What are the differences between acids and bases
 Christopher Lucas
Christopher Lucas An acid increases the concentration of H+ ions. A base is a substance that releases hydroxide (OH-) ions in aqueous solution, donates electrons and accepts protons.
What are three differences between acids and bases?
Bases usually exist in the solid-state except for ammonia which exists as a gas. Acids are sour in taste. Bases taste bitter. Acids release hydrogen ions (H+) when dissolved in water.
What are two key differences between acids and bases?
Differences Between Acid and Base A base is an aqueous substance that donates electrons, accept protons or release hydroxide (OH-) ions. An acid is a proton donor. While a base is a proton acceptor. Acetic acid (CH₃COOH) and sulphuric acid are two examples of Acid.
What is the difference between acids and bases for kids?
Acids and bases are two special kinds of chemicals. … Whether a liquid is an acid or base depends on the type of ions in it. If it has a lot of hydrogen ions, then it is an acid. If it has a lot of hydroxide ions, then it is a base.What is the main difference between acid and base class 10?
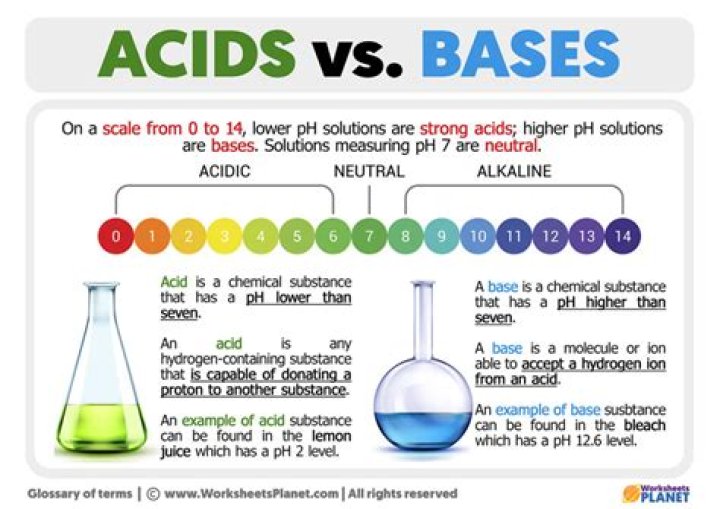
ACIDSBASESThe pH values of acids are basically less than 7The pH values of bases are basically more than 7.Acids turn blue litmus red.The Base turns red litmus blue.Acids remain colourless when dissolved in phenolphthaleinBases makes solution pink coloured when dissolved in phenolphthalein
What is the difference between an acid and a base list some properties of each and explain how do you name them?
Essentially, acids accept electron pairs and donate hydrogen protons. In contrast, bases donate electrons and accept hydrogen protons. In addition to these definitions, acids and bases can be classified by their properties. These properties include pH, taste, texture, reactivity, and conductivity.
Why is it important to know the difference between acids and bases?
Additionally, it’s important to know the difference between acids and bases because mixing the two together can cause a reaction. While mixing vinegar and baking soda can create a great cleaning agent, mixing strong acids and bases can create toxic fumes or even explosions.
What is the difference between acids and bases on the pH scale?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. … pHs less than 7 are acidic while pHs greater than 7 are alkaline (basic).What is the difference between acid and base for Class 7?
Any compound with a pH value between zero to seven is considered acidic, whereas a pH value between seven to fourteen is a base. An acid is called a proton donor, while a base is called a proton acceptor (Kolb, 1978).
What is the use of acid and base?Uses of AcidsUses of BasesNitric acid – fertilizer production, explosivesMagnesium hydroxide – Milk of Magnesia (antacid)Trichloroacetic acid – removes warts, fouls wineAmmonium hydroxide – smelling saltsAcetyl salicylic acid – aspirinAluminum hydroxide – fire extinguishers
Article first time published onWhat is the reaction between acids and bases called?
The reaction of an acid with a base is called a neutralization reaction. The products of this reaction are a salt and water.
What is the difference between acids bases and salts?
An acid is defined as a substance whose water solution tastes sour, turns blue litmus red and neutralizes bases. A substance is called base if its aqueous solution tastes bitter, turns red litmus blue or neutralizes acids. Salt is a neutral substance whose aqueous solution does not affect litmus.
