What is covalent catalysis
 Isabella Bartlett
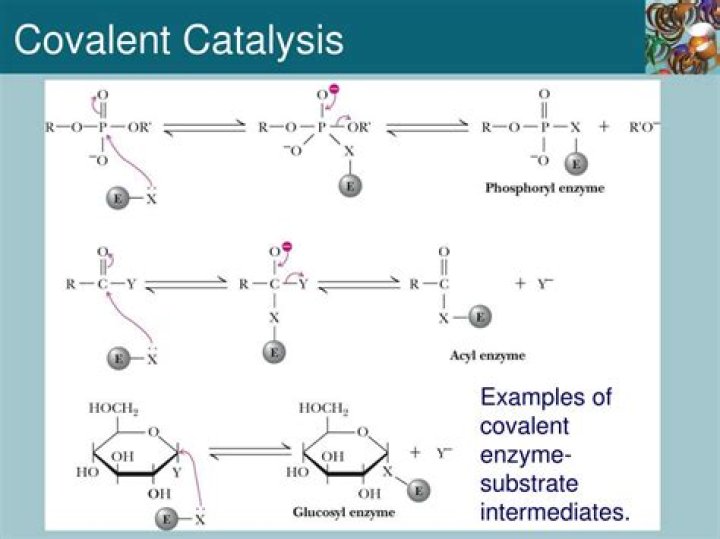
Isabella Bartlett Covalent catalysis involves the formation of a covalent bond between the enzyme and at least one of the substrates involved in the reaction. Often times this involves nucleophilic catalysis which is a subclass of covalent catalysis.
What is meant by covalent catalysis?
Definition. Covalent Catalysis is one of the four strategies that an enzyme will employ to catalyze a specific reaction. Covalent catalysis occurs when the substrate(s) in an enzymatic reaction become temporarily covalently attached to the enzyme during the catalytic reaction.
Which amino acids are involved in covalent catalysis?
This includes methionine, phenylalanine, tyrosine and tryptophan. Chymotrypsin contains a serine amino acid within the active site that plays a major nucleophilic role that catalyzes the hydrolysis of peptide bonds. The active site utilizes covalent catalysis and carries out a two-step process.
What is an example of covalent catalysis?
Another example of covalent catalysis is a diverse group of proteolytic enzymes known as the serine proteases. Their name ‘serine proteases’ comes from the presence of a reactive Ser residue in their active site and this residue takes part in catalytic activity following a common mechanism.Which is not involved in covalent catalysis?
Explanation: Hydrolysis of acetyl imidazole is an example of acid base catalysis, and not covalent catalysis.
What is the mechanism of enzyme catalysis?
An enzyme attracts substrates to its active site, catalyzes the chemical reaction by which products are formed, and then allows the products to dissociate (separate from the enzyme surface). The combination formed by an enzyme and its substrates is called the enzyme–substrate complex.
What do you mean by enzyme catalysis?
Enzyme catalysis is the increase in the rate of a process by a biological molecule, an “enzyme”. Most enzymes are proteins, and most such processes are chemical reactions. … Enzymes are often highly specific and act on only certain substrates.
What is proximity and orientation?
Proximity effects describe the orientation and movement of the substrate molecules when binding to enzyme active sites, and are most readily observed by comparing equivalent inter- and intramolecular reactions. … Proximity effects and orientation effects on reaction rates.What is the mechanism of enzyme catalysis Class 12?
Mechanism of enzyme catalyzed reaction is known as lock and key Mechanism. There specificity is due to the presence of active sites. The shape of active site of any given enzyme is like acavity such that only a specific substrate can fit into it. In the same way a key fit into a lock.
Why are chymotrypsin and subtilisin considered examples of convergent evolution?Why are chymotrypsin and subtilisin considered examples of convergent evolution? Because their polypeptide chains have different folds, but their active sites have identical residues.
Article first time published onHow does covalent catalysis lower activation energy?
The charge loss in the reaction during transitional state will then cause hydrolysis to accelerate. The residue becomes covalently attached to the substrate throughout the catalytic reaction adding an additional intermediate which helps stabilize later transition states by lowering the activation energy.
How does chymotrypsin use covalent catalysis?
Chymotrypsin, a protease, is an enzyme that cleaves the carbonyl side of certain peptide bonds by both general acid-base catalysis, but primarily covalent catalysis. In this mechanism, a nucleophile becomes covalently attached to a substrate in a transition state with an acyl-enzyme.
What are the 4 main catalytic strategies?
Serine proteases use four of the major catalytic mechanism during the reaction cycle: Acid-Base Catalysis, Covalent Catalysis, Electrostatic Interactions, and Desolvation.
What is enzyme catalysis with Example Class 12?
Common examples of enzyme catalysis reactions (1) Normal conversion of glucose into ethanol by zymase (enzyme) present in yeast. (2) Hydrolysis of urea (NH2CONH2) by urease (enzyme) present in soyabean. … (5) Hydrolysis of sugar into glucose and fructose by invertase (enzyme) present in yeast.
