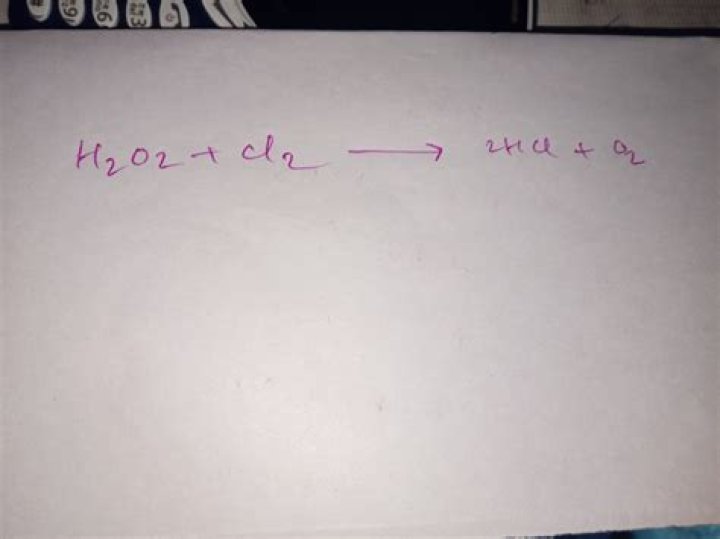
What is the product of the reaction of h2o2 with cl2
 William Burgess
William Burgess When Hydrogen peroxide reacts with chlorine it forms hydrogen chloride and oxygen.
What is the product of a reaction between h2 and O2?
When molecular hydrogen (H2) and oxygen (O2) are combined and allowed to react together, energy is released and the molecules of hydrogen and oxygen can combine to form either water or hydrogen peroxide.
What type of chemical reaction is 2H2O → 2H2 O2?
The chemical reaction 2H2+O2→2H2O 2 H 2 + O 2 → 2 H 2 O is classified as a synthesis reaction.
What is the product of the reaction of H₂o₂ hydrogen peroxide with HOCl?
This finding is explained by the consumption reaction between H 2 O 2 and HOCl (H 2 O 2 + HOCl → 1 O 2 + H 2 O + H + + Cl − ) [75, 76].What type of reaction is CaCO3 CaO CO2?
(a) Decomposition: A substance breaks down to smaller species. [e.g. CaCO3(s) → CaO(s) + CO2(g) Decomposition of limestone to calcium oxide (quicklime) and carbon dioxide on heating.]
What is the product of the reaction of H2O with Cl2?
1H2O + Cl2 → HCl + HClO2Cl2 + HOH → HCl + HClO3Cl2 + HOH → H2O + HCl + HClO
How does H2O2 react with PbS?
Lead sulfide (PbS) reacts with hydrogen peroxide ( H2O2) and produce lead sulfate (PbSO4) and water as products. This reaction is a redox reaction. … PbS is a black precipitate and PbSO4 is a white precipitate.
Is h2o2 a reactant or product in this equation?
It is a chemical reaction because it is not possible to rearrange water and oxygen tor form hydrogen peroxide after they are split apart. Hydrogen peroxide is the reactant, and oxygen and hydrogen are the products.Which oxide gives H2O2 on treatment with dil h2so4?
The oxide that gi Answer : Barium peroxide (BaO2) as we can get from its name, has a per-oxy linkage in it. Due to which its treatment with H2SO4 gives H2O2 .
What is h2o2 in chemistry?hydrogen peroxide, (H2O2), a colourless liquid usually produced as aqueous solutions of various strengths, used principally for bleaching cotton and other textiles and wood pulp, in the manufacture of other chemicals, as a rocket propellant, and for cosmetic and medicinal purposes.
Article first time published onWhat type of reaction is 2agcl 2ag Cl2?
Therefore, this reaction is a photo-decomposition reaction.
What type of chemical equation is the following 2NaCl → 2Na Cl2?
An example of a synthesis reaction is the combination of sodium (Na) and chlorine (Cl) to produce sodium chloride (NaCl). This reaction is represented by the chemical equation: 2Na + Cl2 → 2NaCl.
