemistry to denature proteins and dissolve proteins and protein aggregates immersed in water, such as in SDS-PAGE.
What is the function of 5% stacking gel in polyacrylamide gel electrophoresis?
The purpose of stacking gel is to line up all the protein samples loaded on the gel, so that they can enter the resolving gel at the same time.What is the role of SDS in protein electrophoresis?
SDS-PAGE separates proteins primarily by mass because the ionic detergent SDS denatures and binds to proteins to make them uniformly negatively charged. Thus, when a current is applied, all SDS-bound proteins in a sample will migrate through the gel toward the positively charged electrode.
What is the purpose of adding ammonium persulfate to the gel solutions?Ammonium persulfate (APS) is an oxidizing agent that is often used with tetramethylethylenediamine (TEMED, Part No. 17919) to catalyze the polymerization of acrylamide and bisacrylamide to prepare polyacrylamide gels for electrophoresis.
Article first time published onWhat is the role of ammonium persulfate that will be added during polyacrylamide gel formation?
Ammonium persulfate acts as a buffer between pH 8 and 9.
Why Tris buffer is used in SDS PAGE?
Tris is the buffer used for most SDS-PAGE. Its pKa of 8.1 makes it an excellent buffer in the 7-9 pH range. … SDS in the buffer helps keep the proteins linear. Glycine is an amino acid whose charge state plays a big role in the stacking gel.
Why do we use a mixture of acrylamide and bisacrylamide in SDS PAGE?
Acrylamide forms linear polymers whereas Bisacrylamide cross links these linear polymer. More numbers of cross links means smaller pore size. So, ratio of Acrylamide and Bisacrylamide determines pore size. … Just to help out a little more, we generally use a 30% Bis-Acrylamide solution for our standard SDS-PAGE.
What is the structure of bis acrylamide?
PubChem CID8041StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaC7H10N2O2SynonymsN,N’-METHYLENEBISACRYLAMIDE 110-26-9 N,N’-Methylenediacrylamide N,N’-Methylene-bis-acrylamide Methylenebisacrylamide More…
Why is SDS used in SDS-PAGE?
SDS (sodium dodecyl sulfate) is an anionic detergent that unfolds and denatures proteins, coating proteins in negative charge. It is added in excess to the proteins, so that the proteins’ intrinsic charge is covered, and a similar charge-to-mass ratio is obtained for all proteins.
What does SDS stand for in SDS-PAGE?
In SDS-PAGE, the use of sodium dodecyl sulfate (SDS, also known as sodium lauryl sulfate) and polyacrylamide gel largely eliminates the influence of the structure and charge, and proteins are separated solely based on polypeptide chain length.
What is the role of beta mercaptoethanol in SDS-PAGE?
The role of beta-mercaptoethanol is to break all the disulfide bonds and denature the protein of interest.
How do you make BIS with Tris gel?
- 5X Low MW Running Buffer. 250 mM MES. 250 mM Tris. …
- 5X High MW Running Buffer. 250 mM MOPS. 250 mM Tris. …
- 200X Running Buffer Reducing Agent. 1 M sodium bisulfite. Add to running buffer at 5mM final concentration.
- 3.5X Gel Buffer. 1.25 M bis-Tris (pH 6.5-6.8 with HCl)
How do you choose acrylamide percentage?
The smaller the size of the protein of interest, the higher the percentage of acrylamide/bis. The bigger the size of the protein of interest, the lower the percentage of acrylamide/bis.
How does the percentage of acrylamide affect mobility?
Hard gels (12-20% acrylamide) retard the migration of large molecules more than they do small ones. In certain cases, high concentration acrylamide gels are so tight that they exclude large molecules from entering the gel but allow the migration and resolution of low molecular weight components of a complex mixture.
What two roles does the SDS play in SDS-PAGE electrophoresis?
The Role of SDS (et al.) SDS is a detergent that is present in the SDS-PAGE sample buffer where, along with a bit of boiling, and a reducing agent (normally DTT or β-ME to break down protein–protein disulfide bonds), it disrupts the tertiary structure of proteins.
What is the purpose of using SDS for protein sample buffer and SDS-PAGE 2?
SDS is a strong detergent and present in high concentrations in the buffer that prepares samples for electrophoresis. Before samples such as cells can be run on a protein gel, SDS needs to lyse cell membranes and solubilize all proteins.
How do SDS denature proteins?
SDS is an amphipathic surfactant. It denatures proteins by binding to the protein chain with its hydrocarbon tail, exposing normally buried regions and coating the protein chain with surfactant molecules. … For this reason, separation on a polyacrylamide gel in the presence of SDS occurs by mass alone.
What is separating gel in SDS PAGE?
Polyacrylamide gels are formed by the reaction of acrylamide and bis-acrylamide (N,N’-methylenebisacrylamide) that results in highly cross-linked gel matrix. The gel acts as a sieve through which the proteins move in response to the electric field.
What is separating and stacking gel?
PAGE has two phases: a stacking gel and a separating gel. Under an applied electric field, the stacking gel concentrates the SDS-loaded linear protein molecules while the separating gel separates the proteins on the basis of molecular weight.
What is the purpose of adding APS and TEMED?
Ammonium Persulfate (APS) and TEMED catalyze the polymerization of acrylamide solutions into gel matrices. These gels are then used to separate a variety of macromolecules by size in the presence of an electric field.
What is ammonium persulfate in?
Ammonium persulfate is a standard ingredient in hair bleach. Persulfates are used as oxidants in organic chemistry. For example, in the Minisci reaction.
Why we add TEMED and APS at the end in making of stacking & running gel?
While making gel solution, TEMED has to be added last since it will immediately start to react with APS, and catalyze the polymerization of acrylamide and bisacrylamide. As a consequence, the following mixing and casting steps have to be completed as quickly as possible.
What is the purpose of tris buffer?
Tris is the main buffering component; its chief role is to maintain the pH of the buffer at a stable point, usually 8.0. Additionally, tris likely interacts with the LPS (lipopolysaccharide) in the membrane, serving to destabilize the membrane further.
Why tris buffer is prepared as an electrophoretic running buffer during the SDS-PAGE run?
Most SDS-PAGE gels, running buffers, and blotting buffers are buffered with Tris. … At this pH, ionized chloride ions migrate rapidly, raising the pH behind them and creating a voltage gradient with a zone of low conductivity, which causes glycine (from the running buffer) to ionize and migrate behind the chloride front.
Why do we use two pH in SDS-PAGE?
The main reason is to differentiate the rate of migration while the proteins are stacking into a tight band in the wells, before they enter resolving gel for separation. The respective pH influences the charge of ions in the running buffer, and thus their migration when electric current is turned on.