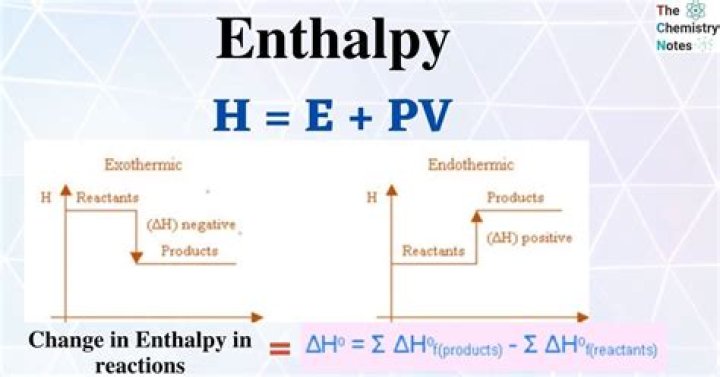
What is the enthalpy at equilibrium
 Emily Sparks
Emily Sparks overall ΔH for equilibrium is zero. the reverse reaction is endothermic.
What is enthalpy in equilibrium?
Enthalpy: The total amount of potential energy stored in a substance. … Entropy increases as a substance goes from solid to liquid to gas. 8. Equilibrium: A system where the rate of forward change is equal to the rate of reverse change. At equilibrium there is no net change.
What is the entropy at equilibrium?
The entropy of an isolated system never decreases: in equilibrium, the entropy stays the same; otherwise the entropy increases until equilibrium is reached.
What happens to enthalpy at equilibrium?
In an equilibrium, the forwards reaction has a negative enthalpy change. If we increase the temperature the equilibrium will shift to the left. The forward reaction is exothermic: it produces heat. … Alternatively, in an equilibrium, the forwards reaction has a positive enthalpy change.What does it mean if enthalpy is 0?
Jul 15, 2017. The CHANGE in enthalpy is zero for isothermal processes consisting of ONLY ideal gases. For ideal gases, enthalpy is a function of only temperature. Isothermal processes are by definition at constant temperature. Thus, in any isothermal process involving only ideal gases, the change in enthalpy is zero.
How do I calculate enthalpy?
Use the formula ∆H = m x s x ∆T to solve. Once you have m, the mass of your reactants, s, the specific heat of your product, and ∆T, the temperature change from your reaction, you are prepared to find the enthalpy of reaction. Simply plug your values into the formula ∆H = m x s x ∆T and multiply to solve.
What do we mean by enthalpy?
enthalpy, the sum of the internal energy and the product of the pressure and volume of a thermodynamic system. … According to the law of energy conservation, the change in internal energy is equal to the heat transferred to, less the work done by, the system.
What is the effect of pressure on equilibrium?
If the pressure is increased, the position of equilibrium moves in the direction of the fewest moles of gas. On the left, there are 3 moles of gas (1 + 2), but on the right there is only 1. Therefore, if the pressure is increased, the position of equilibrium will move to the right and more methanol will be produced.What happens to equilibrium when volume is decreased?
When there is a decrease in volume, the equilibrium will shift to favor the direction that produces fewer moles of gas. When there is an increase in volume, the equilibrium will shift to favor the direction that produces more moles of gas.
Is entropy change 0 at equilibrium?At equilibrium, the total entropy change is zero.
Article first time published onWhy is the change in entropy 0 at equilibrium?
According to the Second Law of Thermodynamics, a change takes place in the direction which corresponds to an increase in Stotal. When the Stotal is zero, there will be no tendency for the reaction to go in either direction; it will be at equilibrium. …
Does entropy change in equilibrium?
At equilibrium, the change in entropy is zero, i.e., ΔS=0 (at equilibrium).
What is the enthalpy of O2?
Species NameFormulaΔfH°(298.15 K)DioxygenO2 (g, singlet)94.383
What is the enthalpy of co2?
The enthalpy of formation of carbon dioxide at 298.15K is ΔHf = -393.5 kJ/mol CO2(g).
What does the h of a reaction represent?
