Its true that ZnO is more soluble in water than in ethanol. On the other hand, though alcohal promotes the conversion of hydroxide to oxide, if there is no water in the reaction mixture, the hydrolysis process would be slow..
Keeping this in view, what is zinc oxide soluble in?
Chemical properties Zinc oxide is an amphoteric oxide. It is nearly insoluble in water, but it will dissolve in most acids, such as hydrochloric acid: Solid zinc oxide will also dissolve in alkalis to give soluble zincates: ZnO + 2 NaOH + H2O → Na2[Zn(OH)4]
One may also ask, why is ZnO insoluble in water? Morphology of ZnO nanoparticles has no effect on its solubility in water. Actually small sized ZnO nanoparticles are easily dispersed in water as compared to large particles. But remember as size decreases, the aggrigation process increases and particles settles immediately.
Similarly, you may ask, is acetone soluble in ethanol?
For example, ethanol is miscible with both water and hexane. Substances composed of small polar molecules, such as acetone and ethanol, are usually soluble in water.
How do you dissolve zinc?
Yes, zinc dissolves in hydrochloric acid by chemical reaction. Zinc being an active metal readily reacts with HCl acid at room temperature liberating bubbles of hydrogen gas. The other product is the salt zinc chloride, which is highly soluble in water.
Related Question Answers
What are the side effects of zinc oxide?
Side Effects - hives.
- itching.
- skin rash.
Is zinc soluble in water?
Zinc is insoluble in water but reacts readily with non-oxidising acids, forming zinc (II) and releasing hydrogen. It also dissolves in strong bases. Water solubility of zinc compounds varies, with zinc acetate, zinc nitrate, zinc sulfate, zinc chloride, zinc chlorate and zinc perchlorate all readily soluble in water.Is zinc oxide soluble in oil?
Zinc Oxide is not soluble in water, thus it requires an oil or other such carrier agent in order to function effectively. Zinc Oxide USP functions as a(n): Emollient. Lubricant.How do you clean up zinc oxide?
How to Clean Zinc Oxide Corrosion From Metal Surfaces - Remove any items sitting on top of the galvanized metal surface.
- Scour the stain away with a nylon scrub brush.
- Pour a cleaning product directly onto the stain if it does not disappear after using the nylon brush.
- Scrub the cleaning product into the stained area with the nylon brush.
Is zinc oxide absorbed well?
Zinc oxide appears to be less well absorbed than other zinc compounds when given without food and may be minimally absorbed by some individuals.What type of reaction is zinc oxide and hydrochloric acid?
Zinc oxide reacts with hydrochloric acid to form zinc chloride and water. Zinc oxide is insoluble in water.What is the pH of zinc oxide?
7.2
What can zinc oxide be used for?
Zinc oxide topical (for the skin) is used to treat diaper rash, minor burns, severely chapped skin, or other minor skin irritations. Zinc oxide rectal suppositories are used to treat itching, burning, irritation, and other rectal discomfort caused by hemorrhoids or painful bowel movements.Is acetone the same as ethanol?
The key difference between acetone and ethanol is that acetone is a ketone whereas ethanol is an alcohol. Acetone is an organic compound with the formula (CH3)2CO while ethanol is a simple alcohol with the chemical formula C2H6O.Is ethanol polar or nonpolar?
Ethanol is a very polar molecule due to its hydroxyl (OH) group, with the high electronegativity of oxygen allowing hydrogen bonding to take place with other molecules. Ethanol therefore attracts polar and ionic molecules. The ethyl (C2H5) group in ethanol is non-polar. Ethanol therefore attracts non-polar molecules.What is the difference between acetone and pure acetone?
Acetone is a powerful solvent that removes nail polish quickly and easily, but can be drying to the cuticles. Non-acetone polish removers contain ethyl acetate or nethyl ethyl keytone as their active ingredient.Is ethanol miscible in water?
Ethanol has a 2 carbon chain and a OH group. As water is polar it attracts OH group. Because of the strength of the attraction of the OH group, first three alcohols (methanol, ethanol and propanol) are completely miscible. They dissolve in water in any amount.Is acetone polar or nonpolar?
Acetone molecules are polar because of the positive and negative charges formed by their carbonyl group. The molecules that compose acetone do have nonpolar covalent bonds within their overall structure, such as their carbon to hydrogen and carbon to carbon bonds.What is acetone soluble?
Acetone is a colorless, mobile, flammable liquid readily soluble in water, ethanol, ether, etc. , and itself serves as an important solvent.What are common solvents?
Solvent Molecules A solvent is a liquid that dissolves a solute. The solvent is the component of a solution that is present in greater amount. Perhaps the most common solvent in everyday life is water. Many other solvents are organic compounds, such as benzene, tetrachloroethylene, or turpentine.Does oil dissolve in ethanol?
The solubility of vegetable oils in aqueous ethanol depends on the concentration of alcohol and temperature of the system. In 95% ethanol they are miscible between 90° and 100°C. Addition of a good solvent, like n-hexane, increases the solubility of oils, and the solubility temperatures are lowered.Why is ethanol used as a solvent?
Because ethanol can be produced by fermentation, this is a useful way for countries without an oil industry to reduce imports of petrol. Ethanol is widely used as a solvent. It is relatively safe, and can be used to dissolve many organic compounds which are insoluble in water.Why ZnO is amphoteric in nature?
Zno is an amphoteric oxide,because it can react with both acids and bases. while in general most oxides only react with acids alone due to its basic nature. Amphoretic oxides contradicts the Bronsted –Lowrey theory of acids and bases. Al2O3,SnO and ZnO are typical examples for amphoteric oxides.Why zinc oxide is back titrated?
Assay of zinc oxide is a type of back titration because its reaction with sulphuric acid is slow. Excess of sulphuric acid is added to the reaction mixture and excess of acid is back titrated with sodium hydroxide. 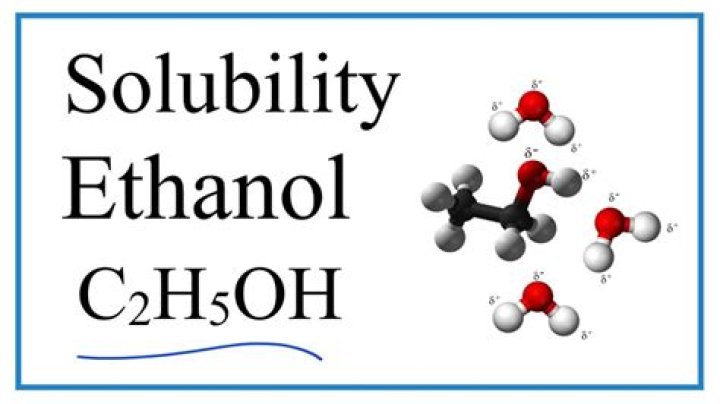
 Christopher Martinez
Christopher Martinez 




