What happens during positron emission
 Rachel Young
Rachel Young Positron emission increases the number of neutrons and decreases the number of protons, making the nucleus more stable. In positron emission, the atomic number Z decreases by one while the mass number A remains the same. … It undergoes positron emission to form sodium-23.
What happens when positron is emitted?
Positron Emission They are emitted from the nucleus of some radioisotopes that are unstable because they have an excessive number of protons and a positive charge. Positron emission stabilizes the nucleus by removing a positive charge through the conversion of a proton into a neutron.
What happens to the atomic number during positron emission?
In positron emission, the atomic number Z decreases by 1 while the mass number A remains the same. As with beta decay, the kinetic energy -ΔE is split between the emitted particles – in this case the positron and neutrino.
What happens during positron emission quizlet?
During positron emission, a proton in the nucleus is converted into a neutron and a positron, and then the positron is emitted.Does a positron emission release an electron?
Positron emission is the release of a positron and an electron neutrino in the process of radioactive decay. Electron capture is a process which emits an electron neutrino. Both these processes take place in proton-rich nuclei.
What is emitted during beta decay?
In electron emission, also called negative beta decay (symbolized β−-decay), an unstable nucleus emits an energetic electron (of relatively small mass) and an antineutrino (with little or possibly no rest mass), and a neutron in the nucleus becomes a proton that remains in the product nucleus. …
What is positron emission formula?
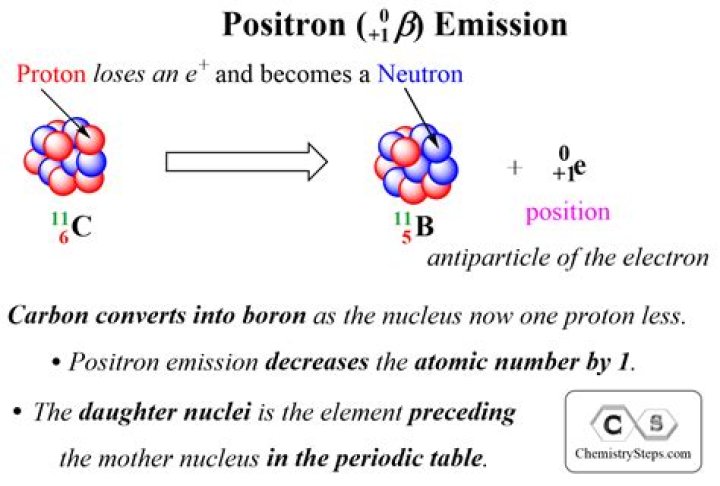
Like the beta particle, a positron is immediately ejected from the nucleus upon its formation. The symbol for a positron in an equation is e+01. For example, potassium-38 emits a positron, becoming argon-38. K3819→Ar3818+e01. Positron emission decreases the atomic number by one, but the mass number remains the same.
What happens to the decaying positron emission?
What happens to the decaying proton during positron emission? Turns into a neutron and a gamma ray and a positron are released. What is the mass number and charge of the emitted positron? The charge is positive.What happens to a proton during positron decay?
In positron emission, also called positive beta decay (β+-decay), a proton in the parent nucleus decays into a neutron that remains in the daughter nucleus, and the nucleus emits a neutrino and a positron, which is a positive particle like an ordinary electron in mass but…
What happens to the decaying neutron during the beta decay?Beta decay occurs when, in a nucleus with too many protons or too many neutrons, one of the protons or neutrons is transformed into the other. In beta minus decay, a neutron decays into a proton, an electron, and an antineutrino: n Æ p + e – +.
Article first time published onWhy does positron decay have the same effect on the nucleus as electron capture?
The capture of an electron has the same effect on a nucleus as the emission of a positron: one of its protons transforms into a neutron, diminishing the global electric charge of the nucleus by 1 unit. … The captured electron belongs to the group of electrons orbiting around the nucleus.
How do you know if positron emission or electron capture?
In positron emission, a proton is converted to a neutron by emitting a positron and a neutrino. In electron capture, an outside electron is pulled inside the nucleus and combined with a proton to make a neutron, emitting only a neutrino.
What isotope produces boron 11 when it emits a positron?
Commonly used isotopes include: Carbon-11 or 11C is a radioactive isotope of carbon that decays 100% into Boron-11 by positron emission.
When a positron is emitted from the nucleus of an atom the mass number?
When a nucleus emits a beta particle (positron), its atomic number decrease by 1 and its mass number remains unchanged. A proton and an electron combine to form a neutron. The mass number stays the same, but the atomic number decreases by 1.
Which particle is emitted during testing of weld in the decay of positron?
Positron emission, beta plus decay, or β+ decay is a subtype of radioactive decay called beta decay, in which a proton inside a radionuclide nucleus is converted into a neutron while releasing a positron and an electron neutrino (νe).
Why does beta decay by positron emission happen?
A positron is the antiparticle of the electron, i.e. it has a +1 charge and the same mass as an electron. Notice that nuclear transmutations take place in both cases, and that each type of beta decay brings the initial ratios of protons to neutrons in the nucleus to 1:1, this being the main reason for their occurrence.
How does a neutron change into a proton?
Protons and neutrons consist of fundamental particles called quarks. A down quark within the neutron transforms into an up quark, changing the neutron into a proton (and changing the atomic element as a result).
What happens to an atom's atomic After beta decay its mass #?
Beta decay leaves the mass number unchanged, so the change of nuclear spin must be an integer.
What happens during beta decay quizlet?
What happens during beta decay? A neutron in an unstable nucleus changes into a negatively charged beta particle and a proton. The new proton remains inside the nucleus so now the nucleus has one less neutron and one more proton. The atomic mass stays the same but the atomic number increases by 1.
Why do a positron and a beta particle have the same mass?
Why do a positron and a beta particle have the same mass? Beta Particles are nothing but fast moving Electrons. Positron is the anti particle of the electron and that is the reason they have same mass, but opposite electronic charges.
What isotope is produced for the reaction in which oxygen 15 undergoes positron emission?
The oxygen-15 isotope decays with a half-life of about two minutes to nitrogen-15, emitting a positron.
What happens when a nucleus is stabilized by converting a proton into a neutron?
Positron emission occurs when a nucleus has too few neutrons and too many protons (the neutron to proton ratio is too low). The nucleus becomes more stable by changing a proton into a neutron and a positron (positively charged electron). The neutron remains in the nucleus and the positron is ejected at high speed.
What radiation emits positrons?
Positrons are emitted in the positive beta decay of proton-rich (neutron-deficient) radioactive nuclei and are formed in pair production, in which the energy of a gamma ray in the field of a nucleus is converted into an electron-positron pair.





