What are elements in Group 1 called
 Andrew Campbell
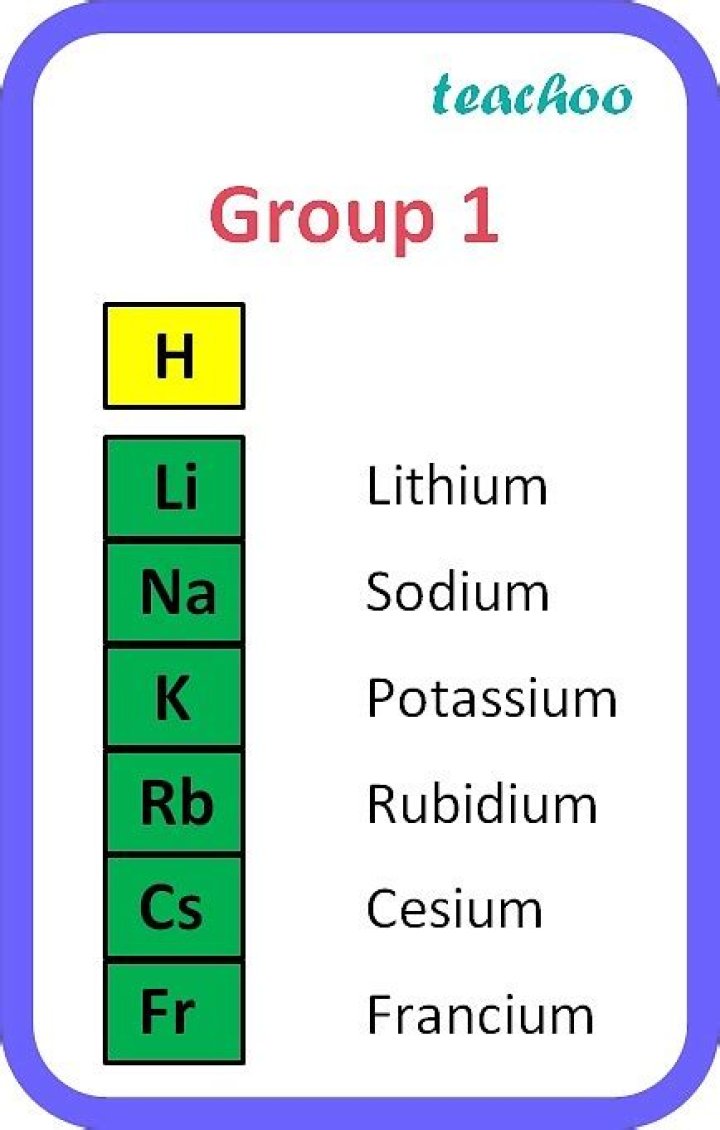
Andrew Campbell The alkali metals are six chemical elements in Group 1, the leftmost column in the periodic table. They are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium
What do we call the elements in Group 1?
Group 1A — The Alkali Metals. Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
What is the first element in Group 1?
Alkali metals are the chemical elements found in Group 1 of the periodic table. The alkali metals include: lithium, sodium, potassium, rubidium, cesium, and francium.
What are the elements of group 2 called?
alkaline-earth metal, any of the six chemical elements that comprise Group 2 (IIa) of the periodic table. The elements are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).Are Group 1 elements metals or nonmetals?
The elements in group 1 are called the alkali metals. The alkali metals share similar physical and chemical properties .
What are Group 3 elements called?
Group III consists of two subgroups: group IIIb and group IIIa. Group IIIa consists of scandium, yttrium, and lanthanium, which is generally considered with the lanthanoids, and actinium, which is classified with the actinoids. Group IIIb, the main group, comprises boron, aluminium, gallium, indium, and thallium.
What is the name of group 7A?
Group 7A (or VIIA) of the periodic table are the halogens: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The name “halogen” means “salt former”, derived from the Greek words halo- (“salt”) and -gen (“formation”).
