What are eclipsed and staggered conformations
 Rachel Young
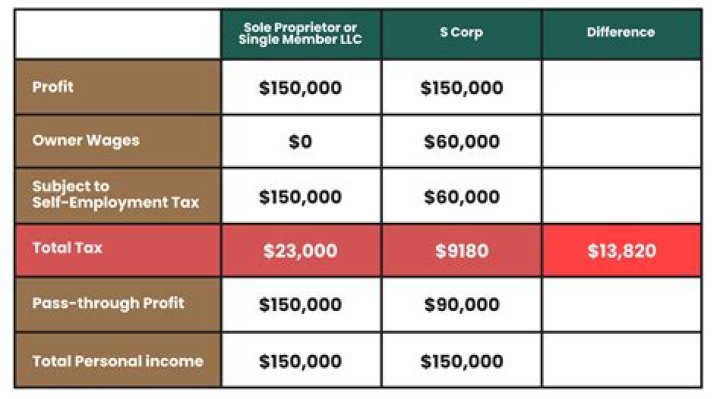
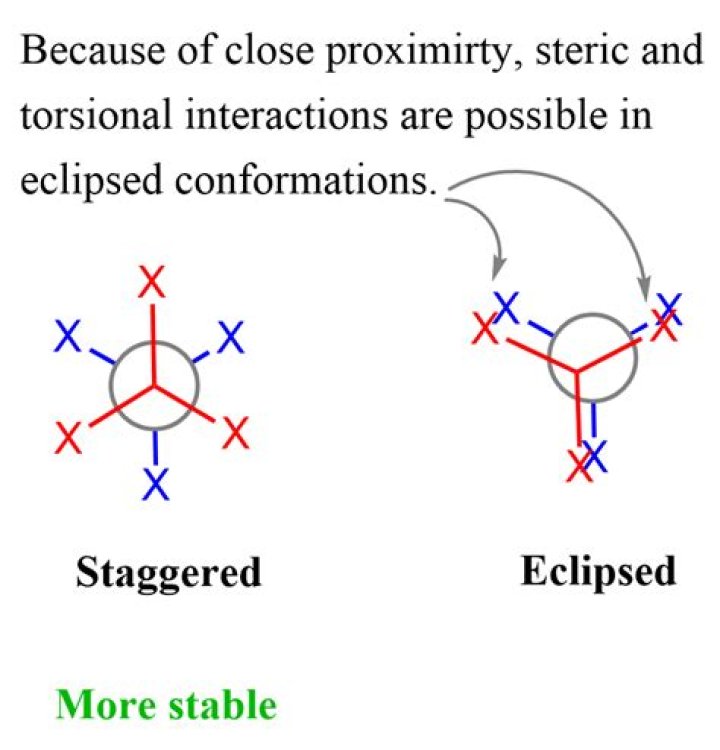
Rachel Young In an eclipsed conformation the carbons are aligned so that the hydrogens are lined up with each other. This creates steric hindrance between them. In a staggered conformation the atoms are all equally spaced from each other.
What is a eclipsed conformation in chemistry?
In chemistry an eclipsed conformation is a conformation in which two substituents X and Y on adjacent atoms A, B are in closest proximity, implying that the torsion angle X–A–B–Y is 0°.
What are staggered and eclipsed conformations of alkanes?
Molecules with different conformations are called conformational isomers or conformers. The two extreme conformations of ethane coming from the C-C rotation shown above are: the staggered conformation with all of the H atoms spread out, and the eclipsed conformation with all of the H atoms overlapped.
What is meant by staggered conformation?
In organic chemistry, a staggered conformation is a chemical conformation of an ethane-like moiety abcX–Ydef in which the substituents a, b, and c are at the maximum distance from d, e, and f. This requires the torsion angles to be 60°.How do you tell if a molecule is staggered or eclipsed?
These structures give the structure of a molecule when looked through the carbon backbone from front-back direction. The main difference between staggered conformation and eclipsed conformation is that staggered conformation has a lower potential energy whereas eclipsed conformation has the maximum potential energy.
What is partially eclipsed conformation?
The partially eclipsed structure of n-butane is. the dihedral angle between two methyl group is 120∘. The dihedral angle between two methyl group in anti-staggered conformation of n-butane is 180∘. The dihedral angle between two methyl group in fully-eclipsed conformation of n-butane is 0∘.
Is staggered or eclipsed conformation more stable?
…with respect to the other—the eclipsed conformation is the least stable, and the staggered conformation is the most stable. The eclipsed conformation is said to suffer torsional strain because of repulsive forces between electron pairs in the C―H bonds of adjacent carbons.
In which type of projection we can get staggered and eclipsed conformations?
A sawhorse projection can reveal staggered and eclipsed conformations in molecules.Why is eclipsed conformation less stable?
The ethane molecule depicted here is in the eclipsed conformation. This is the least stable form of ethane. It is least stable because there is a minimum distance between electron densities of C-H bonds, and therefore a maximum amount of electron-electron repulsion. This interaction is called torsional strain.
