What does the triple point on a phase diagram describe
 Rachel Young
Rachel Young The triple point represents the combination of pressure and temperature that facilitates all phases of matter at equilibrium. The critical point terminates the liquid/gas phase line and relates to the critical pressure, the pressure above which a supercritical fluid forms.
What does the triple point represent in a phase diagram?
phase diagram …the three curves, called the triple point, represents the unique conditions under which all three phases exist in equilibrium together. A phase diagram for two components usually shows melting curves on a temperature-composition diagram.
What does the triple point on a phase diagram describe quizlet?
What does the triple point on a phase diagram describe? The lines show the conditions of temper- ature and pressure at which two phases exist in equilibrium.
What does the triple point describe?
Definition of triple point : the condition of temperature and pressure under which the gaseous, liquid, and solid phases of a substance can exist in equilibrium.What is a triple point during a phase change?
Triple Point Explained: In physics and chemistry, the triple point of a substance is the temperature and pressure at which three phases (gas, liquid, and solid) of that substance may coexist in thermodynamic equilibrium.
Which of the following describes the triple point of water?
Which of the following describes the triple point of water? The particular value of temperature and the particular value of pressure at which liquid water, solid ice, and water vapor can coexist. … Which temperature scale has the zero at the lowest possible temperature?
What is triple point pressure of water in phase diagram?
The triple point for water occurs at a pressure of 4.6 torr and 0.01oC. Click on the diagram to see a microscopic view of the phase(s) that exist for a given temperature and pressure. At this temperature and pressure, liquid and gaseous water exist in equilibrium.
Which statement about the critical point and the triple point must be true?
The triple point and critical point of every substance is different. Therefore, the only true statement is: D. Both the triple point and the critical point of a substance are dependent on the substance only, but not on temperature or pressure.What is meant by triple point and eutectic point?
The triple point is the temperature and pressure at which solid, liquid, and vapour phases of a particular substance co-exist in equilibrium. The term Eutectic point is the temperature and pressure at which a certain liquid mixture transforms into two solid phases at the same time upon cooling the liquid.
What does the critical point on a phase diagram describe about a substance quizlet?critical point: The point above which a substance cannot be liquefied regardless of the pressure applied. The conditions (temperature, pressure and sometimes composition) at which a phase boundary ceases to exist.
Article first time published onWhat phases are present at upper triple point?
Thus, the phases at the upper triple point are diamond, graphite, and liquid.
What is triple point of water Why is it called triple point?
Triple point of water is defined as the temperature and pressure at which liquid water , solid ice and water vapour can coexist in a stable equilibrium . As , at triple point of water , both solid (ice) and vapour state are present so we can say that boiling point and freezing point become same .
What substances have a triple point?
Triple PointsSubstanceTemperature [°C]Pressure [kPa]Ammonia-77.756.076Argon-189.3468.9Arsenic8203628
Why is triple point important?
At the triple point, all three phases (solid, liquid, and gas) are in equilibrium. Since the triple point is a point, there is only one temperature and one pressure where the three phases will exist. This fact often helps in identifying compounds or in problem solving.
Do all elements have a triple point?
The only point at which all three phases may exist in equilibrium is the triple point.
What is Eutectoid point in phase diagrams?
The location on a phase diagram indicating the eutectoid composition and eutectoid temperature of an alloy. The eutectoid point also indicates the location at which three solid phases co-exist. In the iron-carbon phase diagram, this point indicates a temperature of 1333 F and a carbon content of 0.8%.
What is eutectic point in phase diagrams?
The point, E, where the liquidus curves and solidus intersect, is termed the eutectic point. At the eutectic point in this two component system, all three phases, that is Liquid, crystals of A and crystals of B, all exist in equilibrium.
Which statement is correct about the critical point of a phase diagram?
Solid, liquid and gas are present in equilibrium.
Which statement about the triple point of a substance is correct?
The triple point for a substance varies with the pressure. The three phases (solid, liquid, gas) are in equilibrium. Three phases (solid,liquid, gas) are indistinguishable in appearance.
What phase is present at point A on the phase diagram?
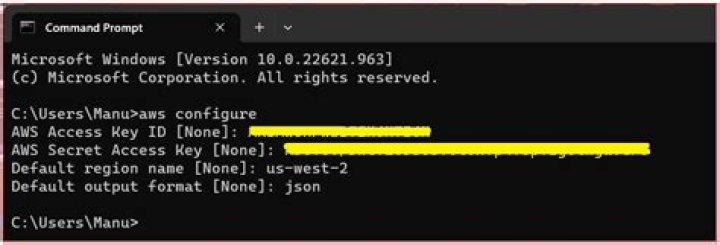
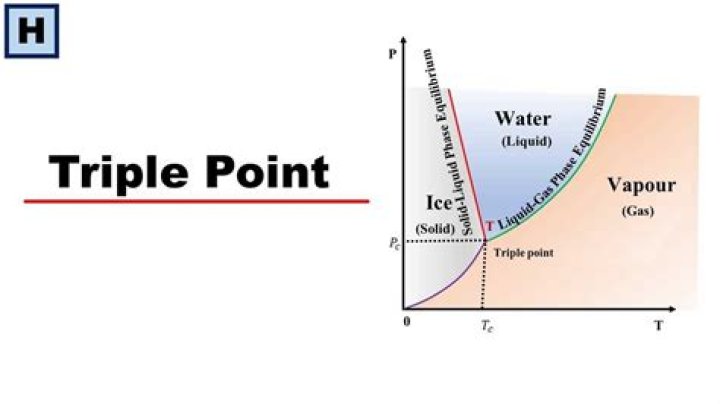
Finally, point A, where the solid/liquid, liquid/gas, and solid/gas lines intersect, is the triple pointThe point in a phase diagram where the solid/liquid, liquid/gas, and solid/gas lines intersect; it represents the only combination of temperature and pressure at which all three phases are in equilibrium and can …
What is the temperature and pressure of the triple point quizlet?
The triple point of a substance is the temperature and pressure at which three phases of a substance may coexist in thermodynamic equilibrium. -In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in equilibrium.
What is a critical point chemistry quizlet?
Critical point. The temperature and pressure at which the gas and liquid states of a substance become identical and form one phase. Critical temperature. The temperature above which a substance cannot exist in the liquid state.
Which phase is denser solid or liquid?
The solid phase is more dense than the liquid phase. The line that separates solid and liquids bends right. IF the solid phase is less dense than the liquid phase.
What is the function of phase diagram?
A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions (pressure, temperature, volume, etc.) at which thermodynamically distinct phases (such as solid, liquid or gaseous states) occur and coexist at equilibrium.
Which phases can coexist at equilibrium at point A on the phase diagram?
Finally, point A, where the solid/liquid, liquid/gas, and solid/gas lines intersect, is the triple point; it is the only combination of temperature and pressure at which all three phases (solid, liquid, and gas) are in equilibrium and can therefore exist simultaneously.
What do curved lines on a phase diagram represent?
The curved lines represent conditions of pressure and temperature at which two phases exist in equilibrium. What do the curved lines on a phase diagram represent? … Gas, volume, and temperature.
Is the triple point the melting point?
The triple point and critical point are labeled. The solid green line represents the melting point of most liquids, and the dotted green line represents the unusual behavior of water. … The point where liquid become stable is called the triple point, where all three phases (solid, liquid and gas) are all in equilibrium.
Does the phase diagram of helium have a triple point?
Helium does not. Unlike the usual triple point where colder temperatures result in the material becoming solid, the phase diagram of helium shows that it remains a liquid (a superfluid, in fact) even down to absolute zero.