Why are Group 1 called alkali metals?
The Group 1 elements are called the alkali metals. … All the Group 1 elements are very reactive . They must be stored under oil to keep air and water away from them. Group 1 elements form alkaline solutions when they react with water, which is why they are called alkali metals.What are the Group 17 elements called?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
Why are Group 1 called alkaline earth metals?Group 1 elements are called alkali metals because their oxides are soluble in water and group 2 elements are called alkaline earth metals as their oxides form in the earth and are water soluble.
Article first time published onWhat is the largest element in Group 1?
Trends: DecreasingMelting PointhighestFirst Ionisation EnergylargestElectronegativityhighestGroup 1 ElementsnamelithiumsymbolLi
What properties do Group 1 elements have?
Group one elements share common characteristics. They are all soft, silver metals. Due to their low ionization energy, these metals have low melting points and are highly reactive. The reactivity of this family increases as you move down the table.
What is the property of Group 1?
Alkali metals are soft solids and can be easily cut. Alkali metals have low densities as compared to heavy metals such as iron and copper. Alkali metals are good conductors of heat and electricity. Alkali metals have low melting and boiling points as compared to heavy metals such as copper and iron.
Can Group 1 metals form Coloured compounds?
CompoundIonColourIron(III) oxide, Fe 2O 3Fe 3+Red-brown
What is 1A in periodic table?
Alkali metals belong to group 1A of the periodic table, which includes lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
What is the name of group 8A elements?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
What is group 0 on the periodic table called?
The elements in group 0 are called the noble gases . They exist as single atoms .
What are Group 5A elements called?
Group 5A (or VA) of the periodic table are the pnictogens: the nonmetals nitrogen (N), and phosphorus (P), the metalloids arsenic (As) and antimony (Sb), and the metal bismuth (Bi).
What are Group 11 elements called?
The Group 11 is composed of copper (Cu), silver (Ag), and gold (Au). Silver and Au belong to the so-called noble metals. All these metals occur in variable oxidation stages, mainly +1, and +2, (Table II-11.1).
What are Group 15 elements called?
nitrogen group element, any of the chemical elements that constitute Group 15 (Va) of the periodic table. The group consists of nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and moscovium (Mc).
What are Group 18 elements known as?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
What is a group of 14 called?
Why is group 14 called crystallogens? – Quora.
What element is in Group 4 Period 5?
It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf).
What is the reactivity of Group 1 elements?
The reactivity of Group 1 elements increases as you go down the group because: … the outer electron gets further from the nucleus as you go down the group. the attraction between the nucleus and outer electron gets weaker as you go down the group – so the electron is more easily lost.
Which statement about the elements in group 1 is correct?
Option A is correct: Hydrogen is evolved when they react with water. Explanation: Group 1 elements in the periodic table are known as Alkali metals. The members of this group are Hydrogen (H), Lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr).
What is the most active metal in Group 1?
Statement 1: Lithium is the most active metal in the first group of the Periodic Table.
Why are the elements in Group 1 of periodic table called alkali metals '? Mention the symbol of a post Uranium element?
The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids). Uranium is a chemical element with the symbol U and atomic number 92. … A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons.
What type of chlorides are formed by the elements of Group 1?
- LiCl.
- NaCl.
- KCl.
- RbCl.
- FrCl.
What are the physical and chemical properties of Group 1 elements?
- shiny.
- soft.
- silvery.
- highly reactive at standard temperature and pressure.
- readily lose their outermost electron to form cations with a charge of +1.
What kind of structure does Group 1 form?
Group 1 – the alkali metals They must be stored under oil to keep air and water away from them. When they react with water, Group 1 elements form metal hydroxides which are alkaline (with a pH above 7).
Is Caesium a metal?
Caesium is a soft, gold-coloured metal that is quickly attacked by air and reacts explosively in water. The most common use for caesium compounds is as a drilling fluid.