How many staggered conformations are there?This process can be continued all around the 360°circle, with three possible eclipsed conformations and three staggered conformations, in addition to an infinite number of variations in between.
Article first time published onWhat are alkane conformations?
Conformation of Alkanes deals with the isomers of alkanes that form due to slight changes in their structure, especially in their carbon-carbon bonds. The conformations start with ethane and occur in all alkanes higher than ethane.
What are conformations in chemistry class 11?
Due to this rotation, different spatial arrangements of carbon atoms in space are observed which can change into one another. Such spatial arrangement of carbon, hydrogen atoms which can be converted into one another by rotation around a C-C single bond is called confirmation or conformer or rotamer.
What is conformation in chemistry?
conformation, any one of the infinite number of possible spatial arrangements of atoms in a molecule that result from rotation of its constituent groups of atoms about single bonds.
What is the energy difference between the eclipsed conformations?
The energy of the eclipsed conformation is approximately 3 kcal/mol (12 kJ/mol) higher than that of the staggered conformation. Torsional strain (or eclipsing strain) is the name give to the energy difference caused by the increased electrostatic repulsion of eclipsing bonds.
What is meant by skew conformation?
1 any conformation of a six‐membered ring form of a monosaccharide or monosaccharide derivative when three adjacent ring atoms and the remaining nonadjacent ring atom are coplanar.
Why is the eclipsed conformation higher in energy than the staggered conformation?
The staggered conformation of ethane is a more stable, lower energy conformation than the eclipsed conformation because the eclipsed conformation involves unfavorable interactions between hydrogen atoms. Specifically, the negatively charged electrons in the bonds repel each other most when the bonds line up.
Is Gauche the same as staggered?
In Staggered Conformation, no group lies parallel to another one in a compound. In Gauche form, More bulky and less bulky groups lie parallel to each other.
Does eclipsed conformation have steric strain?
Steric strain is higher in the case of eclipsed conformation because the bonds are in the closest position to each other, however, it is the least in the case of staggered since the bonds are the furthest apart.
What is staggered in organic chemistry?
Illustrated Glossary of Organic Chemistry – Staggered. Staggered (staggered conformation): Two atoms and/or groups whose dihedral angle is 60o, or close to 60o. In other words, their bonds to the axis of rotation are not aligned. Also a conformation which contains this arrangement.
What is staggered Newman projection?
A staggered conformer in a Newman projection is one in which the bonds from the rear carbon bisect the angles of the bonds in the front carbon. … This puts the atoms on the rear carbon at the maximum distance from those in the front. It also minimizes the van der Waals repulsions between groups.
What is torsional strain?
Torsional strain is the resistance to bond twisting. In cyclic molecules, it is also called Pitzer strain. Torsional strain occurs when atoms separated by three bonds are placed in an eclipsed conformation instead of the more stable staggered conformation.
What eclipsed means?
transitive verb. : to cause an eclipse of: such as. a : obscure, darken. b : to reduce in importance or repute. c : surpass her score eclipsed the old record.
Why staggered conformation is stable?
The increase in potential energy is due to the repulsion between electrons in the bond. This increase in the potential energy is known as the torsional strain. … Thus, the staggered conformation is more stable than the eclipsed conformation because staggered conformation has no torsional strain.
Which is the most stable staggered conformation?
In the most stable conformation, the two methyl groups lie as far apart from each other as possible with a dihedral angle of 180 degrees. This particular staggered conformation is called anti. The other staggered conformation has a Me-Me dihedral angle of 60 degrees and is called gauche.
What are Fischer projections used for?
A Fischer projection or Fischer projection formula is a convention used to depict a stereoformula in two dimension without destroying the stereochemical information, i.e., absolute configuration, at chiral centers.
What is the difference between sawhorse and Newman projections?
The key difference between Newman and Sawhorse projection is that Newman projection is the side-on appearance of a molecule, whereas Sawhorse projection is the angular appearance of a molecule. The Newman projection and the Sawhorse projection are important in determining the conformations of alkanes.
Is a sawhorse a projection?
Sawhorse projection: A representation of molecular structure, from an oblique angle. Somewhat similar to Newman and zig-zag (Natta) projections. The sawhorse projection is named after its similar appearance to a carpenter’s sawhorse.
Where is the lowest energy chair conformation?
To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
What are the conformations of alkanes and cycloalkanes?
Cycloalkanes have one or more rings of carbon atoms, and contain only carbon-hydrogen and carbon-carbon single bonds. The naming of cycloalkanes follows a set of rules similar to that used for naming alkanes. … Larger rings like cyclohexane, have much lower ring straing because they adopt non-planar conformations.
What is the conformation of butane?
Generally, Butane has four conformation isomers which are fully eclipsed, gauche, eclipsed, and anti butane conformational isomers. Lets us discuss these isomers below. When we look at the chemical structure of butane, we can see that it has two substituents which are methyl groups attached to the two end carbon atoms.
What are configurational stereoisomers?
Stereoisomers: Two molecules with the same constitution but different stereochemistry. … Configurational Isomers: Two molecules with the same constitution but different configuration (i.e. – same IUPAC name for constitution, different IUPAC prefix for configuration).