What is the heat capacity of glass
 Rachel Young
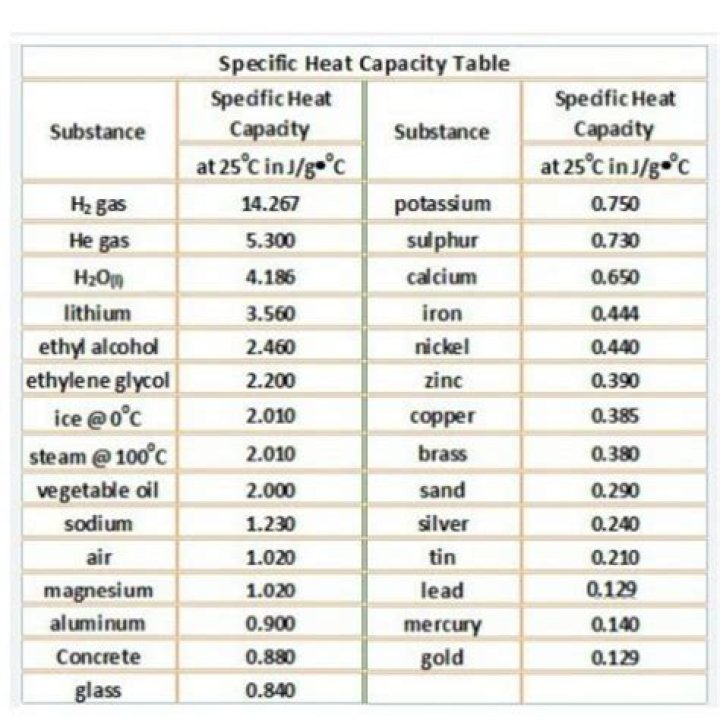
Rachel Young Specific heat capacity is expressed with units J kg-1 °C-1 or J kg-1 °K-1. The specific heat capacity of a type of glass is 840 J kg-1 °C-1. … Some substances have low specific heat capacities while some have higher specific heat capacities.
What is the specific heat capacity of glass J kg C?
MaterialJ/792792Gold130130
What is the thermal property of glass?
Thermal PropertyCommon Values of Borosilicate GlassLinear Thermal Expansionα = 30 – 60 x 10-7/°CThermal ConductivityK = 1 Watt/m°CSpecific HeatC = 800 J/kg°C
Does glass have a low heat capacity?
However, at lower temperatures, glass has a much higher heat capacity than crystals. … In the late 1950’s, scientists found that at temperatures of around 10 K (-263°C) glasses need much more heat to warm up than crystals.Is R The heat capacity?
The heat capacity at constant volume, Cv, is the derivative of the internal energy with respect to the temperature, so for our monoatomic gas, Cv = 3/2 R. The heat capacity at constant pressure can be estimated because the difference between the molar Cp and Cv is R; Cp – Cv = R.
What is the specific heat capacity if it takes 1000 J of energy to heat 25 g of this substance by 100 C give your answer in J kg C?
What is the specific heat capacity if it takes 1000 J of energy to heat 25 g of this substance by 100°C? Give your answer in J/kg°C. c = Q m Δ T = 1000 J 0.025 k g × 100 ∘ C = 400 J / ( k g ∘ C ) .
How do you find heat capacity?
To calculate heat capacity, use the formula: heat capacity = E / T, where E is the amount of heat energy supplied and T is the change in temperature. For example, if it takes 2,000 Joules of energy to heat up a block 5 degrees Celsius, the formula would look like: heat capacity = 2,000 Joules / 5 C.
Is glass a good conductor of heat?
Glass is a good conductor of heat when it is found in fiberglass. Glass allows radiant heat to pass through freely. The reason that glass is such a valuable material is that it exhibits a very low absorption of electromagnetic radiation in the visible range.Is glass affected by heat?
When Can Glass Shatter from Heat? Glass is a durable material that stands up well to summer heat. … However, Glass can suffer a thermal break when the temperature gets too hot. Thermal breaks tend to occur when the glass expands and contracts due to temperature differences.
What is glass thermal conductivity?Glass is made of sand and other minerals that are melted together at very high temperatures to form a material that is ideal for a wide range of uses. Since it is an amorphous solid material, it has not high thermal conductivity. Its thermal conductivity is about k = 1 W/m.K.
Article first time published onDoes glass have a high specific heat capacity?
The specific heat capacity of a typical sample of glass is 0.837 J⋅K−1g−1 . In other words, it takes twice as much heat to raise the temperature of 1 g of wood by 1 K as it does to raise the temperature of 1 g of glass. … Glass is tightly-packed with semiconductive properties, transferring heat well throughout the glass.