In chemistry, the letter “H” represents the enthalpy of a system. Enthalpy refers to the sum of the internal energy of a system plus the product of the system’s pressure and volume. The delta symbol is used to represent change. Therefore, delta H represents the change in enthalpy of a system in a reaction.
Is enthalpy a constant?
Enthalpy is a state function, and the change in enthalpy of a system is equal to the sum of the change in the internal energy of the system and the PV work done. Enthalpy is a state function whose change indicates the amount of heat transferred from a system to its surroundings or vice versa, at constant pressure.
What is entropy and enthalpy?
Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound. Enthalpy is zero for elemental compounds such hydrogen gas and oxygen gas; therefore, enthalpy is nonzero for water (regardless of phase).
Is enthalpy extensive or intensive?
Intensive properties are properties that do not depend on the quantity of matter. For example, pressure and temperature are intensive properties. Energy, volume and enthalpy are all extensive properties.
What is enthalpy combustion?
The enthalpy of combustion of a substance is defined as the heat energy given out when one mole of a substance burns completely in oxygen.
How do you calculate enthalpy of enthalpy of reaction?
This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. and the standard enthalpy of formation values: ΔH fo[A] = 433 KJ/mol. ΔH fo[B] = -256 KJ/mol.
What does Le Chatelier's principle say?
– [Instructor] Le Chatelier’s principle says, if a stress is applied to a reaction mixture at equilibrium, the net reaction goes in the direction that relieves the stress. Change in the concentration of a reactant or product is one way to place a stress on a reaction at equilibrium.
What affects equilibrium constant?
Changing concentrations Equilibrium constants are not changed if you change the concentrations of things present in the equilibrium. The only thing that changes an equilibrium constant is a change of temperature. The position of equilibrium is changed if you change the concentration of something present in the mixture.
What affects Le Chatelier's principle?
Le Chatelier’s principle can be used to predict the behavior of a system due to changes in pressure, temperature, or concentration. … Increasing the concentration of reactants will drive the reaction to the right, while increasing the concentration of products will drive the reaction to the left.
What happens to equilibrium when concentration is increased?
When the concentration of reactants is increased, the equilibrium shifts to the right and there will be more product than before. There will also be more reactants than before (more reactants were added).
What happens when an inert gas is added to an equilibrium?
On adding inert gas at constant volume the total pressure of the system is increased, but the partial pressure of each reactant and product remains the same. Hence no effect on the state of equilibrium.
Why is Gibbs free energy 0 at equilibrium?
Both the forward and backward reactions occur at the same rate. If the Gibbs energy change for the forward reaction is G, then the Gibbs energy change is -G for the backward reaction. Total Gibbs energy at equilibrium is G+(-G)=0. This is why Gibbs energy is zero at equilibrium.
What is the relationship between K and Delta G?
Both K and ΔG° can be used to predict the ratio of products to reactants at equilibrium for a given reaction. ΔG° is related to K by the equation ΔG°=−RTlnK. If ΔG° < 0, then K > 1, and products are favored over reactants at equilibrium.
What is Gibbs energy class 11?
Gibbs Energy is the maximum (or reversible) work that a thermodynamic system can perform at a constant temperature and pressure. The reversible work in thermodynamics implies a special method in which work is carried out such that the system remains in perfect equilibrium with all its surroundings.
When system is in equilibrium then entropy is maximum?
The point of maximum entropy is the equilibrium, where temperatures are equal and heat flow is zero.
How does enthalpy affect the spontaneity of a reaction?
If a reaction is exothermic ( H is negative) and the entropy S is positive (more disorder), the free energy change is always negative and the reaction is always spontaneous. … If the enthalpy change H and the entropy change S are both positive or both negative, the spontaneity of the reaction depends on the temperature.
What equation relates entropy and enthalpy?
Entropy ( S ) defines the degree of randomness or disorder in a system. where at constant temperature, the change on free energy is defined as: ΔG=ΔH−TΔS . Therefore, the free energy expression provides a relationship between enthalpy and entropy. Thus, for a system at equilibrium, ΔG=0 , and then we find that ΔS=ΔHT .