What is alpha decay process
 Rachel Young
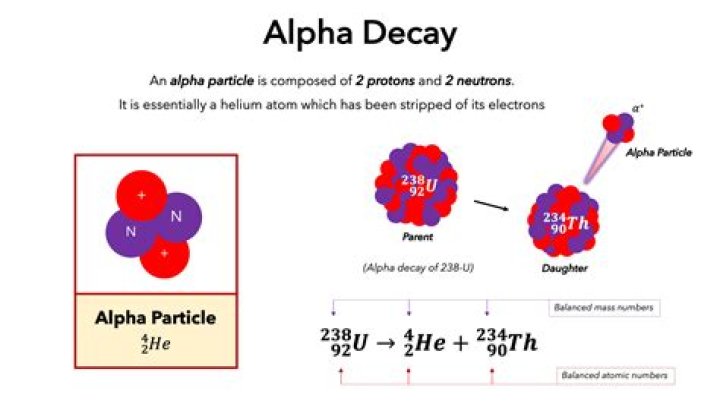
Rachel Young Alpha decay is a nuclear decay process where an unstable nucleus changes to another element by shooting out a particle composed of two protons and two neutrons. This ejected particle is known as an alpha particle and is simply a helium nucleus. Alpha particles have a relatively large mass and a positive charge.
What do you mean by alpha decay?
Alpha decay is the nuclear decay process whereby the parent nucleus emits an alpha particle. The alpha particle, structurally equivalent to the nucleus of a helium atom and denoted by the Greek letter α, consists of two protons and two neutrons.
What are the conditions of alpha decay?
Alpha (α) decay occurs when the N/Z ratio in a nucleus is too low. In this process, an “alpha particle,” which is the same as a helium nucleus 4He, is emitted. Examples for alpha decay: U 92 238 → T 90 234 h + H 2 4 e .
What is beta decay process?
Beta decay occurs when, in a nucleus with too many protons or too many neutrons, one of the protons or neutrons is transformed into the other. … However within a nucleus, the beta decay process can change a proton to a neutron. An isolated neutron is unstable and will decay with a half-life of 10.5 minutes.What is alpha decay and beta decay?
Alpha decay – A common mode of radioactive decay in which a nucleus emits an alpha particle (a helium-4 nucleus). Beta decay – A common mode of radioactive decay in which a nucleus emits beta particles. The daughter nucleus will have a higher atomic number than the original nucleus.
What are the 3 types of beta decay?
- Beta-minus decay. Nuclei that are rich in neutrons tend to decay by emitting an electron along with an antineutrino. …
- Beta-plus decay. Neutron-deficient nuclei tend to decay by positron emission or electron capture (see below). …
- Electron capture. …
- Double beta decay.
What is neutrino and antineutrino?
An antineutrino is the antiparticle partner of the neutrino, meaning that the antineutrino has the same mass but opposite “charge” of the neutrino. Although neutrinos are electromagnetically neutral (they have no electric charge and no magnetic moment), they may carry another kind of charge: lepton number.
How tunnel effect explain alpha decay?
The quantum tunneling or “tunnel effect” describes the fact that a particle behaves as both a particle and a wave in the infinitesimally small world where quantum mechanics replaces classical mechanics.What is the charge of alpha particle?
alpha particle, positively charged particle, identical to the nucleus of the helium-4 atom, spontaneously emitted by some radioactive substances, consisting of two protons and two neutrons bound together, thus having a mass of four units and a positive charge of two.
What elements most commonly decay by alpha decay?Alpha decay usually occurs in heavy nuclei such as uranium or plutonium, and therefore is a major part of the radioactive fallout from a nuclear explosion.
Article first time published onWhat quantities are conserved in alpha decay?
Some quantities that are conserved in nuclear decays are: the charge, the total number of neutrons and protons, total energy, the total momentum of the system, and the total lepton number. To determine if a particular decay is possible or not, one often considers these conserved quantities.
What is the difference between alpha and beta and gamma?
They are alpha (α) particles, beta (β) particles, and gamma (γ) particles. The main difference between alpha beta and gamma particles is that alpha particles have the least penetration power while beta particles have a moderate penetration power and gamma particles have the highest penetration power.
What is the relation between alpha and gamma?
The relationship between the coefficients of linear expansion α, superficial expansion β and cubical expansion γ of a solid is α:β:γ=1:2:k.
What is alpha and beta in chemistry?
