It exists in crystalline form with 10 water molecules. This turns it into the familiar washing soda. = 286 u. Therefore the molar mass of Na2CO3•10H2O is 286g..
In this way, what is the molecular mass of Na2CO3 10h2o?
Answer and Explanation: The molar mass of Na2 CO3 • 10H2 O is 286.1412 g/mol.
Beside above, what is the formula of sodium carbonate decahydrate? Sodium Carbonate Decahydrate Na2CO3. 10H2O Molecular Weight -- EndMemo.
Keeping this in view, what is the molar mass of sodium carbonate decahydrate?
Sodium carbonate decahydrate
| PubChem CID: | 151402 |
| Molecular Formula: | CH20Na2O13 |
| Synonyms: | Sodium carbonate decahydrate 6132-02-1 Carbonic acid disodium salt, decahydrate UNII-LS505BG22I LS505BG22I More |
| Molecular Weight: | 286.14 g/mol |
| Dates: | Modify: 2020-01-11 Create: 2005-06-24 |
What is 10h2o?
10H2O signifies that during crystallization of sodium carbonate 10 molecules of water is being incororated per one molecule of sodium carbonate. This is referred as water of crystallization . It doesnt make it wet but make it crystalline.
Related Question Answers
What is Na2CO3 10h2o called?
The common name of Na2CO3. 10H2O is washing soda. The recrystallization of sodium carbonate from water produces washing soda (Na2CO3. It contains 10 molecules of water of crystallisation and is also known as sodium carbonate decahydrate.How many moles are in Na2CO3?
1 moles
How do I calculate molecular mass?
Key Points - The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol).
- The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms.
Is the molecule of Na2CO3 10h2o wet?
In general, it is pure white dry solid particles having 10 water molecules present as a single unit with sodium carbonate and it is also denoted as Na2CO3. Even the molecule of na2co3.10h2o is really wet because it consists of 10 molecules of water in it so it is wet.What is the common name for Na2CO3?
Sodium carbonate
What is the chemical name of washing soda?
Sodium carbonate
What is sodium carbonate used for?
Uses: The main uses of sodium carbonate are as water softener, food processing aid, pH modifier, swimming pool chemical and electrolyte. It is also used in the manufacture of glass, paper, soaps and detergents, and many other useful chemicals.What is the Colour of hydrated sodium carbonate?
At room temperature, sodium carbonate (Na2CO3) is an odourless, greyish white powder which is hygroscopic. This means when it is exposed to air, it can spontaneously absorb water molecules.What is the solubility of sodium carbonate?
Water
At what temperature does sodium carbonate decompose?
Thermal decomposition of Sodium Carbonate. Does Sodium Carbonate (Na2CO3) decompose completely at >900ºC (1000ºC or 1100ºC) to Sodium Oxide?What is the difference between washing soda and borax?
Borax is a softer molecule with a lower pH, so it is able to dissolve much more easily than washing soda. This means that it is able to work for longer and is less likely to damage your clothes in the wash due to these softer molecules. Washing soda on the other hand, doesn't dissolve as easily and may leave a residue.How much does one sucrose molecule weigh in grams?
Answer and Explanation: One sucrose molecule weighs about 5.679 x 10-22 grams.What is the mass of water?
Mass of one mole of water = 2 g/mol + 16 g/mol = 18 g/mol.What is the chemical name of Na2CO3 10h2o?
Chemical name for Na2CO3. 10H2O is Washing Soda ( Sodium carbonate decahydrate ) and for Na2CO3 is Sodium Carbonate.How do you convert from moles to grams?
Moles to Grams Conversion Formula. In order to convert the moles of a substance to grams, you will need to multiply the mole value of the substance by its molar mass. More commonly written for this application as: where, is the molar mass of the substance.Is sodium carbonate a strong or weak base?
Description: Sodium Carbonate is the disodium salt of carbonic acid with alkalinizing property. When dissolved in water, sodium carbonate forms carbonic acid and sodium hydroxide. As a strong base, sodium hydroxide neutralizes gastric acid thereby acting as an antacid.Is Na2CO3 an acid or base?
Na2CO3 is neither an acid nor a base. It is a salt. A salt in chemistry is formed by the reaction of an acid and a base. Now depending on the strength of the respective acid and base from which the salt is derived, it may be categorised as an acidic, basic or a neutral salt.Is caco3 soluble in water?
Calcium carbonate has a very low solubility in pure water (15 mg/L at 25°C), but in rainwater saturated with carbon dioxide, its solubility increases due to the formation of more soluble calcium bicarbonate. Calcium carbonate is unusual in that its solubility increases as the temperature of the water decreases.What is the pH of sodium carbonate?
The PH value of Common Acids and Bases: Sodium carbonate, also known as washing soda, is a common ingredient in laundry detergents. When dissolved in water, it tends to form solutions with pH values between 11 and 12. 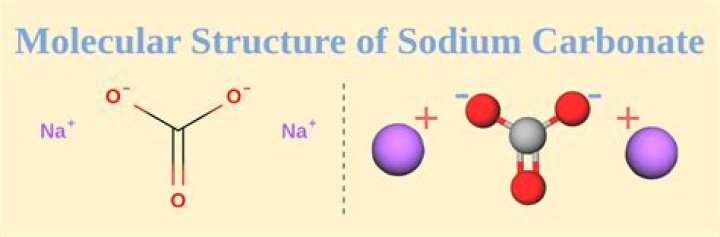
 Christopher Lucas
Christopher Lucas