Protons, neutrons, and electrons: Both protons and neutrons have a mass of 1 amu and are found in the nucleus. However, protons have a charge of +1, and neutrons are uncharged. Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of -1..
Also, what is the mass of a proton?
Proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
Also, what is the mass of a electron? The invariant mass of an electron is approximately 9.109×10−31 kilograms, or 5.489×10−4 atomic mass units. On the basis of Einstein's principle of mass–energy equivalence, this mass corresponds to a rest energy of 0.511 MeV. The ratio between the mass of a proton and that of an electron is about 1836.
Additionally, what is the approximate ratio of the mass of an electron to that of a proton?
The mass of an electron is only about 1/2000 the mass of a proton or neutron, so electrons contribute virtually nothing to the total mass of an atom. Electrons have an electric charge of −1, which is equal but opposite to the charge of a proton, which is +1.
Are protons and electrons equal?
The number of protons in the nucleus of the atom is equal to the atomic number (Z). The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus.
Related Question Answers
Where are electrons found?
Electrons are found in shells or orbitals that surround the nucleus of an atom. Protons and neutrons are found in the nucleus. They group together in the center of the atom.How do you figure out neutrons?
Take note that the nucleus of an atom is composed of protons and neutrons. And the number of particles present in the nucleus is referred as mass number (Also, called as atomic mass). So, to determine the number of neutrons in atom, we only have to subtract the number of protons from the mass number.How do you find electrons in an ion?
The charge on the ion tells you the number of electrons. If the charge is positive, subtract that number from the atomic number to get the number of electrons. You have more protons. If the charge is negative, add the amount of charge to the atomic number to get the number of electrons.Why is proton positively charged?
A proton has positive charge of 1, that is, equal but opposite to the charge of an electron. The charge is believed to be from the charge of the quarks that make up the nucleons (protons and neutrons).Why is electron negatively charged?
Electrons are the negatively charged particles of atom. Together, all of the electrons of an atom create a negative charge that balances the positive charge of the protons in the atomic nucleus. Electrons are extremely small compared to all of the other parts of the atom.What is atomic mass number?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is different for each different isotope of a chemical element.What is mass made up of?
These particles are each made up of three quarks moving at breakneck speeds that are bound together by gluons, the particles that carry the strong force. The energy of this interaction between quarks and gluons is what gives protons and neutrons their mass.Who invented Proton?
Rutherford
How many grams is a proton?
The charge on the proton is +1. The proton has a mass of about 1 amu. In grams, this amounts to roughly 1.66 x 10-24 g, about 1837 times the mass of the electron.What is Proton made of?
Along with neutrons, protons make up the nucleus, held together by the strong force. The proton is a baryon and is considered to be composed of two up quarks and one down quark.What is charge and mass of proton?
Protons. A proton is one of three main particles that make up the atom. The other two particles are the neutron and electron. Protons are found in the nucleus of the atom. Protons have a positive electrical charge of one (+1) and a mass of 1 atomic mass unit (amu), which is about 1.67×10−27 kilograms.What is the mass of deuteron?
But the mass of the deuteron is 1875.6 MeV, implying that, upon energy grounds, it is stable agains such a decay. The free neutron yields an energy of 0.78 MeV in beta decay, but the 2.2 MeV binding energy of the deuteron prevents its decay.Do Protons have mass?
Protons and neutrons have approximately the same mass, about 1.67 × 10-24 grams. Scientists define this amount of mass as one atomic mass unit (amu) or one Dalton. Although similar in mass, protons are positively charged, while neutrons have no charge.What is inside a proton?
A proton is made up of 3 quarks, namely uud, but it is also constituted, as jinawee pointed out, of virtual quarks and antiquarks who are constantly being created and annihilated via strong force which is mediated by gluons, described by Quantum Chromodynamics (QCD).How is a proton formed?
A quark is a tiny particle which makes up protons and neutrons. After the invention of the particle accelerator, it was discovered that electrons are fundamental particles, but neutrons and protons are not. Neutrons and protons are made up of quarks, which are held together by gluons.What is an a particle?
Particles are tiny bits of matter that make up everything in the universe. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms and molecules are called microscopic particles. Subatomic particles are particles that are smaller than atoms.Which is heavier electron or proton?
Atomic particles Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. However, one proton is about 1,835 times more massive than an electron.Why is the charge to mass ratio of a proton different?
Why is the charge of the mass ratio of an electron the same irrespective of the gas taken in a tube while the charge of the mass ratio is different for protons? So for different gases their corresponding nuclei will have different number of neutrons so the ratio turns out to be different.How do you find the mass number?
Together, the number of protons and the number of neutrons determine an element's mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. 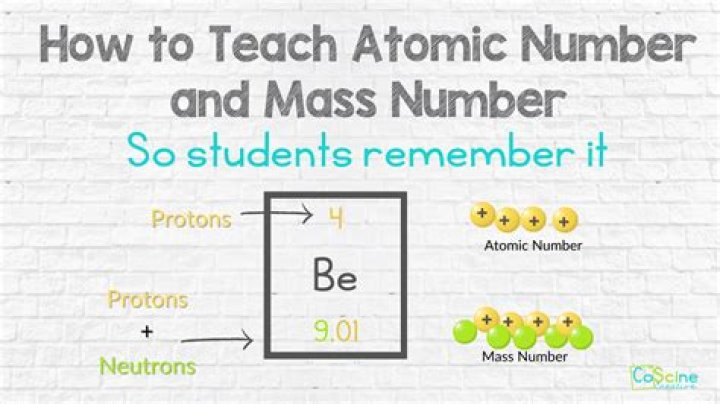
 Rachel Young
Rachel Young