What are the 5 differences between an acid and a alkali?
Acids have a pH less than 7. Alkalis have a pH greater than 7 Neutral substances have a pH equal to 7. … Acids contain lots of hydrogen ions, which have the symbol H+. Alkalis contain lots of hydroxide ions, symbol OH-.
What are the differences between acids and bases Brainly?
ACIDS HAVE SOUR IN TASTE WHEREAS BASE HAVE BITTER IN TASTE ACIDS PH VALUE IS LESS THAN 7 BUT BASE HAVE THE PH VALUE IS MORE THAN 7 ACIDS TURNS BLUE LITIMUS TO RED WHEREAS BASE TURNS RED TO BLUE. HCL, HNO3, CH3COOH, ETC ARE SOME EXAMPLES OF ACIDS AND NAOH, MG(OH) 2, ETC ARE SOME EXAMPLES OF BASE.
How do you tell the difference between an acid and a base in a chemical equation?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).
How does a base differ from an acid quizlet?
An acid is a substance that releases hydrogen ions into an aqueous solution. A base is a substance that releases hydroxide ions.
What is the difference between acids and bases on the basis of their behavior when dissolved in water?
When dissolved in water, acids donate hydrogen ions (H+). Hydrogen ions are hydrogen atoms that have lost an electron and now have just a proton, giving them a positive electrical charge. Bases, on the other hand, mixed with water yield hydroxide ions (OH-).
What are the characteristics of bases and acids?
In general, acids are sour/tart, feel like water, may sting when touched, react strongly with metals, conduct electricity since they ionize in solutions, and turn blue litmus paper red. Bases are bitter, slippery and smooth, do not react with metals usually, conduct electricity, and turn red litmus paper blue.
What are the similarities and differences between acids and bases?
Similarities between Acids and Bases. The following are the similarities between the two: Most strong acids and bases are both corrosive in nature. They tend to corrode or rust metals. Both, acids and bases change the colour of litmus paper.
What are some everyday examples of acids and bases in your household?
- Ammonia.
- Baking soda – sodium bicarbonate.
- Washing soda – sodium carbonate.
- Soap – Either sodium or potassium hydroxide.
- Detergents.
- Shampoo.
- Borax.
- Chlorine bleach.
What are properties of bases?
- Aqueous solutions of bases are also electrolytes. …
- Bases often have a bitter taste and are found in foods less frequently than acids. …
- Bases also change the color of indicators. …
- Bases do not react with metals in the way that acids do.
What do all acids and bases have in common explain with experiment?
Common in acids and bases When acid and bases are dissolved in water solution, acid gives H+ ions whereas base gives OH– ions. So both acid and bases are electrolytes which conducts electricity.
What is difference between salt and base?
Bases contain a huge amount of negative ions. They are of bitter taste and are very slippery. Salt is formed when acid and base both are mixed together and are neutralized. The negative ions form water whereas the positive ions form salt.
What is the difference between acids bases and alkalis?
An acid is a substance that has a pH lower than 7. For example, hydrochloric acid, sulphuric acid, citric acid, ethanoic acid (vinegar). A base is a substance that neutralises an acid – those that dissolve in water are called alkalis. An alkali is a soluble substance with a pH higher than 7.
What is difference between base and alkali?
The difference between an alkali and a base is: Alkali compounds are types of bases that dissolve in water whereas the base neutralizes the acid. All alkali are bases but all bases are not alkalis. Alkali is used for metals of group 1 in the periodic table whereas the base is a compound having OH ions.
What is the main difference between a base and an alkali?
BaseAlkaliA substance which is used to neutralise acid is known as base.Alkali is also a base. Bases which are soluble in water are known as alkali.Bases do not dissolve in water.All alkalis are base but all bases are not alkalis.
Why is it important to understand the difference of an acid and base solution What benefits does it bring to us in our daily lives?
Acids and bases are important in living things because most enzymes can do their job only at a certain level of acidity. Cells secrete acids and bases to maintain the proper pH for enzymes to work. For example, every time you digest food, acids and bases are at work in your digestive system.