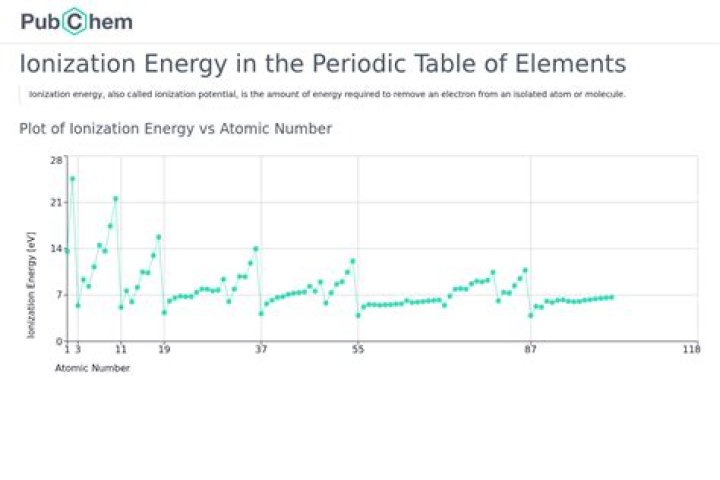
What is the ionization energy on the periodic table
 William Burgess
William Burgess Ionization energy, also called ionization potential, is a property that all elements on the periodic table have. … It is the amount of energy required to remove an electron from a neutral atom, which forms an ion. It is usually measured in kJ/mol, and the measurement is based on an isolated atom in its gaseous phase.
What is the ionisation energy of an element?
The ionization energy of a chemical species (i.e., an atom or molecule) is the energy required to remove electrons from gaseous atoms or ions. This property is also referred to as the ionization potentia and is measured in volts.
How do you find the ionization energy of a photoelectron?
PES involves a given energy of photon to ionize a molecule. As the excess energy, will be in the form of kinetic energy, is calculated by the photoelectron spectrometer it is possible to calculate ionization energy of a molecule, by rearranging the following equation: Ek=hν−EI, to solve for EI, ionization energy.
Why does ionization energy increase from left to right on the periodic table?
On the periodic table, first ionization energy generally increases as you move left to right across a period. This is due to increasing nuclear charge, which results in the outermost electron being more strongly bound to the nucleus.What is sodium ionization energy?
This energy is usually expressed in kJ/mol, or the amount of energy it takes for all the atoms in a mole to lose one electron each. … For instance, the ionization energy of Sodium (alkali metal) is 496KJ/mol (1) whereas Chlorine’s first ionization energy is 1251.1 KJ/mol (2).
Is second ionization energy?
An element’s second ionization energy is the energy required to remove the outermost, or least bound, electron from a 1+ ion of the element. Because positive charge binds electrons more strongly, the second ionization energy of an element is always higher than the first.
What is the first ionization energy?
By definition, the first ionization energy of an element is the energy needed to remove the outermost, or highest energy, electron from a neutral atom in the gas phase.
Why does ionization energy decrease?
On the periodic table, first ionization energy generally decreases as you move down a group. This is because the outermost electron is, on average, farther from the nucleus, meaning it is held less tightly and requires less energy to remove.What increases ionization energy?
Ionization energy (IE) is the energy required to remove the highest-energy electron from a neutral atom. In general, ionization energy increases across a period and decreases down a group. … Down a group, the number of energy levels (n) increase and the distance is greater between the nucleus and highest-energy electron.
Why does ionization energy happen?Ionization is the process by which ions are formed by gain or loss of an electron from an atom or molecule. If an atom or molecule gains an electron, it becomes negatively charged (an anion), and if it loses an electron, it becomes positively charged (a cation). Energy may be lost or gained in the formation of an ion.
Article first time published onWhat is ionisation energy Class 11 chemistry?
Periodic Properties of Class 11. The amount of energy required to remove the most loosely bound electron of the outermost shell (i.e. the outermost electron) from one mole of an isolated gaseous atom of an element in its ground state to produce a cation is known as ionization energy of that element.
How do you find the ionization energy of sodium?
- wavelength λ=242nm=2. 42×10−7m.
- Energy (E)=hc/λ
- =0. 0821×10−17 J / atom.
- 0821×10−17 J energy is sufficient for ionisation of one Na atom , so it is the ionisation energy of Na .
- hence, I. E=0. 0821×10−17 J/atom.
- =0. 0821×10−17×6. 022×1022 J/mol.
How do you find the ionization energy of aluminum?
- Electron Affinity of Aluminum is 42.5 kJ/mol.
- Electronegativity of Aluminum is 1.61.
- First Ionization Energy of Aluminum is 5.9858 eV.
- X + e– → X– + energy Affinity = – ∆H.
- Affinities of Nonmetals vs.
What is hydrogen ionization energy?
For a hydrogen atom, composed of an orbiting electron bound to a nucleus of one proton, an ionization energy of 2.18 × 10−18 joule (13.6 electron volts) is required to force the electron from its lowest energy level entirely out of the atom. …
What is ionization energy of oxygen?
First Ionization Energy of Oxygen is 13.6181 eV.
What is the ionization energy of carbon?
First Ionization Energy of Carbon is 11.2603 eV.
What is 3rd ionization energy?
The third ionization energy is the energy it takes to remove an electron from a 2+ ion. (That means that the atom has already lost two electrons, you are now removing the third.) And 2nd ionization energy is higher than 1st ionization energy, 3rd is higher than 2nd, and so forth.
What is ionization energy MCAT?
What is ionization energy? Ken Tao is an MCAT expert and explains that it’s the amount of energy required to remove an electron from an atom or ion. Ionization energy increases from left to right across the periodic table and decreases going down the periodic table.
What has the highest ionization energy?
Thus, helium has the largest first ionization energy, while francium has one of the lowest.
What is ionization energy How does it vary in period of the periodic table?
The ionization energy of an element increases as one moves across a period in the periodic table because the electrons are held tighter by the higher effective nuclear charge.
Which group has the highest ionization energy?
Noble Gases have a very stable electron configuration, therefore, they have the highest amount of ionization energy within their periods.
What is low ionization energy?
Definition. Ionization energy is the minimum energy necessary to remove one mole of electrons from one mole of atoms in the gaseous state. … If the ionization energy is low, that means that it takes only a small amount of energy to remove the outermost electron.
How does ionization happen?
Ionization, in general, occurs whenever sufficiently energetic charged particles or radiant energy travel through gases, liquids, or solids. … Pulses of radiant energy, such as X-ray and gamma-ray photons, can eject electrons from atoms by the photoelectric effect to cause ionization.
How do you find the ionization energy of hydrogen?
For hydrogen, first orbit energy is –2.18 × 10– 18 J/atom (or – 1312.3 KJ/mole), and the ionization energy is + 2.18 × 10–18 J/atom (or + 1312.3 KJ/mole).
What is ionization enthalpy Class 12?
Ans. Ionization Enthalpy is defined as the amount of energy an isolated gaseous atom would take to lose an electron in its ground state. Whenever an electron is expelled from an atom, it takes a specific amount of energy to expel it, hence the enthalpies of chemical elements for ionisation are always optimistic.