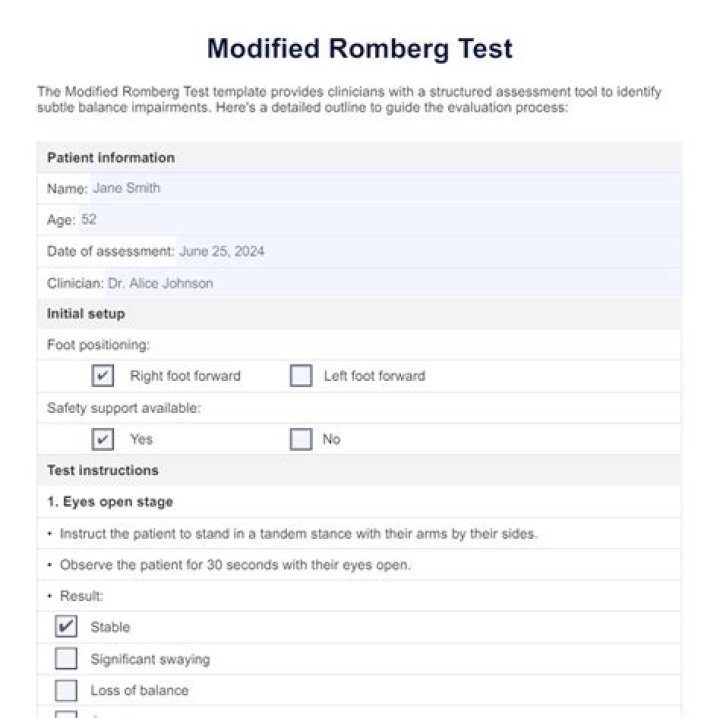
The core difference is that heat deals with thermal energy, whereas temperature is more concerned with molecular kinetic energy. Heat is the transfer of thermal energy, whereas temperature is a property the object exhibits..
Also question is, what is the relationship between thermal energy and temperature?
Temperature: The temperature does not depend on the quantity of the substance – it is related to the average kinetic energy of the particles. Thermal energy: The thermal energy depends on the quantity of the substance – it is related to the total kinetic energy of the particles.
One may also ask, is thermal energy and temperature the same? Thermal Energy, Temperature, and Mass Thermal energy and temperature are closely related. Both reflect the kinetic energy of moving particles of matter. However, temperature is the average kinetic energy of particles of matter, whereas thermal energy is the total kinetic energy of particles of matter.
Likewise, people ask, what is the difference between heat and thermal energy?
The difference between heat and thermal energy is that thermal energy is not in the process of being transferred; it is not in transit, but remains as part of the internal energy of the system; heat, on the other hand, is energy in transit, i.e. energy in the process of being transferred from a hotter system towards ..
What is an example of thermal energy?
Thermal energy is a form of kinetic energy. Examples of Thermal Energy: 1. Thermal energy from a hot stove is transferred to a metal pot and causes the water molecules to move faster increasing the temperature of the water.
Related Question Answers
How is thermal energy measured?
Thermal energy itself is expressed in British thermal units (Btu), calories and joules. One Btu is the amount of heat necessary to raise 1 lb. of water through 1 degree Fahrenheit. A calorie is the amount of thermal energy needed to raise the temperature of 1 gram of water by 1 degree Celsius.Does thermal energy increase with temperature?
When the temperature of an object increases, the average kinetic energy of its particles increases. Therefore, the thermal energy of an object increases as its temperature increases.What factors influence thermal energy?
What 3 factors influence the amount of thermal energy in an object, and how do they each affect it? Mass, temperature, and phase. More mass, more Thermal Energy. Higher temperature, more Thermal Energy.What is the similarities of heat and temperature?
Heat measures both kinetic and potential energy contained by molecules in an object. On the other hand, temperature measures average kinetic energy of molecules in substance. The main feature of heat is that it travels from hotter region to cooler region.What do you mean by thermal?
Thermal means relating to or caused by heat or by changes in temperature.What is difference between heat and energy?
The difference between heat and thermal energy is that thermal energy is not in the process of being transferred; it is not in transit, but remains as part of the internal energy of the system; heat, on the other hand, is energy in transit, i.e. energy in the process of being transferred from a hotter system towardsWhat are the three ways to transfer heat?
Heat can travel from one place to another in three ways: Conduction, Convection and Radiation. Both conduction and convection require matter to transfer heat. If there is a temperature difference between two systems heat will always find a way to transfer from the higher to lower system.What do you mean by thermal energy?
Thermal energy is the internal energy of an object due to the kinetic energy of its atoms and/or molecules. The atoms and/or molecules of a hotter object have greater kinetic energy than those of a colder one, in the form of vibrational, rotational, or, in the case of a gas, translational motion.How do you distinguish between temperature and heat give example?
Temperature is the degree of hotness and coldness of a body. Heat is the total kinetic energy and potential energy obtained by molecules in an object. Temperature is the average K.E of molecules in a substance. Heat flows from hot body to cold body.What is the definition of energy in physics?
Energy, in physics, the capacity for doing work. It may exist in potential, kinetic, thermal, electrical, chemical, nuclear, or other various forms. There are, moreover, heat and work—i.e., energy in the process of transfer from one body to another.What two factors does thermal energy depend on?
An object's thermal energy is dependent upon its temperature and mass. The higher the temperature of a given quantity of a substance, more is its thermal energy. Similarly, for the same temperature, higher mass of a substance will contain more thermal energy.How are thermal energy and temperature similar and different?
Thermal energy and temperature both depend on the kinetic energy of the particles in a substance. The particles in a substance are moving at many different speeds. Temperature does not depend upon the mass of an object. The thermal energy of a substance is a measure of the total kinetic energy of its particles.What is the unit of heat?
As a form of energy, heat has the unit joule (J) in the International System of Units (SI). However, in many applied fields in engineering the British thermal unit (BTU) and the calorie are often used. The standard unit for the rate of heat transferred is the watt (W), defined as one joule per second.Does higher thermal energy mean higher temperature?
Temperature is the average kinetic energy of particles of an object. Warmer objects have faster particles and higher temperatures. If two objects have the same mass, the object with the higher temperature has greater thermal energy. 
 Emily Sparks
Emily Sparks