What is the pOH?
 William Burgess
William Burgess .
Hereof, how do you calculate pOH?
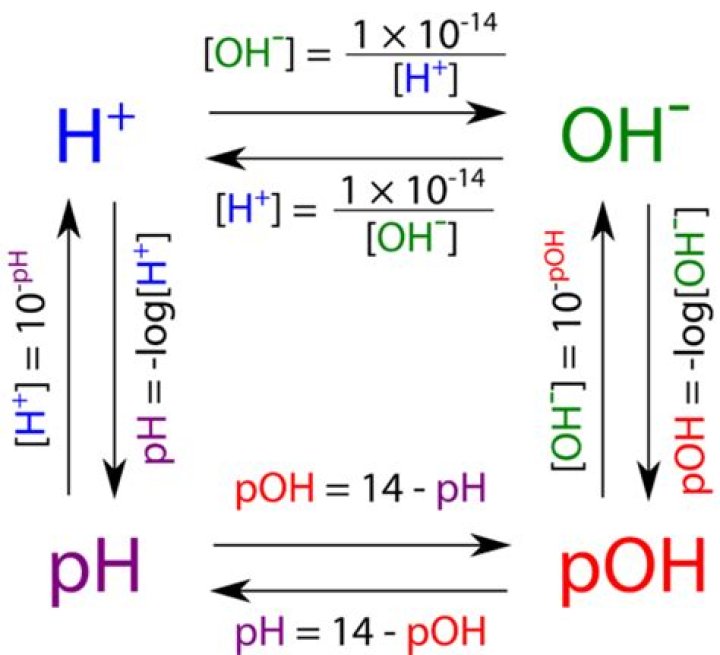
To find the pOH, simply subtract the pH from 14. In order to calculate the pOH, take the negative log of the hydroxide ion concentration. To find the pH, simply subtract pOH from 14.
Also Know, what is the difference between pH and pOH? Re: pH and pOH difference So pH is the measure of hydrogen ion concentration, [H+], while pOH is a measure of the hydroxide ion concentration, [OH-]. The scales for both are 1-14. However in most cases, the question will ask for pH. So remember that pH+pOH=14.
Thereof, what is the pOH of a solution?
The pOH Concept. As with the hydrogen-ion concentration, the concentration of the hydroxide ion can be expressed logarithmically by the pOH. The pOH of a solution is the negative logarithm of the hydroxide-ion concentration. A basic solution has a pOH less than 7, while an acidic solution has a pOH of greater than 7.
What is the relationship between pOH and pH?
pH and pOH denote the negative log of the concentration of hydrogen or hydroxide ions. High pH means that a solution is basic while high pOH means that a solution is acidic. Neutral solutions have pH and pOH of 7.
Related Question AnswersWhat does pOH stand for?
POH| Acronym | Definition |
|---|---|
| POH | Pull Out of Hole (oil drilling industry) |
| POH | Potential of Hydroxide |
| POH | Pat on Head |
| POH | Past Obstetric History |
What is pOH equal to?
pOH is a measure of hydroxide ion (OH-) concentration. It is used to express the alkalinity of a solution. Aqueous solutions at 25 degrees Celcius with pOH less than 7 are alkaline, pOH greater than 7 are acidic and pOH equal to 7 are neutral.What is pOH value?
Updated October 25, 2019. pOH is a measure of hydroxide ion (OH-) concentration. It is used to express the alkalinity of a solution. Aqueous solutions at 25 degrees Celcius with pOH less than 7 are alkaline, pOH greater than 7 are acidic and pOH equal to 7 are neutral.What is the formula for the pH?
To calculate the pH of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The pH is then calculated using the expression: pH = - log [H3O+].Can pH be negative?
It's definitely possible to calculate a negative pH value. In practice, any acid that yields a concentration of hydrogen ions with a molarity greater than 1 will be calculated to have a negative pH. For example, the pH of 12M HCl (hydrochloric acid) is calculated to be -log(12) = -1.08.What is meant by pOH?
Chemistry Terminology: Definition of pOH D. Updated October 25, 2019. pOH is a measure of hydroxide ion (OH-) concentration. It is used to express the alkalinity of a solution. Aqueous solutions at 25 degrees Celcius with pOH less than 7 are alkaline, pOH greater than 7 are acidic and pOH equal to 7 are neutral.Why does pH and pOH equal 14?
Correct answer: Explanation: pH and pOH are the log concentrations of protons and hydroxide ions, respectively. The sum of pH and pOH is always 14. This is because the product of proton concentration and hydroxide concentration must always equal the equilibrium constant for the ionization of water, which is equal to .How is pOH calculated?
The calculation for pOH follows the same rules as for pH, but uses the concentration of OH- ions: pOH = - log(OH-). For example, find the pOH of a 0.02 M solution of sodium hydroxide (KOH). The concentration of OH- ions is 0.02 moles per liter. pOH = - log(.What is meant by pH value?
pH is a measure of hydrogen ion concentration, a measure of the acidity or alkalinity of a solution. The pH scale usually ranges from 0 to 14. Aqueous solutions at 25°C with a pH less than 7 are acidic, while those with a pH greater than 7 are basic or alkaline.Is HCl a strong acid?
Why is hydrochloric acid known as the strong acid? It has a very low pH, extremely acidic, higher concentration of H+ ions than OH- ions and its extent of ionisation/dissociation of H+ ions when dissolved in acid is high. HCl completely dissociates. So HCl a strong acid.How do you find the pH of HCl?
Since HCl is a strong acid, it completely ionizes, and the pH of HCl in solution can be found from the concentration (molarity) of the H+ ions, by definition equal to 0.100 M. (The conjugate base of the acid, which is the chloride ion Cl–, would also have a concentration of 0.100 M.) The pH is thus –log(0.100) = 1.000.What is pH and pOH scale?
pH + pOH = 14. The pOH scale is similar to the pH scale in that a pOH of 7 is indicative of a neutral solution. A basic solution has a pOH of less than 7, while an acidic solution has a pOH of greater than 7. The pOH is convenient to use when finding the hydroxide ion concentration from a solution with a known pH.What is pH and pOH of a solution?
pH and pOH denote the negative log of the concentration of hydrogen or hydroxide ions. High pH means that a solution is basic while high pOH means that a solution is acidic. So here the basic definition of pH is that it's equal to negative log base 10 of the concentration of protons in your solution.What is the pH full form?
PH stands for Potential of Hydrogen. It refers to the hydrogen ion concentration in a solution. It is the measure of the acidity or alkalinity of a solution. The PH value ranges from 0 to 14 on a pH scale.What is pOH mean?
POH| Acronym | Definition |
|---|---|
| POH | Pull Out of Hole (oil drilling industry) |
| POH | Potential of Hydroxide |
| POH | Pat on Head |
| POH | Past Obstetric History |