What type of reaction is 2Na Cl2 2NaCl?
The reaction 2Na + Cl2 = 2NaCl is an example of Combination reaction. Decomposition reaction will occur when one reactant breaks down into two or more products, e.g. AB => A + B. In Combination Reaction, two or more elements/compounds combine to form a single compound, e.g. A + B => AB.
What are the products of CaCO3?
Calcium carbonate decomposes to form carbon dioxide and lime, an important material in making steel, glass, and paper.
When CaCO3 decomposes the products are?
When calcium carbonate is heated it decomposes into calcium oxide and carbon dioxide.
How does H2O2 react with FeSO4?
Fe 2+ + H2O2 → Fe 3+ + OH – + . OH. … adding the iron catalyst (as a solution of FeSO4); and. adding slowly the H2O2. If the pH is too high, the iron precipitates as Fe(OH)3 and catalytically decomposes the H2O2 to oxygen — potentially creating a hazardous situation.
What is the Colour of product when PbS and H2O2 reacted?
So, after getting the product from the reaction we can say that the on reacting hydrogen peroxide with lead sulphide we get the a white product of the lead sulphate.
What happens when H2O2 reacts with acidified FeSO4?
2FeSO4 + H2SO4 + H2O2 → Fe2(SO4)3 + 2H2O(l), have hydrogen peroxide acts as oxidizing agent.
How do you make Cl2 HCl?
Cl2 can be prepared from HCl by Deacon’s process. The chemical reaction is 4HCl + O2 = 2Cl2 + 2H2O. On the other hand, the HCl can be prepared from the Cl2 by treating it with the water.
What is the formula for a chlorine molecule?
Formula and structure: the chemical structure of chlorine gas is Cl2 and its molecular weight is 70 g/mol. Its structure is Cl-Cl, which is also called the element form of the chlorine element, consist in 2 atoms of chlorine joined by a covalent bond.
Which substance produces H2O2 on reaction with h2so4?
Hence, only sodium peroxide (Na2O2 ) will give H2O2.
Which of the following gives H2O2 on treatment?
From the reactions, we can conclude that the sodium peroxide, and barium peroxide forms a hydrogen peroxide; but sodium peroxide reacts with the water to form hydrogen peroxide.
Which compound gives hydrogen peroxide on electrolysis?
Answer: Hydrogen peroxide can be produced by electrolysis of sulphuric acid.
What are the reactants and the products in the reaction?
The substance(s) to the left of the arrow in a chemical equation are called reactants. A reactant is a substance that is present at the start of a chemical reaction. The substance(s) to the right of the arrow are called products . A product is a substance that is present at the end of a chemical reaction.
What would be product formed by the decomposition reaction of H2O2?
Hydrogen peroxide, H2O2 (very slowly) decomposes naturally into water and oxygen gas.
Is the reaction of Na and Cl2 exothermic?
When a sodium atom transfers an electron to a chlorine atom, forming a sodium cation (Na+) and a chloride anion (Cl-), both ions have complete valence shells, and are energetically more stable. … The reaction is extremely exothermic, producing a bright yellow light and a great deal of heat energy.
What do you see when sodium reacts with chlorine?
Sodium and chlorine react vigorously when heated, giving an orange flame and clouds of white sodium chloride.
What is the chemical name of 2H2O?
Heavy water (2H2O) The number 2 indicates that there are two atoms of hydrogen in a molecule of water. There is also one atom of oxygen but the number one is omitted from a chemical formula. Water molecule (formally known as dihydrogen monoxide) is composed of two hydrogen atoms and one oxygen atom.
What does 3% H2O2 mean?
When you use peroxide, go for medical-grade peroxide, which is 3% strength. That means it’s 97% water and 3% peroxide. “Medical grade is strong enough for household use,” says Dr. Beers. You can also buy a more concentrated form of hydrogen peroxide, called food-grade peroxide, with strength as high as 35%.
Is 2agcl 2ag cl2 an endothermic reaction?
Answer: endothermic reaction it is.
Which type of reaction is 2agbr 2ag br2?
It is photo decomposition reaction.