What is a key difference between what happens in a covalent catalysis mechanism as opposed to the other catalytic strategies?
The intermediate is stabilized by the interactions of the oxyanion hole. The reaction mechanism is shown on Fig. 8.25. How does the enzyme chymotrypsin bind, and specifically, hydrolyze its substrate?
What is the role of entropy in enzyme catalysis?
The idea that enzymes accelerate their reactions by entropic effects has played a major role in many prominent proposals about the origin of enzyme catalysis. … It is also implied that the binding entropy is equal to the activation entropy, (ΔSw‡)′, of the corresponding solution reaction.
What are three mechanisms of enzyme catalysis?
The chemical nature of enzyme catalysis Enzyme-catalysis can include, within a single reaction mechanism, acid-base, electrostatic and covalent catalysis as well as proximity effects, orbital steering and stress/strain factors.
What is a transient covalent bond?
A transient covalent bond is formed between the enzyme and the substrate. … A new pathway from substrate(s) to product(s) is formed that is faster than the uncatalyzed reaction.
What is an example of an enzyme catalyzed reaction?
Enzymes are highly specific catalysts for biochemical reactions, with each enzyme showing a selectivity for a single reactant, or substrate. For example, the enzyme acetylcholinesterase catalyzes the decomposition of the neurotransmitter acetylcholine to choline and acetic acid.
What are the important characteristics of enzyme catalysis?
Characteristics of enzyme catalysis: The effectiveness of a catalyst is maximum at its optimum temperature. The activity of the biochemical catalysts declines at either side of the optimum temperature. Biochemical catalysis is dependent upon the pH of the solution.
What is the shape selective catalysis?
Shape-selective catalysis is the molecular-sieving function in action during a catalytic reaction that distinguishes between the reactant, the product or the transition state species in terms of the relative sizes of the molecules and the pore space where the reaction occurs.
What is the enzyme?
An enzyme is a substance that acts as a catalyst in living organisms, regulating the rate at which chemical reactions proceed without itself being altered in the process. The biological processes that occur within all living organisms are chemical reactions, and most are regulated by enzymes.
What is transition state stabilization?
Transition state stabilization (by electrostatic interactions, including hydrogen bonds) is found to be central to catalysis by the enzyme. … The active site is clearly complementary to the transition state for the reaction, stabilizing it more than the substrate, so reducing the barrier to reaction.
What is metal ion catalysis?
Metal ion catalysis, or electrostatic catalysis, is a specific mechanism that utilizes metalloenzymes with tightly bound metal ions such as Fe2+, Cu2+, Zn2+, Mn2+, Co3+, Ni3+, Mo6+ (the first three being the most commonly used) to carry out a catalytic reaction. … Enzymes can catalyze a reaction by the use of metals.
Which statement expresses the correct relationship between Cosubstrates coenzymes and cofactors?
Which statement expresses the correct relationship between cosubstrates, coenzymes, and cofactors? Cosubstrates are coenzymes that transiently bind to enzymes, while coenzymes are cofactors that are organic compounds.
What is the difference between trypsin chymotrypsin and elastase?
However, each one of these proteases differs in their specificity; that is, they differ in the type of amino acids that they cleave. Chymotrypsin cleaves peptides on the carboxyl end of large,hydrophobic side chains, trypsin cleaves on the carboxyl end of large, positively-charged side chains such as arginine and …
Why is chymotrypsin called a serine protease?
They are called the serine proteases for two reasons: They hydrolyze proteins. They have an essential Ser residue at the active site which is critical for catalysis. In fact this Ser is much more reactive then other serines in the protein.
What is the basis of chymotrypsin's substrate specificity?
Chymotrypsin, like most enzymes, is specific in the types of substrates with which it reacts. As a protease, it cleaves polypeptides, and its inherent specificity allows it to act only on the carboxy-terminal of aromatic residues.
What is nucleophilic catalysis in chemistry?
Nucleophilic catalysis is the enzymatic analogy to anchimeric assistance by neighboring groups in organic reaction mechanisms. Anchimeric assistance is the process by which a neighboring functional group assists in the expulsion of a leaving group by intermediate covalent bond formation.
What is the function of carboxypeptidase?
Carboxypeptidase M (EC 3.4. 17.12) belongs to the family of the carboxypeptidases. These enzymes remove C-terminal amino acids from peptides and proteins and exert roles in the physiological processes of blood coagulation/fibrinolysis, inflammation, food digestion and pro-hormone and neuropeptide processing.
What is the role of histidine?
Histidine is required for synthesis of proteins. It plays particularly important roles in the active site of enzymes, such as serine proteases (e.g., trypsin) where it is a member of the catalytic triad. Excess histidine may be converted to trans-urocanate by histidine ammonia lyase (histidase) in liver and skin.