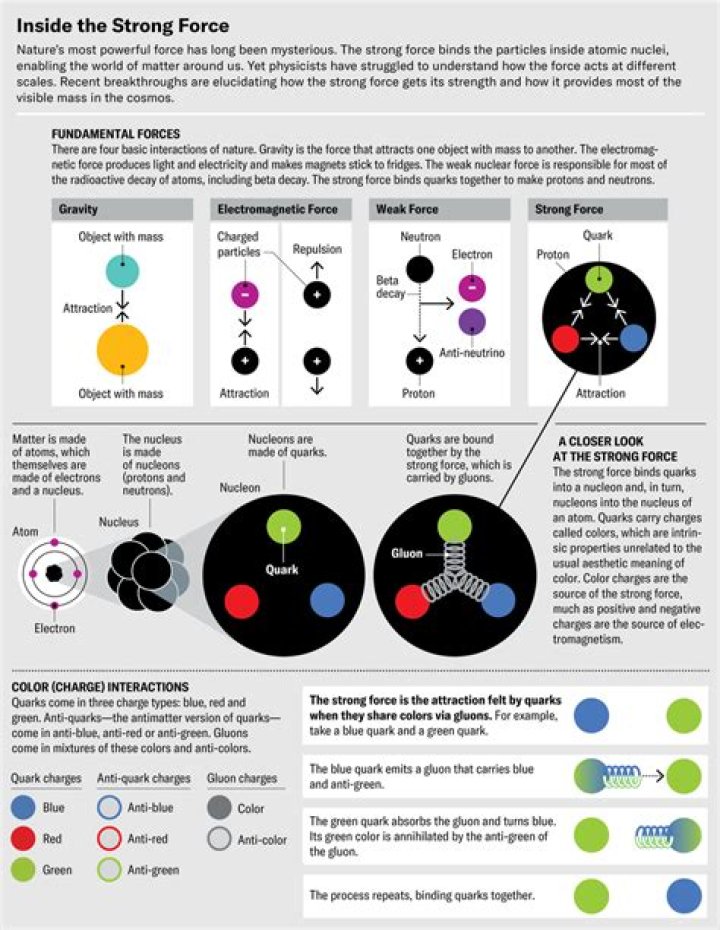
electromagnetic force
.
Similarly, what causes electrons to orbit the nucleus?
The nucleus is heavy and positively charged. The electron is light and negatively charged. They attract, so if the electron moves sideways at the right speed it will bend toward the nucleus and just keep on bending in a circle, the same as a satellite orbiting earth.
Secondly, what is the force that holds the nucleus of an atom together? nuclear force
In this way, how do electrons move around the nucleus of an atom?
Protons and neutrons make up the nucleus in the center of the atom. “Early on, we thought that electrons were revolving around the nucleus like planets,” Cooley said. If you add energy, electrons can move to a higher orbital level and then back again when the energy returns to normal.
What are electrons made of?
So in answer to the question "What are electrons made up of?" is, electrons are made up of charge, mass, and angular momentum. To make an electron, reach out across the electric field (which is otherwise just sitting there) and introduce enough wiggle to create −1.602×10−19 coulombs of charge.
Related Question Answers
Do electrons orbit the nucleus of an atom?
The electrons do not orbit the nucleus in the manner of a planet orbiting the sun, but instead exist as standing waves. Thus the lowest possible energy an electron can take is similar to the fundamental frequency of a wave on a string.Why do electrons not fall into the nucleus of an atom?
Electrons are not little balls that can fall into the nucleus under electrostatic attraction. Rather, electrons are quantized wavefunctions that spread out in space and can sometimes act like particles in limited ways. An electron in an atom spreads out according to its energy.Do electrons have a negative charge?
Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite. Neutrons have no charge.What do electrons orbit around?
Electrons are found in different levels -- or orbitals -- surrounding the nucleus. The electrons can be found at any point in their orbital. Protons and neutrons make up the nucleus in the center of the atom. “Early on, we thought that electrons were revolving around the nucleus like planets,” Cooley said.Do atoms spin?
Elementary particles have a fundamental property called 'spin' that determines how they align in a magnetic field. MIT researchers have created a new physical system in which atoms with clockwise spin move in only one direction, while atoms with counterclockwise spin move in the opposite direction.Why do electrons have mass?
The electron has a mass that is approximately 1/1836 that of the proton. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer de Broglie wavelength for a given energy.Why do electrons have a negative charge?
Electrons are the negatively charged particles of atom. Together, all of the electrons of an atom create a negative charge that balances the positive charge of the protons in the atomic nucleus. Electrons are extremely small compared to all of the other parts of the atom.Why do electrons move and not protons?
Protons interact in ways that electrons do not. Electrons are not affected by the strong force, and so they only get trapped by the electrical attraction to the nucleus which is much weaker in ionized atoms. Therefore it is easier for electrons to move away from one atom to another, transferring charge.Does an electron move?
With all of this in mind, an electron in a stable atomic state does not move in the sense of a solid little ball zipping around in circles like how the planets orbit the sun, since the electron is spread out in a wave. Furthermore, an electron in a stable atomic state does not move in the sense of waving through space.Where do electrons get their energy?
It requires an external source of energy in order to change to a higher-energy state or else to leave the atom and become a free electron again. Typically it gets this energy either by absorbing a photon or by absorbing some kinetic energy when its atom hits another atom.Why do electrons stay in the atom?
The answer is that electrons do in fact “fly right into the nucleus of an atom.” The lowest energy orbitals in most atoms do overlap the nucleus, and so the electrons spend some of their time moving through that volume of space. Electrons and protons aren't little cannonballs orbiting around each other.Why is an atom electrically neutral?
Atoms are electrically neutral because they have equal numbers of protons (positively charged) and electrons (negatively charged). If an atom gains or loses one or more electrons, it becomes an ion.What is the overall charge of an atom?
The overall charge of an atom is zero. Atoms are made up of positively charged particles called protons and negatively charged particles called electrons as well as non-charged particles called neutrons.What are valence electrons used for by an element?
Valence electrons are the outermost shell electrons of an atom which it uses to form bonds with other atoms.Can a proton move?
Protons and neutrons never move from object to object. The energy that comes from these charged particles is called electrical energy.How fast do electrons move around an atom?
A calculation shows that the electron is traveling at about 2,200 kilometers per second. That's less than 1% of the speed of light, but it's fast enough to get it around the Earth in just over 18 seconds. Read up on what happens when nothing can go faster than the speed of light.What force holds electrons in the atom?
the electromagnetic force
What is the space between nucleus and electrons?
The empty space between the atomic cloud of an atom and its nucleus is just that: empty space, or vacuum. That's the simple answer, but there are a few subtleties: 1) Sub-atomic particles such as electrons, protons and neutrons need to be treated as quantum objects.What force of attraction holds the electron in the hydrogen atom?
the electromagnetic force
 Isabella Bartlett
Isabella Bartlett