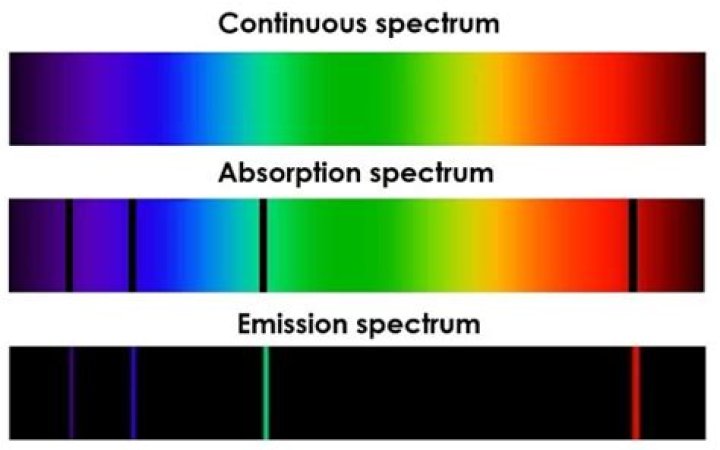
What are emission and absorption spectra why dark lines appear in the absorption spectra
 John Peck
John Peck Emission lines refer to the fact that glowing hot gas emits lines of light, whereas absorption lines
Why do dark lines appear in absorption spectra?
spectra. …or molecules, whereas in an absorption spectrum, portions of a continuous spectrum (light containing all wavelengths) are missing because they have been absorbed by the medium through which the light has passed; the missing wavelengths appear as dark lines or gaps.
What are the black lines in absorption spectrum?
Absorption lines are usually seen as dark lines, or lines of reduced intensity, on a continuous spectrum. This is seen in the spectra of stars, where gas (mostly hydrogen) in the outer layers of the star absorbs some of the light from the underlying thermal blackbody spectrum.
What is emission and absorption spectra?
The emission spectrum is the spectrum of radiation emitted by a substance that has absorbed energy. … The absorption spectrum is the opposite of the emission spectrum. It is the spectrum formed by electromagnetic radiation that has passed through a medium, in which radiation of some frequencies is absorbed.What do dark lines in a spectrum mean?
When we see dark lines in a spectrum, they correspond to certain wavelengths being missing due to absorption by matter (in the form of atoms/molecules) on their way. So the dark line represents “absence of light” in a spectrum, not any particular wavelength (color) of light.
What causes emission lines?
Emission lines occur when the electrons of an excited atom, element or molecule move between energy levels, returning towards the ground state. The spectral lines of a specific element or molecule at rest in a laboratory always occur at the same wavelengths.
Why are there dark lines in an absorption spectrum quizlet?
Dark lines in the absorption spectrum represent: particular energies of light coming in from a distant object that are absorbed by material in between. … The electrons in a cloud of hydrogen gas are making the transition from high energy states to the lowest state.
How are absorption and emission lines produced?
When electrons move from a higher energy level to a lower one, photons are emitted, and an emission line can be seen in the spectrum. Absorption lines are seen when electrons absorb photons and move to higher energy levels.What do emission spectra show?
The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an atom or molecule making a transition from a high energy state to a lower energy state. … Therefore, spectroscopy can be used to identify elements in matter of unknown composition.
Why are the dark absorption lines in an absorption spectrum at the exact same wavelength where the bright lines in an emission spectrum usually show up?Atoms can also absorb light of certain energies, resulting in a transition from the ground state or a lower-energy excited state to a higher-energy excited state. This produces an absorption spectrum, which has dark lines in the same position as the bright lines in the emission spectrum of an element.
Article first time published onWhat are continuous emission and absorption spectra how are they produced quizlet?
A continuous spectrum is produced by a hot, high-density light source. An emission spectrum is produced by a hot, low-density light source. An absorption spectrum is produced by a hot, high-density light source shining through a cool, low-density medium. You just studied 16 terms!
What colors of visible light are found in the emission spectrum of hydrogen?
(b) When the light emitted by a sample of excited hydrogen atoms is split into its component wavelengths by a prism, four characteristic violet, blue, green, and red emission lines can be observed, the most intense of which is at 656 nm.
What produces emission lines in a spectrum?
An emission line will appear in a spectrum if the source emits specific wavelengths of radiation. This emission occurs when an atom, element or molecule in an excited state returns to a configuration of lower energy. … The spectrum of a material in an excited state shows emission lines at discrete frequencies.
Why do absorption and emission lines match?
Absorption lines are based on the same physical principle as emission lines: they involve an atom jumping from one particular energy level to another. In this case, however, the jumps must be upwards, from a low level to a higher one.
What causes specific lines to appear in a line spectrum?
Spectral lines are produced by transitions of electrons within atoms or ions. As the electrons move closer to or farther from the nucleus of an atom (or of an ion), energy in the form of light (or other radiation) is emitted or absorbed.…
What is an absorption spectrum definition?
Definition of absorption spectrum : an electromagnetic spectrum in which a decrease in intensity of radiation at specific wavelengths or ranges of wavelengths characteristic of an absorbing substance is manifested especially as a pattern of dark lines or bands.
Why are atomic emission spectra lines whereas molecular emission spectra are normally bands?
Because atoms have quantized energy levels. … Since their energy levels are quantized (i.e. they correspond to the principal quantum number n=1,2,…,N where N is an integer), they give rise to discrete peaks in their spectra, i.e. lines, when these excited electrons relax to their ground state energy levels.
What are the black lines in absorption spectra produced by Mcq?
What are the black lines in absorption spectra produced by? The absorption of light as electrons move from higher energy levels to lower energy levels. The emission of light as electrons move from lower energy levels to higher energy levels.
What is the difference between emission spectra and absorption spectra?
The main difference between emission and absorption spectra is that an emission spectrum has different coloured lines in the spectrum, whereas an absorption spectrum has dark-coloured lines in the spectrum.
What are continuous emission and absorption spectra how are they produced?
Continuous spectra (also called thermal or blackbody spectra) arise from dense gases or solid objects which radiate heat. They emit radiation over a broad range of wavelengths, thus the spectra appear smooth and continuous.
What conditions produce an emission line spectrum quizlet?
The emission line spectrum is produced by electrons jumping up in energy level, while the absorption line spectrum is produced by electrons jumping down in energy level. The only way to decide is to make a graph of the intensity of the light at every wavelength, and then analyze the graph carefully.
What do the different colors in a line spectrum represent Why are the spectra for each element unique?
Each element has its own unique line spectrum and is thus referred to as the “fingerprint” for a particular element. The spectra for each element are unique because each element contains differing numbers of electrons and thus different energy levels.
Why are only 4 lines seen in the hydrogen emission spectrum?
Whilst hydrogen has only one electron, there are many shells or energy levels which that electron can transition between. The gaps between those levels are all different, so each has its own frequency resulting in different lines in the spectra.
Why is there no yellow line in the hydrogen emission spectrum?
Each time an electron changes from a higher energy state to a lower one, a package of energy is emitted. Sometimes the energy emitted is visible. There is no transition in the hydrogen atom that results in the emission of yellow light.
Why are there more emission lines than absorption?
In the emission spectrum, the electrons in the energy levels usually start at random energy levels and so there is more of a variety of wavelengths that could possibly be emitted.
When the absorption spectrum is in the form of dark bands this is known as?
When light passes through gas in the atmosphere some of the light at particular wavelengths is scattered resulting in darker bands. These lines came to be known as ‘spectral lines‘ and were cataloged by heating common elements until they produced light and measuring the wavelengths emitted.