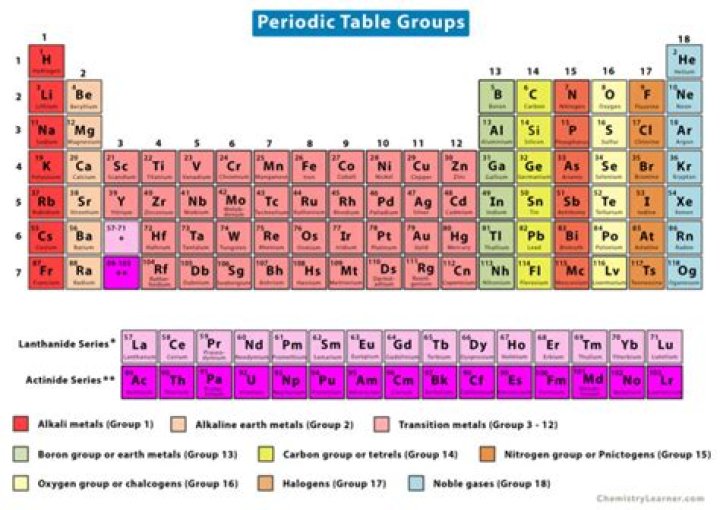
What is the name of Group 11 on the periodic table
 Christopher Martinez
Christopher Martinez The “coinage metals”, copper, silver, and gold, have held great importance in societies throughout history, both symbolically and practically.
What is Group 11 on the periodic table called?
They are also known as the “noble metals.” The name “coinage metals” is not recognized by the International Union of Pure and Applied Chemistry (IUPAC), and can be somewhat misleading as further elements are added to the table. …
What element is Group 11 Period 5?
silver (Ag), chemical element, a white lustrous metal valued for its decorative beauty and electrical conductivity. Silver is located in Group 11 (Ib) and Period 5 of the periodic table, between copper (Period 4) and gold (Period 6), and its physical and chemical properties are intermediate between those two metals.
What is 11 on the periodic table?
Sodium – Element information, properties and uses | Periodic Table.What is Group 11 period 4 on the periodic table?
Copper is in period 4, group 11 | Element project, Periodic table, Copper.
What is RG in periodic table?
roentgenium (Rg), artificially produced transuranium element of atomic number 111.
Are Group 11 metals reactive?
The “coinage metals”, copper, silver, and gold, have held great importance in societies throughout history, both symbolically and practically. Although the most important oxidation state for group 11 is +1, the elements are relatively unreactive, with reactivity decreasing from Cu to Au. …
How can atomic number 11 become stable?
Answer: It can become stable by losing one electron.Is sodium atomic number 11?
Sodium is a chemical element with symbol Na and atomic number 11. Classified as an alkali metal, Sodium is a solid at room temperature.
Is Cl A metal?Chlorine is a non-metal. … The element chlorine is a yellowish-green gas which is extremely reactive and react with various metals.
Article first time published onWhat element is in period 5 Group 11 1b?
Silver is located in Period 5 and Group 11 (Ib) of the periodic table. Silver (Ag), chemical element, a white, shiny metal accounted for its decorative appeal and electrical conductivity.
What element is in period 4 Group 18?
PeriodGroup3Na 11Ar 184K 19Kr 365Rb 37Xe 546Cs 55Rn 86
Which element is in period 4 Group 16?
Selenium. Selenium (Se) is an element in group 16. Selenium is the first nonmetal in period 4, with properties similar to sulfur. Selenium is quite rare in pure form in nature, mostly being found in minerals such as pyrite, and even then it is quite rare.
What element has 12 neutrons metallic and has 11 electrons?
Sodium has a mass number of 23 and an atomic number of 11. It contains 11 protons, 11 electrons, and 12 neutrons.
What is Group 12 on the periodic table called?
zinc group element, any of the four chemical elements that constitute Group 12 (IIb) of the periodic table—namely, zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn).
What is the name of the element in Group 3 Period 4?
“The Element Phosphorus.” It’s Elemental -. N.p., n.d. Web.
Is group 11 a transition metal?
A Group 11 element is one in the series of elements in group 11 (IUPAC style) in the periodic table, consisting of transition metals which are the traditional coinage metals of copper (Cu), silver (Ag), and gold (Au).
How did gold get its name?
Discovery dateapprox 3000BCDiscovered by-Origin of the nameThe name is the Anglo-Saxon word for the metal and the symbol comes from the Latin ‘aurum’, gold.
What is Group 10 on the periodic table called?
Group 10: Transition Metals.
What does RG look like?
RoentgeniumAppearancesilvery (predicted)Mass number[282] (unconfirmed: 286)Roentgenium in the periodic table
What is the group number of Roentgenium?
Group11Melting pointPeriod7Boiling pointBlockdDensity (g cm−3)Atomic number111Relative atomic massState at 20°CSolidKey isotopes
When was Unununium renamed?
Even though Unununium is a fantastic name, in 2004 it was renamed Roentgenium (Rg) to honor the German physicist Wilhelm Conrad Röntgen, who discovered X-rays.
What element atomic number is 11?
Atomic NumberSymbolName11NaSodium12MgMagnesium13AlAluminum14SiSilicon
What is the group name of sodium?
Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
What does 11 mean in sodium?
Mass number We know that the atomic number of sodium is 11. This tells us that sodium has 11 protons and because it is neutral it has 11 electrons. The mass number of an element tells us the number of protons AND neutrons in an atom (the two particles that have a measurable mass).
Which atom is larger Na or Cl?
Na atoms are larger than Cl atoms because the electron cloud of Cl is pulled in more tightly by the increased number of protons in the nucleus. Na+ ions are smaller than Cl- ions because the Na+ ions have electrons in only two energy levels and Cl- ions have electrons in three levels.
What family is chlorine in?
Chlorine is in group 17 of periodic table, also called the halogens, and is not found as the element in nature – only as a compound. The most common of these being salt, or sodium chloride, and the potassium compounds sylvite (or potassium chloride) and carnallite (potassium magnesium chloride hexahydrate).
Is chlorine a halogen?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
What is pool chlorine?
Chlorine is added to the water to kill germs. When it is added to a swimming pool, it forms a weak acid called hypochlorous acid that kills bacteria like salmonella and E. coli, as well as germs that cause viruses such as diarrhea and swimmer’s ear. How is pool water chlorinated?
What element is in period 4 Group 11 1b?
Molybdenum is an important trace mineral for the body and is even considered essential to life.
Which element is in period 3 Group 17?
Chlorine is a halogen in group 17 and period 3. It is very reactive and is widely used for many purposes, such as as a disinfectant. Due to its high reactivity, it is commonly found in nature bonded to many different elements.