What is the thermal expansion of glass?
The glass possesses a low thermal expansion coefficient (6.7×10-7/K) and is thermally stable to around 1000oC.
How do you calculate thermal conductivity of glass?
Glass componentbi valueMgO0.0137CaO0.0123SrO0.0084BaO0.0024
What is water's heat capacity?
One of water’s most significant properties is that it takes a lot of energy to heat it. Precisely, water has to absorb 4,184 Joules of heat (1 calorie) for the temperature of one kilogram of water to increase 1°C. For comparison sake, it only takes 385 Joules of heat to raise 1 kilogram of copper 1°C.
What is the heat capacity of 175 g of liquid water?
The heat capacity of 175 g of liquid water is 732.55 J/°C.
What is the molar heat capacity of gold?
Substancecp in J/g KMolar cp J/mol KBismuth0.12325.7Copper0.38624.5Brass0.380-Gold0.12625.6
What is an example of heat capacity?
The specific heat capacity of a substance is the amount of energy required to raise the temperature of 1 kg of the substance by 1°C. Example: A 250g copper pipe is heated from 10°C to 31°C.
What is heat capacity explain?
heat capacity, ratio of heat absorbed by a material to the temperature change. It is usually expressed as calories per degree in terms of the actual amount of material being considered, most commonly a mole (the molecular weight in grams). The heat capacity in calories per gram is called specific heat.
What is Q heat chemistry?
q = Quantity of Heat. m = Mass of Substance. ΔT = Change in Temperature. Cp = Specific/Molar Heat Capacity.
How do you calculate heat in joules?
Multiply the change in temperature by the specific heat capacity and the mass of your object. This will give you the heat lost or gained in joules. Example: If 10 kilograms of water are heated from 10 degrees Celsius to 50 degrees Celsius, how much energy (in joules) did they absorb?
What is the specific heat capacity of water in J kg K?
The SI unit of specific heat capacity is joule per kelvin per kilogram, J⋅kg−1⋅K−1. For example, the heat required to raise the temperature of 1 kg of water by 1 K is 4184 joules, so the specific heat capacity of water is 4184 J⋅kg−1⋅K−1.
What is specific heat capacity GCSE?
The specific heat capacity of a material is the energy required to raise one kilogram (kg) of the material by one degree Celsius (°C). … This means that it takes 4,200 J to raise the temperature of one kg of water by 1 °C.
Can heat crack glass doors?
Can tempered glass doors break from heat? If the temperature exceeds greater than 300°F, then the glass can spontaneously shatter. … Pre-existing mechanical defects can magnify and push the breakage when constantly exposed to high or fluctuating temperatures since the glass will constantly expand and cool.
Can mirrors withstand heat?
What temperature can a mirror tolerate? Under normal conditions our mirrors can withstand temperatures of minus 15 to plus 80 (dependent on material). At higher temperatures, we recommend Stainless Steel, this mirror remains usable by higher temperatures.
Can sunlight crack glass?
If the glass experiences enough stress, it will crack. Sunlight can also cause stress cracks in a similar way. … When the metal edges of your windshield heat up faster than the middle of the glass, this can cause stress that leads to cracking.
How is glass conductive?
Glass generally does not conduct electricity well, but special coatings designed to conduct electricity can be added in post-production that allow glass to do just that.
Why glass is heat insulator?
Glass is actually an insulator. … Insulators have electrons that are held tightly which means they aren’t shared between other atoms. Glass resists heat and electricity from passing through it. Glass, wood and plastic are all excellent insulators, but not good conductors.
Is glass a heat sink?
The “heat sink” that sits on top of your CPU just moves the heat from the chip out to the fins, where the breeze from the fan can carry it away. So now to your question: glass is a very poor conductor of heat, so it would do a terrible job of transporting it from the CPU chip to those fins. Yes.
Is glass a heat conductor or insulator?
Glass, for instance, is a very good insulator at room temperature, but becomes a conductor when heated to a very high temperature.
Does glass conduct cold?
The glass you touch is at room temperature, which is lower than your fingers, so it feels cold, even if it is somewhat of an insulator. For comparison, glass has a thermal conductivity of 1.05 W/(w K).
Why glass has high thermal conductivity?
It has one of the lowest possible heat conduction a solid (without air trapped in it) can possibly have, this is mostly due to its lack of ordered crystal structure. Since it’s an insulator, the electronic contribution to the thermal conductivity is very small.