The alpha carbon (Cα) in organic molecules refers to the first carbon atom that attaches to a functional group, such as a carbonyl. The second carbon atom is called the beta carbon (Cβ), and the system continues naming in order with Greek letters.
What is positron and neutrino?
Positron and Neutrino The emission of a positron or an electron is referred to as beta decay. The positron is accompanied by a neutrino, an almost massless and chargeless particle. Positrons are emitted with the same kind of energy spectrum as electrons in negative beta decay because of the emission of the neutrino.
What is neutrinos Upsc?
A neutrino is a subatomic particle that is very similar to an electron, but has no electrical charge and a very small mass, which might even be zero. Neutrinos are one of the most abundant particles in the universe. Because they have very little interaction with matter, however, they are incredibly difficult to detect.
What are quarks made?
Quarks are a type of particle that constitute matter. Look around you…all of the matter that you see is made up of protons and neutrons, and these particles are composed of quarks. There are three pairs (or families) of quarks for a total of six. They are: up/down, charm/strange, top/bottom.
Why are neutrinos emitted?
Neutrinos are born in various decays, which is when a particle changes from one type into another. There are a couple of ways this can happen. Sometimes, elementary particles (which are the ones that can’t be broken down any further) transform into different, lighter particles, often creating neutrinos in the process.
What is an example of beta decay?
The decay of technetium-99, which has too many neutrons to be stable, is an example of beta decay. A neutron in the nucleus converts to a proton and a beta particle. The nucleus ejects the beta particle and some gamma radiation. The new atom retains the same mass number, but the number of protons increases to 44.
What causes positron emission?
Positron emission occurs when a proton in a radioactive nucleus changes into a neutron and releases a positron and an electron neutrino.
Why does alpha decay happen?
Alpha decay occurs when a nucleus is unstable because it has too many protons. … The nucleus emits an alpha particle and energy. An alpha particle consists of two protons and two neutrons, which is actually a helium nucleus. Losing the protons and neutrons makes the nucleus more stable.
Do alpha particles have electrons?
An alpha particle is identical to a helium atom that has been stripped of its two electrons; thus, an alpha particle contains two protons and two neutrons. Because an alpha particle has no electrons to balance the positive charge of the two protons, it has a charge of +2 and can be represented as He2+.
How alpha rays are produced?
An alpha particle is produced by the alpha decay of a radioactive nucleus. Because the nucleus is unstable a piece of it is ejected, allowing the nucleus to reach a more stable state.
What is alpha tunneling?
Alpha decay is a quantum tunneling process. This is similar to cluster decay, in which an atomic nucleus emits a small “cluster” of neutrons and protons (e.g., 12C). The height of the Coulomb barrier for nuclei of A « 200 is about 20-25 MeV. … The alpha particle is trapped in a potential well by the nucleus.
How does an alpha particle escape the nucleus?
Essentially, the alpha particle escapes from the nucleus not by acquiring enough energy to pass over the wall confining it, but by tunneling through the wall.
What is called induced radiation?
Induced radioactivity, also called artificial radioactivity or man-made radioactivity, is the process of using radiation to make a previously stable material radioactive. … Neutron activation is the main form of induced radioactivity. It occurs when an atomic nucleus captures one or more free neutrons.
What can stop alpha decay?
In general, alpha particles have a very limited ability to penetrate other materials. In other words, these particles of ionizing radiation can be blocked by a sheet of paper, skin, or even a few inches of air.
What symbol represents an alpha particle?
Equations for Alpha Decay For example, an alpha particle (helium nucleus) is represented by the symbol 42He, where He is the chemical symbol for helium, the subscript 2 is the number of protons, and the superscript 4 is the mass number (2 protons + 2 neutrons).
What is the chemical symbol of polonium?
polonium (Po), a radioactive, silvery-gray or black metallic element of the oxygen group (Group 16 [VIa] in the periodic table).
What happens to the nucleus during alpha decay?
Alpha decay is a nuclear decay process where an unstable nucleus changes to another element by shooting out a particle composed of two protons and two neutrons. … This ejected particle is known as an alpha particle and is simply a helium nucleus.
What happens when alpha particle is emitted?
When an atom emits an alpha particle in alpha decay, the atom’s mass number decreases by four due to the loss of the four nucleons in the alpha particle. The atomic number of the atom goes down by two, as a result of the loss of two protons – the atom becomes a new element.