The atoms become ions. forms the ionic bond between lithium and oxygen. The formula for lithium oxide is Li2O..
In this manner, what is the formula of lithium oxide?
Li2O
Furthermore, how do you balance lithium and oxygen? 1 Answer
- in this case Lithium Oxide (which is Li2O )
- Oxygen is a reactant in the form O2.
- Li+O2→Li2O.
- By making 2 lots of Li2O as products, we end up with 2 Oxygen atoms on the right side (the same number as the left side).
- Li+O2→2Li2O.
- 4Li+O2→2Li2O.
Beside above, what is lithium oxygen?
Lithium-oxygen cells are made of two electrodes, an anode and a cathode, separated by a substance called electrolyte. When the battery is powering another device, oxygen molecules on the cathode combine with lithium ions from the electrolyte to form a solid compound called lithium peroxide.
What type of structure is lithium oxide?
Lithium oxide
| Names |
| Refractive index (nD) | 1.644 |
| Structure |
| Crystal structure | Antifluorite (cubic), cF12 |
| Space group | Fm3m, No. 225 |
Related Question Answers
What is lithium oxide used for?
Typical Applications. Lithium oxide is used as a flux in ceramic glazes; and creates blues with copper and pinks with cobalt. Lithium oxide reacts with water and steam, forming lithium hydroxide and should be isolated from them.What is the charge of lithium?
Lithium has 3 electrons, 2 in inner shell and 1 in outer shell. If lithium loses one electron then it is said to have a charge of +1 if 2 electrons lost then the charge would be +2 and if all the electrons are lost then charge would be +3.Is lithium oxide a solid liquid or gas?
Lithium oxide is a white solid also known as lithia, it is produced when lithium metal burns in the presence of oxygen. Oxide compounds are not conductive to electricity.How do you write chemical formulas?
Part 1 Writing Chemical Formulas of Covalent Compounds - Memorize the prefixes for number of atoms.
- Write the chemical symbol for the first element.
- Add the number of atoms as a subscript.
- Write the chemical symbol for the second element.
- Add the number of atoms present as a subscript.
- Practice with some examples.
Is Lithium a metal?
Lithium. Lithium is part of the alkali metal group and can be found in the first column of the periodic table right below hydrogen. Like all alkali metals it has a single valence electron that it readily gives up to form a cation or compound. At room temperature lithium is a soft metal that is silvery-white in color.What holds ions together?
Ionic Bonding. An ionic bond is held together by the electrostatic attraction between ions that are near one another. Electrostatic attraction is the attraction between atoms that have opposite charge and holds the atoms together in ionic bonds.What is the chemical name of Li2O?
Lithium oxide
| PubChem CID: | 166630 |
| Structure: | Find Similar Structures |
| Molecular Formula: | Li2O |
| Synonyms: | Lithium oxide 12057-24-8 Lithium oxide anhydrous 12142-77-7 Lithium oxide (LiO) More |
| Molecular Weight: | 29.9 g/mol |
Is lithium oxide a covalent bond?
Lithium oxide is an ionic compound formed between a metal (Li) and a non- metal (O) by the complete transfer of electrons from Li to O to give Li+ cations and O2– anions. These ions are held in place in the crystal lattice by strong electrostatic attractions between the positively and negatively charged ions.What is the product of lithium and oxygen?
Two lithium atoms will each give one electron to the oxygen atom. The atoms become ions. forms the ionic bond between lithium and oxygen. The formula for lithium oxide is Li2O.What type of reaction is lithium and oxygen?
Lithium burns with a strongly red-tinged flame if heated in air. It reacts with oxygen in the air to give white lithium oxide. With pure oxygen, the flame would simply be more intense. For the record, it also reacts with the nitrogen in the air to give lithium nitride.What happens to lithium in air?
Lithium will ignite and burn in oxygen when exposed to water or water vapors. Lithium is flammable, and it is potentially explosive when exposed to air and especially to water, though less so than the other alkali metals. The metal reacts with hydrogen gas at high temperatures to produce lithium hydride (LiH).How does a lithium air battery work?
The lithium-air battery works by combining lithium ion with oxygen from the air to form lithium oxide at the positive electrode during discharge.What is the pH of lithium?
Lithium hydroxide is normally used for pH control of primary water, the pH being between 6.9 and 7.4 at operating temperature.What does lithium and water make?
Lithium metals reacts slowly with water to form a colourless solution of lithium hydroxide (LiOH) and hydrogen gas (H2). The resulting solution is basic because of the dissolved hydroxide. The reaction is exothermic, but the reaction is slower than that of sodium (immediately below lithium in the periodic table).Is oxygen a compound?
Oxygen in the atmosphere is a molecule because it contains molecular bonds. It is not a compound because it is made from atoms of only one element - oxygen. This type of molecule is called a diatomic molecule, a molecule made from two atoms of the same type.Why is Li2O linear?
The shape of Li2O will be linear. In Valence Shell Electron Pair Repulsion (VSEPR) theory, pairs of electrons that surround the central atom of a molecule or ion are arranged as far apart as possible to minimise electron-electron repulsion.Which is more reactive lithium or sodium?
Within the alkali metals, sodium is more reactive than lithium but less reactive than potassium. Its ionization energy is higher than potassium but lower than lithium. Sodium is also more metallic than lithium.What is the balanced symbol equation for lithium and water?
Answer and Explanation: In this reaction, 2 mole of Li reacts with 2 mole of water to form 2 mole of LiOH and 1 mole hydrogen gas. Therefore, the correct equation is 2Li+2H2O→2LiOH+H2 2 L i + 2 H 2 O → 2 L i O H + H 2 .What is the symbol equation for lithium and water?
Lithium metals reacts slowly with water to form a colourless solution of lithium hydroxide (LiOH) and hydrogen gas (H2). The resulting solution is basic because of the dissolved hydroxide. The reaction is exothermic, but the reaction is slower than that of sodium. 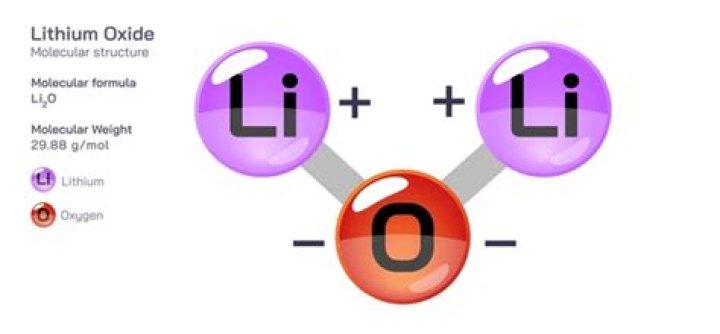
 Andrew White
Andrew White